LAUNDRY AGENTS, STAIN REMOVAL, OCCUPATIONAL HAZARDS AND ENVIRONMENTAL CONCERN
|
|
|
- Ann Adams
- 5 years ago
- Views:
Transcription
1 LAUNDRY AGENTS, STAIN REMOVAL, OCCUPATIONAL HAZARDS AND ENVIRONMENTAL CONCERN 1. Introduction Laundry agents or aids are the materials used to improve laundering results (bleaches, optical whiteners) or to accomplish specific functions or effects (soaks, stain removers, softeners and stiffeners). The important laundry agents or aids are water, laundry soap, detergents, stiffeners, bleaches, alkaline agents, acid agents, organic solvents and absorbents. 2. Objective: After learning this module you able to 1. Uses of laundry agents, stain removal, dry cleaning, 2. Discuss the handling guest laundry and valet service 3. Explain the importance occupational hazards and environmental concern in the laundry departments. 3. LAUNDRY AGENTS WHAT IS USED IN THE LAUNDRY? A. WATER: water is the most important material for the laundry process. It just be available in large quantities in the laundry. Hard water should not be used because the compounds in hard water react with soap or detergent. Which form a thick paste that redeposits on the fabric and made the fabric look dull and dirty. Only soft water should be used for the laundering process; if hard water has to be used, it must be chemically treated to free it from compounds before using. B. SOAP: Soap is technically also a type of detergent is being used from ancient period. In older times, ashes of wood fire under the cooking pot were mixed with left over fat which was then used for washing clothes. Now a-days soap is made of fatty acid and alkali combined at different temperature. Soap-free detergents have properties similar to soap-such as foaming, wetting, and cleaning- but they are able to make soluble salts out of the calcium, magnesium, and other metal salts that make water hard and render ordinary soap insoluble. While selecting laundry soaps, the following criteria must be kept in mind: 1
2 The soap should be a clear, pale colour as dark-coloured soaps may contain impurities that aren t easily visible. The soap should feel firm when pressed if it is soft, it may contain excessive amounts of water and will be wasteful in use. C. SYNTHETIC DETERGENTS: Detergent (cleaning agent) refers to mixtures of chemical compounds including alkylbenzenesulfonates, which are similar to soap but are less affected by hard water. While rinsing, attention must be given so that no trace of detergent remains in the fabric because they may react with the fibers and damage the clothes. Clothes should not be dipped in synthetic detergent for a long time as soiling or dirt gets redeposited. D. ALKALI: Alkali gives better results if used with soap and detergent. Alkali is added before adding soap or detergent as jut helps in converting hard water into soft water. For cotton fabric, strong alkali should be used while for wool, silk or similar fabric mild alkali should be used. Common alkalis used are washing soda, sodium phosphate, sodium hydroxide, sodium metaslicate. 4. STIFFENERS AND SOFTENERS Fabric softeners are added with sours in the final wash cycle BLEACHES These are chemicals capable of whitening fabrics and removing stains by destroying pigmented matter. Various type of bleaches and the fabric to be used safely is indicated in table -1 TABLE 1- Types of bleaches and their uses Name of the bleach Nature Fabrics on which used Bleaching powder Sodium chlorate Sodium chlorite Sodium hypochlorite Sodium perborate Sodium percarbonate Sodium peroxide Cotton Cotton Cotton Cellulosic All Cellulosic Cellulosic 2
3 Hydrogen peroxide Potassium permanganate Sodium hydrosulphide Sulphurous acid Oxalic acid Reducing Reducing /reducing All Cotton Proteinic Proteinic Cellulosic and proteinic 4.2. TYPES OF BLEACH Bleach can be classified into the following categories: A. OXIDIZING BLEACHES: These bleaches release oxygen, which combines with the stains to form a colorless compound. The bleach left in contact with the fabric until the stain is removed, or else the fabric will be weakened. B. OPEN AIR AND SUNLIGHT: This is the world s oldest and cheapest method of bleaching. Hanging clothes out in the sun to dry keeps white clothes sparkling. C. SODIUM HYPOCHLORITE (JAVELLE WATER): This is made using the following ingredients: 277g washing soda; 57 g chloride of lime; ½ liter boiling water and 1 liter cold water. To make Javelle water, dissolve the washing soda in boiling water, mix the chloride of lime with cold water, allow settling and straining off the clear liquid without stirring and then mix this filtrate with the washing soda solution. Allow the precipitate of calcium carbonate that is formed to settle and again strain off the clear liquid and store the residue in dark-colored bottles as it is unstable to light. Used to bleach white cottons and linen and never on any other fabric. Javelle water should not be used in silk and wool as these fabrics are dissolved on it. Dilute the Javelle water with an equal amount of hot water and dip the stain into the bleach until it is removed. A small amount of ammonia in the rinse water will help to remove the smell of bleach from the fabric. D. SODIUM CHLORIDE: This is suitable for cellulosic and synthetic fibres. It is a finely divided crystalline powder obtained when chlorine peroxide reacts with sodium peroxide.sodium chloride is used in its anhydrous form for bleaching. The bleach is not hygroscopic and is extremely stable. Cold aqueous solutions have only a mild oxidizing power. One heating or acidification, they develop a strong oxidizing power. 3
4 E. HYDROGEN PEROXIDE: This is an effective bleach, yet not harmful to most fabrics. Teaspoon of concentrated ammonia solution or of sodium perborate may be added to each gallon of the diluted solution to make the action stronger. F. SODIUM PERBORATE: This is made from borax, caustic soda and hydrogen peroxide. It is used in many oxygen washing powders. To prepare the bleach, sodium perborate is dissolved in water. G. POTASSIUM PERMANGANATE: This has a high content of oxygen which enables it to combine with and remove obstinate stains such as perspiration and mildew. It can be used on animal as well as vegetable fibres. During bleaching, the articles will be stained a characteristic brown due to the formation of manganese dioxide. This is removed by dipping the fabric in one of the following solutions: Sodium hypochlorite Oxalic acid (diluted) Two volumes of hydrogen peroxide acidified with a teaspoon of vinegar (acetic acid) to one volume of bleach. The brown stain disappears almost at once. The fabric is then thoroughly rinsed. H. BLEACHING POWDER: This has been the traditional bleach for cotton fabrics. It has now been superseded by sodium chloride and other bleaches. A good bleaching powder contains 38% available chlorine. Bleaching powder solutions are alkaline in nature due to the presence of free lime. I. REDUCING BLEACHES: Reducing bleaches work by removing oxygen from the colouring matter of the stain. J. SODIUM HYDROSULPHITE: This is a valuable agent for bleaching all fibres, particularly wool and silk which cannot be treated. Sodium hydrosulphite should be stored in an airtight container in a moisture free place away from heat as it can decompose by absorbing oxygen and evolving sulphur dioxide gas. It can be used in its concentrated form to remove spots caused by grass, faecal matter, leather polish, mildew, ink, potassium permanganate and dye stains. K. SODIUM BISULPHITE: This is a mild reducing agent produced by the partial neutralization of sulphurous acid by caustic soda. Neutralization or thorough rinsing must follow 4
5 the bleaching process, otherwise sulphuric acid will appear in the fabric through the action of oxygen. L. SODIUM THIOSULPHATE: This is bleach for cottons, it is made by mixing 7 gm sodium thiosulphate, 3.5 kg of 36% acetic acid and 8 cups of water. M. OVER BLEACHING: The over bleaching of cotton and linen during laundering is one of the main causes of general weakening of the fabrics. The fibres then become brittle and harsh, emitting a distinct crackle when rubbed together to guard against over bleaching, always use a bleach of known strength. Keep the temperature below 60 o C (140 o F) and add measured quantities of dilute bleach gradually. Chlorine bleach especially should not be used at temperature above 71 o C (160 o F). 5. THE RIGHT WAY TO BLEACH 5.1. NEVER USE BLEACH DIRECTLY ONTO THE FABRIC: Never pour bleach, whether liquid or powder, directly onto the fabric. It must first be diluted in a small container and then added to the water in the tub before the fabrics are immersed. Fabric is pre-soaked in bleach for minutes. Hot water and agitation help to dissolve the bleach and hasten its action USE THE RIGHT AMOUNT OF BLEACH: Bleach should be used according to the manufacturer s directions or as per the guidelines given above. All measurements should be precise WASH OFF THOROUGHLY: Washing the fabrics thoroughly after bleaching is important because if the bleach remains in the fabric it will weaken and degenerate ANTICHLORS: These are used during the after-wash/bleach rinse to ensure that all the chlorine in the bleach has been removed ALKALINE AGENTS/ALKALINE BUILDERS Some alkaline agents/builders used in laundering are discussed below: 5.6. WASHING SODA (SODIUM CARBONATE, NA 2 CO 3.10H 2 O): This is the most commonly used additive in detergents. It is usually purchased in the form of soda crystals that readily dissolve in boiling water. 5
6 5.7. BORAX (sodium tetra carbonate, Na 2 B4O 7.10H 2 O): This compound occurs naturally and is sold as a white powder. It is a mildly alkaline substance, readily soluble in cold water and can be used safely on any fiber AMMONIUM HYDROXIDE (NH 4 OH): This is sometimes purchased as a concentrated solution. It must be used with care as its pungent vapors may cause coughing and choking. 6. ACIDS AGENTS These are useful for neutralizing alkalis and for stain removal. Mild acids used to neutralize any residual alkalinity in fabrics after washing and rinsing are called sours 6.1.OXALIC ACID : This is a poison and should be kept in jar labeled as such. It is sold in the form of white crystals. Its uses include the removal of obstinate fruit stains, bleaching of brown stains left after the use of potassium permanganate and removal of the tannin base of ink stains together with hydrogen peroxide, 6.2. SALT OF LEMON: This is a compound of potassium oxalate and oxalic acid, referred to as potassium binoxalate. 7. ORGANIC SOLVENTS them. Solvents are applied to the most delicate fabrics either to remove stains or to dry-clean 7.1. CLEANING BENZENE (C 6 H 6 ) or PETROL: This is obtained from the distillation of shale oil or petroleum. It is highly inflammable and should not be kept or used indoors in large quantities; of course, it must never be used, even in small quantities near an open fire either CARBON TETRACHLORIDE (CCl 4 ): This is more expensive than cleaning benzene, but is similar in action and has the advantage of being non-flammable. It is, however, very toxic and should be used only near an open window or in well-ventilated rooms as it is extremely volatile and evaporates quickly ACETONE: This is a useful solvent for many stains. Acetone is an effective spot-cleaning agent for stains caused by cosmetics, nail polish, lipstick, paint, varnish, and shoe polish. Acetone is highly inflammable METHYLATED SPIRIT: This is ethyl alcohol (C 2 H 5 OH) mixed with methyl alcohol (CH 3 OH), which makes it poisonous. 6
7 7.5. PARAFFIN: This is mixture of hydrocarbons. It us a white, waxy solid obtained as a residue from the distillation of petroleum and shale TURPENTINE (C10H16): This is solvent is more expensive than paraffin. It has a distinctive smell and is both inflammable and volatile. It acts as a solvent for grease, varnish, paint and printers ink. 8. ABSORBENTS These are substances suitable for removing grease spots from all fabrics and for the general cleaning of light-colored fabrics that are evenly soiled. 8.1 ph SCALE FOR LAUNDRY AGENTS This is a scale used to determine the ph of the sud in the washing so that it that may be possible to adjust the ph of the sud with the use of chemicals to maximize cleaning efficiency. ph scale for laundry agents STAIN REMOVAL Stain is mark or discoloration caused by the absorption of foreign distance on a textile. Stain removal method aims at treatment and removal of coloured spot from the fabric.stain removal or spot cleaning is a skill that demands special attention,specific techniques and exprenice IDENTIFICATION OF A STAIN Sight: By looking carefully at the stain, to see if it s absorbed or built up stain. Odour: To smell the stain, especially after giving it a light steam treatment, we come to know what they of stain it is. 7
8 Colour: It also gives an important clue of the nature of stain and the agents causing it. Feel: Ton run the hand/fingers over the stain to feel its stiffness, especially of a built- up stain. Location: it helps to pinpoint the cause of stain. Perfume stains with usually be located on the lapels of garments or under the arms CLASSIFICATION OF STAINS Animal stains Vegetable stains Chemical stain (blood, eggs, milk, meat, (tea, coffee, coco, fruit juice, (Nail polish, rust, perspiration, urine and faeces) henna betel leaf) grease, acids, etc) 9.3. PRINCIPLES OF STAIN REMOVAL Stains should be treated immediately so as to prevent them from spreading as we as from being absorbed by the fabric. The action involves sponging or washing the stain with water or mild detergent solution GENERAL RULES OF STAIN REMOVAL 1. Always treat the stain immediately. 2. If a stain is not removed after immediate action, the specific removal may be required. Before attempting specific removal first ascertain the nature of the fabric, the nature of the stain, age of the stain, colour of the fabric, etc. 3. Select appropriate stain removal reagents and methods accordingly. Before attempting stain removal, check the reaction on the fabric and the reagent also. 4. Always start with the mildest method and reagent, gradually progression to stronger methods and reagents. 5. Stretch the stain over a pad of clean white absorbent cloth with a piece of clean cotton wool, apply the reagent on the stain. 8
9 6. Start applying the reagent to the outer edge of the stain and work toward the centre. This will prevent the stain from spreading as well as formation of a ring. 7. Sponging is preferable to soaking and rubbing with the reagent. 8. Rubbing and washing instantaneously is preferred to soaking and the washing. 9. Always neutralize the effect of every chemical used by thoroughly rinsing the reagent at each step before attempting the use of another method or reagent. 10. The need of professional help if required should be ascertained at the time of stain removal. 9.5.GENERAL RULES FOR REMOVAL OF UNKNOWN STAIN 1. Soak stain in Luke warm salted for minimum half an hour. 2. If stain persists, wash in luke warm synthetic detergent solution. 3. If stain still persists, wash in luke warm enzyme detergent solution. 4. If stain still persists, use hot synthetic detergent solution to activate solution per borate, Which is an oxidizing bleaching agent. 5. Use solvent on still persisting stains. 6. If stain is not removed work with mild acidic solution progressing towards strong acidic solution. 7. If stain does not go, use alkaline progressing towards strong alkaline solution. 8. If stain sill persists, appropriate bleaching treatment should be done IMPORTANCE POINTS TO BE KEPT IN MIND 1. After every step, thoroughly rinse in water to neutralize the effect of each cleaning agent. 2. Temperature may be adjusted accordingly keeping in mind the nature of fabric and the stain. Type of chemicals used and the time of application should be accordingly to the nature of the fabric 9
10 10. CLASSIFICATION OF STAIN REMOVERS There are two type of stain removal method they are 1. Physical method 2. Chemical method PHYSICAL METHOD a. Reagent used, which absorbs the stain from the fabric surfaced, e.g., chalked powder, fuller s earth, starch pad. b. Use of grease absorbents c. Heat application method used generally on fresh stains CHEMICAL METHOD Basic chemical reagents used to wash stain are salts, solvents, acids, alkalies. Some chemicals can be mild. Flammable and toxic ones have to be used with care and not on plastic, vinyl or nylon, e.g., benzene, acetone, CCl4, amyl acetate etc. These are used for old or stubborn stain, e.g., bleaching method of stain removal. Stain removal agents 10. A. ORGANIC SOLVENTS A Benzene, white sprit, turpentine B Carbon tetra chloride, per chloro ethylene Both type dissolve grease and require care when use group A is inflammable and should not be used near a naked flame. Group B through non inflammable should be used in ventilated areas as they are harmful when they are harmful when they are inhaled and used for removal of chewing gum stain, grease stain, oil, lipstick, ball point stain, etc. 10. B. ACIDS Oxalic acids, potassium oxalate, salts of lemon acid are frequently used as removers. All these are poisonous and must be carefully handled. Acid must be diluted before use. Some dyes are affected by acid; so acid should be used only one fast colours. Washing with detergent are 10
11 through rinsing is essential to neutralize excess acid and prevent any damage to the fabric. They are used for removal of metal stain, rust, blood and iron stain etc. 10. C. ALKALINE Washing soda, ammonia dye and animal fibre are adversely affected by alkalies so they should be used only on cotton and linen fabric used for removal of old and heavy vegetable stains like tea and coffee. 10. D. BLEACHES The process of changing a colored fabric or substance into a colorless one is known as bleaching. As they weaken the fabric, extreme care should be taken while using them. They whiten the coloured substance either by oxidizing or reducing. OXIDIZING BLEACHES They liberate oxygen, which combine in colour pigments to form a colorless compound. a. Sodium hydrochloride, b. Hydrogen peroxide c. Sodium per borate A. SODIUM HYDROCHLORIDE: This is household bleach which damages animal fiber; it must not be used on silk and woolen articles used for removal of obstinate stain on cotton and linen fabrics, it fixes iron and rust stain. All articles should be thoroughly rinsed with water. B. HYDROGEN PEROXIDES: This is a lower acting that the above it decomposes readily after adding little ammonia to the solution can be used on white fabric. C. SODIUM PER BORATE: It is a safe and quick acting agent present in powered soap and soap less detergent. 11
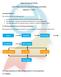
12 11. HANDLING GUEST LAUNDRY Guest Laundry desk attendant Valet Calls (assigns valet) (proceeds to room) Spotter Sorter/Marker Valet & Guest (Remove strain ) (Sorts & marks) (Tally laundry) Washer man / Drycleaner Pressman Packer (Wash or dry clean) (Iron clothes) (Packs or hangs) Guest Desk attendant Valet (Receives clothes) (Get involves) (To delivers clothes) 11. A. GUEST CALLS GUEST CYCLE Dealing with guest laundry is one of the major responsibility of the house keeping department. Guest may avail of laundry service by their telephoning the housekeeping desk and room attendant is to sent to collect the laundry, if the laundry is on premise valet runner will collect the laundry. Generally laundry must picked up on time, laundered and delivered back to the guest on time without any mix ups.usually guest laundry clothes collected in the morning and given back evening. In hotels, which offer this service, the lists are left in the bedroom, together with laundry bags..the guest is expected to place the soiled linen in the laundry bags,fill the laundry forms and handover to valet. 11. B. VALET SERVICE Most hotel provide valet service for the to and fro transfer of guest laundry.valet collect the soiled guest laundry bag and laundry list. Valet verify himself against the list if they does not match, the guest is requested by the valet to rectify the mistake.valet are the responsible for returning the clean guest clothes. 12
13 The usual laundry service for in house guests is referred to as normal service and entails laundry being collected from guest before 10.a.m and being returned by in evening. On week days,there are no additional charges for this service apart from the rate specified in the laundry list. On Sundays and holidays,the normal service rate is charged at 25 % extra.hotels have a provision for an express service, which takes about 2-3 hours,guests are usually paid 50 % extra or double the laundry list rate. 12. Occupational Hazards The most common accidents in industrial laundries involve chemical exposure, sharp objects left in soiled linen, slips from wet floors, exposure to pathogens in contaminated linen and body parts being stuck in machinery. While these problems can usually be avoided by standard precautions and a little common sense, they can and do happen. Exposure to soil and pathogens can be limited by two things. One, of course, is the use of personal protective equipments like barrier gowns, gloves, eyewear, foot coverings and face masks. These items should be worn when handling soiled linen. Infections can also be eliminated by proper hand washing with antibacterial soap. Employees should wash their hands after handling any linen, whether soiled or clean. Since the noise levels in industrial laundries can be quite high, earmuffs and disposable earplugs are often issued, hearing protection is essential. Exposure to chemicals is also common. Since washers require a constant stream of detergent, bleach and other chemicals, the supply must be constantly replenished. Laundries are required to provide a material safety data sheet or MSDS for all chemicals used. Many laundries require their machine operators to be familiar with HAZMAT, if not fully certified. With the constant workload and harsh working conditions, employees can easily become short-tempered. This occasionally leads to problems in work place bullying. For this reason, laundry managers must be competent disciplinarians, ready to deal with employees who have attitude problems. Laundry machines use high pressure steam and dangerous chemicals. Poor maintenance can cause injuries such as severe burns and chemical exposures. Hoses, steam lines and other machine parts must be checked properly and replaced regularly. 13
14 13. ENVIRONMENTAL CONCERNS A more profound problem arises from the heavy use of sodium triophosphate which can comprise up to 50% by weight of detergents. The discharge of soluble phosphates into natural waters has led to problems with eutrophication, or the growth of living things, of lakes and streams, often where it is not desirable. The replacement of sodium triphosphate by zeolites offers some relief to this problem. With respect to the phosphate additives, between 1940 and 1970 "the amount of phosphates in city wastewater increased from 20,000 to 150,000 tons per year." They can also indirectly cause oxygen depletion at greater depths, through microbial breakdown of dead algal cells. Many country started phasing out phosphates in the 1980s in their washing procedures. In 2004, the European Union introduced regulations to require biodegradability in all detergents, and intends to ban phosphates in domestic products from Australia began phasing out the use of phosphates in its detergents in 2011, with an all-out ban expected to take effect in Pursuant to findings published in 2006 by the Shenkar College of Engineering and Design indicating that liquid detergents are "much more environment-friendly" than powdered detergents. Israel's Ministry of the Environment began recommending that consumers prefer liquid detergent over powdered ones for laundry which is not heavily stained. Recent studies have revisited the question of whether existing household phosphate bans are effective in reducing phosphorus concentration in waterways, and subsequent algal blooms. A 2014 case study of Vermont phosphate policies showed that while the bans reduced the phosphate contribution by treated wastewater from households to five percent of the total contribution, algal blooms have still continued to worsen for other reasons. 14. SUMMARY Laundry agents and stain removal or spotting is important functions in the laundry. The section on stain removal is exhaustive as this is a specialized skill. A trial and error method does not work here and there can be no short cuts for the stain removal. It should be understood that the term dry cleaning is misnomer.the actual process dose use liquids apart from the dry powders and agents but these liquids are non aqueous. Dealing with the guest laundry is another important responsibility of the house keeping department,this type of service is done by valets at a stipulated time and need to returned by normal or express service as desired by the guest. Laundry personnel have to interpret certain symbols on the care labels. 14
FABRIC CARE STAIN REMOVAL
 FABRIC CARE GUIDE FABRIC CARE As clothing typically sees the most wear and tear through the laundering process, all Speed Queen products are designed, tested and proven to provide both the highest level
FABRIC CARE GUIDE FABRIC CARE As clothing typically sees the most wear and tear through the laundering process, all Speed Queen products are designed, tested and proven to provide both the highest level
Spot-Cleaning Tips and Remedies
 The best and most convenient way to remove spots and spills is to have your carpet serviced by an Aladdin Professional. However, the following tips and home remedies should help remove most spots and odors.
The best and most convenient way to remove spots and spills is to have your carpet serviced by an Aladdin Professional. However, the following tips and home remedies should help remove most spots and odors.
Surface Materials Cleaning & Maintenance
 Surface Materials Cleaning & Maintenance With regular care and maintenance, your Trendway product will continue to provide years of superior performance and satisfaction. Please follow the finish cleaning
Surface Materials Cleaning & Maintenance With regular care and maintenance, your Trendway product will continue to provide years of superior performance and satisfaction. Please follow the finish cleaning
Carpet Cleaning Guide. Carpet Cleaning 101: An Overview
 Carpet Cleaning Guide Carpet Cleaning 101: An Overview Step One: Identify Carpet Fiber Type Cotton Wool, Silk Nylon Olefin Polyester Acrylic Synthetic Composition Cellulosic Cotton Seed, Jute Protein Based-
Carpet Cleaning Guide Carpet Cleaning 101: An Overview Step One: Identify Carpet Fiber Type Cotton Wool, Silk Nylon Olefin Polyester Acrylic Synthetic Composition Cellulosic Cotton Seed, Jute Protein Based-
How to Remove Stains Many thanks to all who contributed to this list including The Stain Expert.
 How to Remove Stains Many thanks to all who contributed to this list including The Stain Expert. Acids - If the item is washable, spread dry baking soda on the area and dampen with water. Rinse thoroughly
How to Remove Stains Many thanks to all who contributed to this list including The Stain Expert. Acids - If the item is washable, spread dry baking soda on the area and dampen with water. Rinse thoroughly
HOW TO CLEAN AND CARE FOR MARBLE
 HOW TO CLEAN AND CARE FOR MARBLE CONTENTS 3 GET TO KNOW YOUR STONE 4 STAIN AND SCRATCH PREVENTION 6 SEALING 7 STAIN INDENTIFICATION AND REMOVAL 9 FINAL THOUGHTS MARBLE CARE GUIDE 2 GET TO KNOW YOUR STONE
HOW TO CLEAN AND CARE FOR MARBLE CONTENTS 3 GET TO KNOW YOUR STONE 4 STAIN AND SCRATCH PREVENTION 6 SEALING 7 STAIN INDENTIFICATION AND REMOVAL 9 FINAL THOUGHTS MARBLE CARE GUIDE 2 GET TO KNOW YOUR STONE
Uniperol Bleach IT. Technical Information. Europe
 Technical Information TIe/ EU July 2011/I (5/2011)(WJA) Page 1 of 7 First Edition Europe = Registered trade mark of BASF in several countries Uniperol Bleach IT Basic bleaching agent, without optical brightener
Technical Information TIe/ EU July 2011/I (5/2011)(WJA) Page 1 of 7 First Edition Europe = Registered trade mark of BASF in several countries Uniperol Bleach IT Basic bleaching agent, without optical brightener
tech 66 General Statements Regarding the Chemical Resistance of nora Floor Coverings
 This document is written as a general statement to address nora s stain resistance to chemical contamination however facilities must realize that it does not address all the chemicals and chemical compounds
This document is written as a general statement to address nora s stain resistance to chemical contamination however facilities must realize that it does not address all the chemicals and chemical compounds
Procedure: Hazardous Substances and Dangerous Goods Storage and Safe Use
 Procedure: Hazardous Substances and Dangerous Goods Storage and Safe Use Date Description of Original Document/Amendments Prepared/Edited By 09/04/14 Initial Draft Jasmina Lozanovska 30/04/14 Released
Procedure: Hazardous Substances and Dangerous Goods Storage and Safe Use Date Description of Original Document/Amendments Prepared/Edited By 09/04/14 Initial Draft Jasmina Lozanovska 30/04/14 Released
Proper vacuuming requires 3 to 5 passes at slow speed over the same area
 CARPET CLEANING & MAINTENANCE GUIDELINES WOOL & VISCOSE & OR SILK --------- ---------- DAILY CLEANING REGULAR VACUUMING Regular vacuuming of the whole carpet should take place even when the carpet does
CARPET CLEANING & MAINTENANCE GUIDELINES WOOL & VISCOSE & OR SILK --------- ---------- DAILY CLEANING REGULAR VACUUMING Regular vacuuming of the whole carpet should take place even when the carpet does
PARFIX BATHROOM AND KITCHEN
 1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Name: Recommended Use: PARFIX BATHROOM AND KITCHEN Silicone sealant for use on most common building materials. Supplier: DuluxGroup (New Zealand)
1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Name: Recommended Use: PARFIX BATHROOM AND KITCHEN Silicone sealant for use on most common building materials. Supplier: DuluxGroup (New Zealand)
Ultimate stain-removal chart
 realsimple Ultimate stain-removal chart Wipe out any spot, smudge, or splatter with this go-to guide for washable fabrics. AFTER TREATING Wash fabrics as recommended. The sooner, the better! DRINKS COFFEE
realsimple Ultimate stain-removal chart Wipe out any spot, smudge, or splatter with this go-to guide for washable fabrics. AFTER TREATING Wash fabrics as recommended. The sooner, the better! DRINKS COFFEE
Spring Cleaning with Homemade Products
 Spring Cleaning with Homemade Products Try these DIY cleaning products to get your home sparkling clean this spring. As spring approaches, our thoughts are turned to spring cleaning! We ve been cooped
Spring Cleaning with Homemade Products Try these DIY cleaning products to get your home sparkling clean this spring. As spring approaches, our thoughts are turned to spring cleaning! We ve been cooped
Material Safety Data Sheet
 Section 1: Product and Company Identification Product Name Catalog Number Company Cyclic GMP CLIA Kit (High-Sensitivity) SKT-210 StressMarq Biosciences Inc. PO Box 55036 Cadboro Bay Victoria BC V8N4G0
Section 1: Product and Company Identification Product Name Catalog Number Company Cyclic GMP CLIA Kit (High-Sensitivity) SKT-210 StressMarq Biosciences Inc. PO Box 55036 Cadboro Bay Victoria BC V8N4G0
TRIM CLEANING SECTION 10 INDEX INTRODUCTION GENERAL INSTRUCTIONS CLEANING FABRICS WITH VOLATILE CLEANERS. Page
 S TRIM CLEANING SECTION 10 TRIM CLEANING INDEX Subject Page Subject Page Introduction General Instructions Cleaning Fabrics With Volatile Cleaners Cleaning Fabrics With Detergents Precautions for Cleaning
S TRIM CLEANING SECTION 10 TRIM CLEANING INDEX Subject Page Subject Page Introduction General Instructions Cleaning Fabrics With Volatile Cleaners Cleaning Fabrics With Detergents Precautions for Cleaning
SULTRASPOT COLOUR. Application. Physico chemical properties. Properties. Composition & Safety. Use instructions. Storage.
 SULTRASPOT COLOUR Pre-wash and post-wash stain remover for removing tannin stains SULTRASPOT COLOR is an acidic stain remover used for removing tannin and pigment stains from clothes SULTRASPOT COLOR is
SULTRASPOT COLOUR Pre-wash and post-wash stain remover for removing tannin stains SULTRASPOT COLOR is an acidic stain remover used for removing tannin and pigment stains from clothes SULTRASPOT COLOR is
Chemical Name: Dishwasher Detergent. Manufacturer: Cascade. Container size: 20oz. Location: SOC. Disposal: Place empty container in trash.
 Chemical Name: Dishwasher Detergent Manufacturer: Cascade Container size: 20oz. Location: SOC Disposal: Place empty container in trash. Page 1 of 4 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza Cincinnati,
Chemical Name: Dishwasher Detergent Manufacturer: Cascade Container size: 20oz. Location: SOC Disposal: Place empty container in trash. Page 1 of 4 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza Cincinnati,
The Specialist's Free Guide to Stain Removal
 The Specialist's Free Guide to Stain Removal Red Wine, Coffee, and Pet Stains No More! The Specialist Complete Carpet Care Expert Carpet, Upholstery & Area Rug Cleaning (714) 350-9290 info@thespecialistccc.com
The Specialist's Free Guide to Stain Removal Red Wine, Coffee, and Pet Stains No More! The Specialist Complete Carpet Care Expert Carpet, Upholstery & Area Rug Cleaning (714) 350-9290 info@thespecialistccc.com
It Works! Multi Stain Remover Kit with 2 Tubes & 2 Sponges
 It Works! Multi Stain Remover Kit with 2 Tubes & 2 Sponges USE: It Works for clothing stains, carpets, upholstery, car seats, head liners, all types of over garments, leather, vinyl, aluminum rims, jewelry,
It Works! Multi Stain Remover Kit with 2 Tubes & 2 Sponges USE: It Works for clothing stains, carpets, upholstery, car seats, head liners, all types of over garments, leather, vinyl, aluminum rims, jewelry,
Care & Maintenance guide
 Care & Maintenance guide The natural stone you have in your home, office, or commercial building is an investment that will give you many years of beautiful service. Simple care and maintenance will help
Care & Maintenance guide The natural stone you have in your home, office, or commercial building is an investment that will give you many years of beautiful service. Simple care and maintenance will help
SHEPROS. Baby Series BABY DISHWASHER - B-BD 001. Product Description:
 BABY DISHWASHER - B-BD 001 Baby Dishwasher is a water based, low-foaming, non-skin irritating, biodegradable and environmentally friendly cleaner. It is made of powerful Nano Biotech Colloidal Micelles
BABY DISHWASHER - B-BD 001 Baby Dishwasher is a water based, low-foaming, non-skin irritating, biodegradable and environmentally friendly cleaner. It is made of powerful Nano Biotech Colloidal Micelles
Annex A. Composition of laundry detergents
 Annex A Composition of laundry detergents Laundry detergents are composed of a range of ingredients that give the final product its different characteristics. The most common ingredients are listed alphabetically
Annex A Composition of laundry detergents Laundry detergents are composed of a range of ingredients that give the final product its different characteristics. The most common ingredients are listed alphabetically
Safety Data Sheet SANDPIT SANITISER 1. Identification
 Safety Data Sheet SANDPIT SANITISER 1. Identification Trade name of product : SANDPIT SANITISER Other Shipping Name : None allocated Supplier Address : Total Hygiene Solutions 71 Stradbroke Street Heathwood
Safety Data Sheet SANDPIT SANITISER 1. Identification Trade name of product : SANDPIT SANITISER Other Shipping Name : None allocated Supplier Address : Total Hygiene Solutions 71 Stradbroke Street Heathwood
INDEPENDENTLY LABORATORY TESTED
 Bio-Green Cleaning Product Range An Extensive Range of Environmentally Responsible Products A range of BIO-GREEN products have been submitted for independent analysis by Lanfax Laboratories, a premier
Bio-Green Cleaning Product Range An Extensive Range of Environmentally Responsible Products A range of BIO-GREEN products have been submitted for independent analysis by Lanfax Laboratories, a premier
THYME GUARD PAG 1 of 5
 PAG 1 of 5 SECTION 1: PRODUCT AND COMPANY IDENTIFICATION Product Name: Thyme Guard Product Use: Bactericide and fungicide liquid, used as part of a protection program for plants and crops. Supplier: AGRO
PAG 1 of 5 SECTION 1: PRODUCT AND COMPANY IDENTIFICATION Product Name: Thyme Guard Product Use: Bactericide and fungicide liquid, used as part of a protection program for plants and crops. Supplier: AGRO
SPOTTING GUIDE. Rubber or latex based containing metallic oxides and resins. Usually white but darkens with age. Sticky. Remove before drycleaning.
 SPOTTING GUIDE Adhesive Tape Albumin (egg) Animal Stains Asphalt (pitch) Automobile Polish Avocado Rubber or latex based containing metallic oxides and resins. Usually white but darkens with age. Sticky.
SPOTTING GUIDE Adhesive Tape Albumin (egg) Animal Stains Asphalt (pitch) Automobile Polish Avocado Rubber or latex based containing metallic oxides and resins. Usually white but darkens with age. Sticky.
Instrument Cleaning. Dental Clinics
 1 CLEANING FOR INFECTION CONTROL Dental Clinics 2 This presentation is designed to reflect the recommendations given in the following Australian Standards and Guidelines. Throughout this discussion we
1 CLEANING FOR INFECTION CONTROL Dental Clinics 2 This presentation is designed to reflect the recommendations given in the following Australian Standards and Guidelines. Throughout this discussion we
Maintenance Guidelines Bluff
 Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
WHMIS Training Module 2017
 WHMIS 1988 Training Module 2017 Table of Contents 1. WHMIS a. Purpose b. 3 Main Components of WHMIS c. WHMIS Communication System d. Enforcement of WHMIS in Ontario 2. Responsibilities, Hazardous Materials
WHMIS 1988 Training Module 2017 Table of Contents 1. WHMIS a. Purpose b. 3 Main Components of WHMIS c. WHMIS Communication System d. Enforcement of WHMIS in Ontario 2. Responsibilities, Hazardous Materials
Bleaching. Mercerising Dyeing Printing. Finishing
 Subject: Dyeing and Printing Unit 2: Fabric preparatory process for dyeing and printing Quadrant 1 e-text Learning Objectives The learning objectives of this unit are: Outline the Preparatory Process involved
Subject: Dyeing and Printing Unit 2: Fabric preparatory process for dyeing and printing Quadrant 1 e-text Learning Objectives The learning objectives of this unit are: Outline the Preparatory Process involved
TABLE TOP CARE. Edition information pack
 TABLE TOP CARE Edition. 001 information pack INSTRUCTIONS Regular maintenance and care will help to lengthen the lifespan of your table tops and ensure they look as good as possible, for as long as possible.
TABLE TOP CARE Edition. 001 information pack INSTRUCTIONS Regular maintenance and care will help to lengthen the lifespan of your table tops and ensure they look as good as possible, for as long as possible.
MSDS Summary Sheet. Comet Creme Disinfectant Cleanser (Professional Line) TDG Requirements. First Aid Requirements
 MSDS Summary Sheet Revised: September 15, 2003 Product: Comet Creme Disinfectant Cleanser (Professional Line) Part Number(s): L301-CL-007 Department: Sub Assembly Welding, Maintenance, Medical WHMIS Classification
MSDS Summary Sheet Revised: September 15, 2003 Product: Comet Creme Disinfectant Cleanser (Professional Line) Part Number(s): L301-CL-007 Department: Sub Assembly Welding, Maintenance, Medical WHMIS Classification
Chemical Name: Cleanser with Chlorinol. Manufacturer: Comet. Container Size: 21 oz. Location: VLA. Disposal: Place empty container in trash.
 Chemical Name: Cleanser with Chlorinol Manufacturer: Comet Container Size: 21 oz. Location: VLA Disposal: Place empty container in trash. Page 1 of 4 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza
Chemical Name: Cleanser with Chlorinol Manufacturer: Comet Container Size: 21 oz. Location: VLA Disposal: Place empty container in trash. Page 1 of 4 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza
CLEANING, SANITIZING, AND DISINFECTING
 CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
Caring for Sheepskin it s simple
 Caring for Sheepskin it s simple Sheepskin is very easy to look after. The fibre s unique structure gives it a natural durability and robustness. It even repels dirt! Even so, a little care and attention
Caring for Sheepskin it s simple Sheepskin is very easy to look after. The fibre s unique structure gives it a natural durability and robustness. It even repels dirt! Even so, a little care and attention
Cleaning and care instructions
 Cleaning and care instructions designed & produced by Upholstery fabrics, like any other textiles, such as carpets and clothing, are constantly subjected to dust and surface soiling. Regular care and cleaning
Cleaning and care instructions designed & produced by Upholstery fabrics, like any other textiles, such as carpets and clothing, are constantly subjected to dust and surface soiling. Regular care and cleaning
Components CAS No. Range in %
 MATERIAL SAFETY DATA SHEET National Turbine Oil 46 LPP 1. Chemical & Company Identification Trade Name : Turbine Oil 46 LPP Supplier : Techno Lube LLC-Dubai. P.O. Box. 116636. Dubai United Arab Emirates.
MATERIAL SAFETY DATA SHEET National Turbine Oil 46 LPP 1. Chemical & Company Identification Trade Name : Turbine Oil 46 LPP Supplier : Techno Lube LLC-Dubai. P.O. Box. 116636. Dubai United Arab Emirates.
Maintenance Guidelines Compound
 6696 Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
6696 Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
UDATED JANUARY 2016 SPIRALS TEAK FURNITURE PRODUCT CARE WE STRONGLY RECOMMEND USE OF HIGH QUALITY FURNITURE COVERS
 SPIRALS TEAK UDATED JANUARY 2016 SPIRALS TEAK FURNITURE PRODUCT CARE WE STRONGLY RECOMMEND USE OF HIGH QUALITY FURNITURE COVERS The Seasonal Living Spirals Teak Collection is manufactured from Planation
SPIRALS TEAK UDATED JANUARY 2016 SPIRALS TEAK FURNITURE PRODUCT CARE WE STRONGLY RECOMMEND USE OF HIGH QUALITY FURNITURE COVERS The Seasonal Living Spirals Teak Collection is manufactured from Planation
How to care for FABRIC SOFA. Instruction Avoid
 How to care for FABRIC SOFA Instruction Avoid As with all materials of daily use, upholstery fabric requires care. To avoid premature wear and staining, fabric covered furniture should be regularly vacuumed
How to care for FABRIC SOFA Instruction Avoid As with all materials of daily use, upholstery fabric requires care. To avoid premature wear and staining, fabric covered furniture should be regularly vacuumed
WELCOME TO BRODY CHEMICAL
 LAUNDRY 1 WELCOME TO BRODY CHEMICAL Brody Chemical began operations in Salt Lake City, Utah in 1975. Since then, the company has grown from a small local company to one that provides superior products
LAUNDRY 1 WELCOME TO BRODY CHEMICAL Brody Chemical began operations in Salt Lake City, Utah in 1975. Since then, the company has grown from a small local company to one that provides superior products
Go2. Environmentally Sound Cleaning System. #3 #4 Easy as Put the cleaning power of mother nature to work for you! #1 #2
 Put the cleaning power of mother nature to work for you! Go2 Environmentally Sound Cleaning System #1 #2 #3 #4 Easy as 1234 E.logical brand symbolizes biotechnology chemistry providing safer, effective,
Put the cleaning power of mother nature to work for you! Go2 Environmentally Sound Cleaning System #1 #2 #3 #4 Easy as 1234 E.logical brand symbolizes biotechnology chemistry providing safer, effective,
Maintenance Guidelines Ellipsis
 Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
SAFETY DATA SHEET. Basis: Regulation (EC) No 1907/2006 of the European Parliament and of the Council of Europe, 18 th December 2006
 SAFETY DATA SHEET Basis: Regulation (EC) No 1907/2006 of the European Parliament and of the Council of Europe, 18 th December 2006 Date of issue 15.01.2013 Date of update 22.08.2014 1. IDENTIFICATION OF
SAFETY DATA SHEET Basis: Regulation (EC) No 1907/2006 of the European Parliament and of the Council of Europe, 18 th December 2006 Date of issue 15.01.2013 Date of update 22.08.2014 1. IDENTIFICATION OF
Developed by Western Massachusetts Coalition for Occupational Safety and Health Spring, 2001
 Developed by Western Massachusetts Coalition for Occupational Safety and Health Spring, 2001 Hazard Recognition Identify the hazardous chemicals in the salon products that you work with. Identify the hazard
Developed by Western Massachusetts Coalition for Occupational Safety and Health Spring, 2001 Hazard Recognition Identify the hazardous chemicals in the salon products that you work with. Identify the hazard
1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION
 MATERIAL SAFETY DATA SHEET Page 1 of 5 Product: Endocem ZR Product Code: DL1321 & DL1325 (5 pack) 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: Endocem ZR Recommended use: ENDOCEM ZR is
MATERIAL SAFETY DATA SHEET Page 1 of 5 Product: Endocem ZR Product Code: DL1321 & DL1325 (5 pack) 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: Endocem ZR Recommended use: ENDOCEM ZR is
Maintenance Guidelines Scuba
 Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
Maintenance Code W/B-Clean with water-based cleanser or diluted household bleach. Regular Maintenance Vacuum regularly using the proper attachment to avoid pilling. For non-woven textiles, wipe regularly
SAFETY DATA SHEET. Ammonium Oxalate Solution Manufacture of other chemical products, professional, sc ientific, and technical activities. None known.
 SAFETY DATA SHEET BIOPHARM INC. 1. Identification: Product Identifier: Ammonium Oxalate, various aqueous solutions Product code: BA8091 = 3.5% (w/v) BA8023 = 4% (w/v) Synonym(s): Recommended use: Recommended
SAFETY DATA SHEET BIOPHARM INC. 1. Identification: Product Identifier: Ammonium Oxalate, various aqueous solutions Product code: BA8091 = 3.5% (w/v) BA8023 = 4% (w/v) Synonym(s): Recommended use: Recommended
Cleaning Instructions
 Cleaning Instructions Although Tarimatec no maintenance, if that should be cleaned periodically installation to keep it looking good. It is necessary to clean the space between the slats of organic debris
Cleaning Instructions Although Tarimatec no maintenance, if that should be cleaned periodically installation to keep it looking good. It is necessary to clean the space between the slats of organic debris
Professional spotting guide from Prochem Europe
 STAIN REMOVAL GUIDE Professional spotting guide from Prochem Europe 1 STAIN REMOVAL GUIDE Spot & Stain Removal Tips & Precautions Always be aware that no guarantee can be given for complete stain removal
STAIN REMOVAL GUIDE Professional spotting guide from Prochem Europe 1 STAIN REMOVAL GUIDE Spot & Stain Removal Tips & Precautions Always be aware that no guarantee can be given for complete stain removal
sparkle. Natural clinging action saves on the amount of product used. Non-toxic and biodegradable.
 Laundry Detergent & Softener A naturally derived laundry detergent and pre-spotter made primarily from replenish able natural resources. Cleans, softens, brightens, whitens, deodorizes and removes stains.
Laundry Detergent & Softener A naturally derived laundry detergent and pre-spotter made primarily from replenish able natural resources. Cleans, softens, brightens, whitens, deodorizes and removes stains.
1. IDENTIFICATION OF MATERIAL AND SUPPLIER 2. HAZARDS IDENTIFICATION. Product Use. Coffee machine descaler Company Name Cafetto Address
 Page 1 of 5 Date of Issue: 6 March, 2017 1. IDENTIFICATION OF MATERIAL AND SUPPLIER Product Name Restore Product Use Coffee machine descaler Company Name Cafetto Address 12 Coglin Street, Brompton SA 5007,
Page 1 of 5 Date of Issue: 6 March, 2017 1. IDENTIFICATION OF MATERIAL AND SUPPLIER Product Name Restore Product Use Coffee machine descaler Company Name Cafetto Address 12 Coglin Street, Brompton SA 5007,
Safety Data Sheet Mighty Wipe 1. Identification
 Safety Data Sheet Mighty Wipe 1. Identification Trade name of product : Mighty Wipe Other Shipping Name : None allocated Supplier Address : North Queensland Cleaning & Paints 6-8 Industrial Ave Mackay
Safety Data Sheet Mighty Wipe 1. Identification Trade name of product : Mighty Wipe Other Shipping Name : None allocated Supplier Address : North Queensland Cleaning & Paints 6-8 Industrial Ave Mackay
MATERIAL SAFETY DATA SHEET
 MSDS : 1399-2 NAME OF PRODUCT : Acetic acid glacial PRODUCT IDENTIFICATION AND USE USE OF PRODUCT : Acetates manufacture TRANSPORTATION OF DANGEROUS GOODS SHIPPING NAME : Acetic acid glacial WHMIS: B3,
MSDS : 1399-2 NAME OF PRODUCT : Acetic acid glacial PRODUCT IDENTIFICATION AND USE USE OF PRODUCT : Acetates manufacture TRANSPORTATION OF DANGEROUS GOODS SHIPPING NAME : Acetic acid glacial WHMIS: B3,
METAL CARE GUIDE STAINLESS STEEL
 METAL CARE GUIDE TESKO ENTERPRISES WORKS WITH MANY METALS AND APPRECIATES THAT EACH IS UNIQUE IN ITS APPEARANCE AND PROPERTIES. OUR WORK IS ALWAYS GUARANTEED TO BE OF THE FINEST QUALITY REGARDLESS OF THE
METAL CARE GUIDE TESKO ENTERPRISES WORKS WITH MANY METALS AND APPRECIATES THAT EACH IS UNIQUE IN ITS APPEARANCE AND PROPERTIES. OUR WORK IS ALWAYS GUARANTEED TO BE OF THE FINEST QUALITY REGARDLESS OF THE
Potentially Dangerous Household Products
 Potentially Dangerous Household Products Please click on any of the links below to go directly to your specified topic within this document. If You Have a Poisoning Emergency Cleaning, Bleaching Agents
Potentially Dangerous Household Products Please click on any of the links below to go directly to your specified topic within this document. If You Have a Poisoning Emergency Cleaning, Bleaching Agents
Material Safety Data Sheets (MSDS)
 Material Safety Data Sheets (MSDS) TIME: 20 minutes PURPOSE OF THE ACTIVITY: The participant will be able to read and use a Material Safety Data Sheet. MATERIALS NEEDED: q q Copies of the MSDS Sheets for
Material Safety Data Sheets (MSDS) TIME: 20 minutes PURPOSE OF THE ACTIVITY: The participant will be able to read and use a Material Safety Data Sheet. MATERIALS NEEDED: q q Copies of the MSDS Sheets for
Material Safety Data Sheet
 Hazardous Substance, Non-Dangerous Goods 1. MATERIAL AND SUPPLY COMPANY IDENTIFICATION Product name: All in One Stain Remover Other Names: Citric Acid Monohydrate 2-HYDROXY-1,2,3-Propanetricarboxylic Acid,
Hazardous Substance, Non-Dangerous Goods 1. MATERIAL AND SUPPLY COMPANY IDENTIFICATION Product name: All in One Stain Remover Other Names: Citric Acid Monohydrate 2-HYDROXY-1,2,3-Propanetricarboxylic Acid,
Learning Guide. Common stains Identify and remove common stains Level 3 6 credits. Name: Workplace: Issue 1.0
 Learning Guide Common stains 29383 Identify and remove common stains Level 3 6 credits Name: Workplace: Issue 1.0 Copyright 2017 Careerforce All rights reserved. Careerforce (Community Support Services
Learning Guide Common stains 29383 Identify and remove common stains Level 3 6 credits Name: Workplace: Issue 1.0 Copyright 2017 Careerforce All rights reserved. Careerforce (Community Support Services
: Promat Glue K84 : Viscous liquid. : Glue, resistant to high temperature
 SECTION 1 Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Trade name Identification of the product Type of product : : Viscous liquid. : Glue, resistant to
SECTION 1 Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Trade name Identification of the product Type of product : : Viscous liquid. : Glue, resistant to
PROFESSIONAL SPOTTING KIT
 CLEANING SOLUTIONS Table of Contents Fire Restoration Products... 2 Emulsifier/Prespray Products... 3 Interim Cleaning Products... 2-3 Spotting Products... 4 Upholstery Care... 5 Deodorizers... 5 Green
CLEANING SOLUTIONS Table of Contents Fire Restoration Products... 2 Emulsifier/Prespray Products... 3 Interim Cleaning Products... 2-3 Spotting Products... 4 Upholstery Care... 5 Deodorizers... 5 Green
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 Procter & Gamble Household Care Division Ivorydale Technical Center 5299 Spring Grove Avenue Cincinnati, OH 45217-1087 MATERIAL SAFETY DATA SHEET MSDS #: CT: FH/O/2005/ASAM-6CCNRJ Issue Date:
Page 1 of 5 Procter & Gamble Household Care Division Ivorydale Technical Center 5299 Spring Grove Avenue Cincinnati, OH 45217-1087 MATERIAL SAFETY DATA SHEET MSDS #: CT: FH/O/2005/ASAM-6CCNRJ Issue Date:
Material Safety Data Sheet CALCIPAST + I Temporary root canal filling material with Iodoform
 Page 1 of 6 MSDS is prepared in accordance with THE REGULATION (EC) No 1907/2006 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 18 December 2006 concerning the Registration, Evaluation, Authorization
Page 1 of 6 MSDS is prepared in accordance with THE REGULATION (EC) No 1907/2006 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 18 December 2006 concerning the Registration, Evaluation, Authorization
Page 1 of 5. Citric CIP Liquid
 Page 1 of 5 1. IDENTIFICATION OF MATERIAL AND SUPPLIER Citric CIP Liquid Product Name Citric CIP Liquid Code 50341 Product Use For neutralising tanks and lines in the food and drink industry after using
Page 1 of 5 1. IDENTIFICATION OF MATERIAL AND SUPPLIER Citric CIP Liquid Product Name Citric CIP Liquid Code 50341 Product Use For neutralising tanks and lines in the food and drink industry after using
GHS07. H315 - Causes skin irritation. H320 - Causes eye irritation. H335 - May cause respiratory irritation.
 Revision Date: June 2015 Version: 1.0 SECTION 1: IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY Product Identifier Product Name: Blended Fertilizer; Urea-Based Intended Use of the Product :
Revision Date: June 2015 Version: 1.0 SECTION 1: IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY Product Identifier Product Name: Blended Fertilizer; Urea-Based Intended Use of the Product :
GOJO Green Certified Foam Hand Cleaner
 SECTION 1. PRODUCT AND COMPANY IDENTIFICATION Product name : Manufacturer or supplier's details Company name of supplier : GOJO Industries, Inc. Address : One GOJO Plaza, Suite 500 Akron, Ohio 44311 Telephone
SECTION 1. PRODUCT AND COMPANY IDENTIFICATION Product name : Manufacturer or supplier's details Company name of supplier : GOJO Industries, Inc. Address : One GOJO Plaza, Suite 500 Akron, Ohio 44311 Telephone
Safety Data Sheet (OSHA 29 CFR ) Section 1 - Product and Company Identification
 Safety Data Sheet (OSHA 29 CFR 1910.1200) Section 1 - Product and Company Identification Product Company Organocide Plant Doctor Organic Laboratories Phone (772) 286-5581 Product Use Plant nutrition Emergency
Safety Data Sheet (OSHA 29 CFR 1910.1200) Section 1 - Product and Company Identification Product Company Organocide Plant Doctor Organic Laboratories Phone (772) 286-5581 Product Use Plant nutrition Emergency
A;:>IVI Material Safety Data Sheet
 A;:>IVI MAN!, ::i MU;.', COMPANY DETAILS COMPANY NAME: ADDRESS; ADM Australian Dental Manufacturing ACN; 010 967 432 25 Billabong Street Kcnmore Hills Brisbane, Queensland. 4069 TELEPHONE: FAX: EMAIL:
A;:>IVI MAN!, ::i MU;.', COMPANY DETAILS COMPANY NAME: ADDRESS; ADM Australian Dental Manufacturing ACN; 010 967 432 25 Billabong Street Kcnmore Hills Brisbane, Queensland. 4069 TELEPHONE: FAX: EMAIL:
Stone Care Guidelines
 Each stone, created by the earth itself, upholds its own unique properties and geological composition. Because of this, some stones require greater maintenance than others. By following these simple guidelines,
Each stone, created by the earth itself, upholds its own unique properties and geological composition. Because of this, some stones require greater maintenance than others. By following these simple guidelines,
SAFETY DATA SHEET according to Regulation (EC) No. 453/2010 Dental Use
 Page 1 of 8 v SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code : OH/71059 Product name : Mint HV Product description : Dental
Page 1 of 8 v SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code : OH/71059 Product name : Mint HV Product description : Dental
Please Read Before Applying General Directions: Note: You may want to print these for future use!
 Please Read Before Applying General Directions: Note: You may want to print these for future use! Page One: GENERAL DIRECTIONS Page Two: SPECIAL DIRECTIONS FOR CARPET AND UPHOLSTERY Page Three: SPECIAL
Please Read Before Applying General Directions: Note: You may want to print these for future use! Page One: GENERAL DIRECTIONS Page Two: SPECIAL DIRECTIONS FOR CARPET AND UPHOLSTERY Page Three: SPECIAL
Part Numbers: 80227, 80255, 80265, 80256, 80237, 80257, 80253, 80278, 80289, Hazard Rating: Health: 1 Fire: 1 Reactivity: 0
 SAF-T-LOK International Corporation Material Safety Data Sheet SAF-T-EZE NICKEL ANTI-SEIZE Issue Date: 10/19/17 SECTION I - PRODUCT AND COMPANY INFORMATION Product Name: NICKEL ANTI-SEIZE Product Type:
SAF-T-LOK International Corporation Material Safety Data Sheet SAF-T-EZE NICKEL ANTI-SEIZE Issue Date: 10/19/17 SECTION I - PRODUCT AND COMPANY INFORMATION Product Name: NICKEL ANTI-SEIZE Product Type:
71 Stradbroke Street Heathwood Queensland 4110 Australia
 Safety Data Sheet LASER 1. Identification Trade name of product Other Shipping Name Supplier Address : LASER : Corrosive Liquid N.O.S. (contains Phosphoric Acid) : Total Hygiene Solutions 71 Stradbroke
Safety Data Sheet LASER 1. Identification Trade name of product Other Shipping Name Supplier Address : LASER : Corrosive Liquid N.O.S. (contains Phosphoric Acid) : Total Hygiene Solutions 71 Stradbroke
MATERIAL SAFETY DATA SHEET According to ANSI Z /ISO :1994 Para-Pak SAF and FeKal SAF
 MATERIAL SAFETY DATA SHEET According to ANSI Z400.1-1998/ISO 11014-1:1994-9002 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Product Type Fixative - 9002 Product Name Catalogue
MATERIAL SAFETY DATA SHEET According to ANSI Z400.1-1998/ISO 11014-1:1994-9002 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Product Type Fixative - 9002 Product Name Catalogue
O Oxidizing R: Phrases R34 Causes Burns R36/37/38 Irritating to eyes, respiratory system and skin. 41 Risk of serious damage to eyes
 Synca Marketing Inc. MATERIAL SAFETY DATA SHEET CP PEROXIDE SECTION 1 CHEMICAL PRODUCT AND COMPANY INDENTIFICATION Product Identifier WHIMIS Classification BRILLIANCE Product Use Dental Whitening Gel Manufacturer
Synca Marketing Inc. MATERIAL SAFETY DATA SHEET CP PEROXIDE SECTION 1 CHEMICAL PRODUCT AND COMPANY INDENTIFICATION Product Identifier WHIMIS Classification BRILLIANCE Product Use Dental Whitening Gel Manufacturer
NOT CLASSIFIED AS HAZARDOUS ACCORDING TO CRITERIA OF NOHSC. International Beauty Supplies. 6e Narabang Way, Belrose NSW, Australia 2085
 1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address SOIN DU CORPS CYOSLIM LOTION CRYOTONIQUE SB81324 BODY SKIN CARE International Beauty Supplies
1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address SOIN DU CORPS CYOSLIM LOTION CRYOTONIQUE SB81324 BODY SKIN CARE International Beauty Supplies
CARE AND MAINTENANCE
 CARE AND MAINTENANCE CARE AND MAINTENANCE 02 CARE AND MAINTENANCE Quartz Surfaces are high-quality, solid, non-porous products that resist scratches, heat, and stains. However, VICOSTONE Quartz Surfaces
CARE AND MAINTENANCE CARE AND MAINTENANCE 02 CARE AND MAINTENANCE Quartz Surfaces are high-quality, solid, non-porous products that resist scratches, heat, and stains. However, VICOSTONE Quartz Surfaces
Care and Maintenance Guide
 1 Care and Maintenance Guide Cal Flame Barbecue Islands, Fireplaces, and Fire Pits This brief guide describes how to clean, protect, and repair your Cal Flame product. It discusses the following topics:
1 Care and Maintenance Guide Cal Flame Barbecue Islands, Fireplaces, and Fire Pits This brief guide describes how to clean, protect, and repair your Cal Flame product. It discusses the following topics:
MATERIAL SAFETY DATA SHEET Description: SAF LIQUID LAUNDRY DETERGENT Revision Number: 01
 SUPER BRIGHT INDUSTRIAL STRENGTH WASHING LIQUID GENERAL Using an exclusive blend of sodium chelating agents, biodegradable non-ionic surfactants & optical brighteners, this highly concentrated product
SUPER BRIGHT INDUSTRIAL STRENGTH WASHING LIQUID GENERAL Using an exclusive blend of sodium chelating agents, biodegradable non-ionic surfactants & optical brighteners, this highly concentrated product
Material Safety Data Sheet
 1. PRODUCT AND COMPANY IDENTIFICATION Material Safety Data Sheet turtle wax, inc 625 Willowbrook Ctr Pkwy Willowbrook, Illinois 60527 PRODUCT NAME PRODUCT CODE CHEMICAL FAMILY CHEMICAL NAME FORMULA MANUFACTURER
1. PRODUCT AND COMPANY IDENTIFICATION Material Safety Data Sheet turtle wax, inc 625 Willowbrook Ctr Pkwy Willowbrook, Illinois 60527 PRODUCT NAME PRODUCT CODE CHEMICAL FAMILY CHEMICAL NAME FORMULA MANUFACTURER
MATERIAL SAFETY DATA SHEET Description: SAF BLEACH Revision Number: 01 Document Number: MSDS B015
 Description: SAF BLEACH Revision Number: 01 FORMULATION SAF-BLEACH is a concentrated, liquid chlorine stain remover, deodoriser and sanitizer formulated for rapid chlorine release. PRODUCT DESCRIPTION
Description: SAF BLEACH Revision Number: 01 FORMULATION SAF-BLEACH is a concentrated, liquid chlorine stain remover, deodoriser and sanitizer formulated for rapid chlorine release. PRODUCT DESCRIPTION
BIODEGRADABLE SCREEN PREPARATION CHEMICALS
 BIODEGRADABLE SCREEN PREPARATION CHEMICALS 160 Fenmar Drive, Weston, Ontario, Canada, M9L 1M6, Tel: 877-465-6362, Fax: 416-743-1511 12180 Boul.Industriel, Montreal, Quebec, H1B 5R7, Tel: 514-644-4404,
BIODEGRADABLE SCREEN PREPARATION CHEMICALS 160 Fenmar Drive, Weston, Ontario, Canada, M9L 1M6, Tel: 877-465-6362, Fax: 416-743-1511 12180 Boul.Industriel, Montreal, Quebec, H1B 5R7, Tel: 514-644-4404,
SAFETY DATA SHEET. LaserWhite20 Bleaching Gel 46% HP 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING
 LaserWhite20 Bleaching Gel 46% HP 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME: LaserWhite20 Bleaching Gel 46% Hydrogen Peroxide PRODUCT DESCRIPTION: In-office
LaserWhite20 Bleaching Gel 46% HP 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME: LaserWhite20 Bleaching Gel 46% Hydrogen Peroxide PRODUCT DESCRIPTION: In-office
!!! CCS LAVENDER SPIKE OIL ESSENCE!!!!!! ! SAFETY DATA SHEET (SDS)! Page 1 of 5. MSDS for # CLSCL STDIO OILS MED
 Page 1 of 5 SAFETY DATA SHEET (SDS) CCS LAVENDER SPIKE OIL ESSENCE Identification Name: CCS LAVENDER SPIKE OIL ESSENCE Relevant identified uses of the substance Use of the substance/mixture: For oil painting
Page 1 of 5 SAFETY DATA SHEET (SDS) CCS LAVENDER SPIKE OIL ESSENCE Identification Name: CCS LAVENDER SPIKE OIL ESSENCE Relevant identified uses of the substance Use of the substance/mixture: For oil painting
Developed by Western Massachusetts Coalition for Occupational Safety and Health. Funded by The Toxics Use Reduction Institute
 Developed by Western Massachusetts Coalition for Occupational Safety and Health Funded by The Toxics Use Reduction Institute Hair Relaxing Process Health and Safety Concerns Health and Safety Precautions
Developed by Western Massachusetts Coalition for Occupational Safety and Health Funded by The Toxics Use Reduction Institute Hair Relaxing Process Health and Safety Concerns Health and Safety Precautions
SAFETY DATA SHEET according to Regulation (EC) No. 453/2010 Dental Use
 Page 1 of 8 SDS No. V008 SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code : 40005460, 40005462, 40005465 Product name : Product
Page 1 of 8 SDS No. V008 SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code : 40005460, 40005462, 40005465 Product name : Product
carpet care products Providing Quality Maintenance and Chemical Specialty Solutions with Unparalleled Customer Service
 carpet care products Maintaining carpet at a high appearance level extends the life of carpet and protects your investment. Spartan provides the highest quality carpet care products with easy-to-follow
carpet care products Maintaining carpet at a high appearance level extends the life of carpet and protects your investment. Spartan provides the highest quality carpet care products with easy-to-follow
Instructor Guide. Georgia Department of Technical and Adult Education. Decontamination and Infection Control
 Instructor Guide Georgia Department of Technical and Adult Education Decontamination and Infection Control Copyright October 2002 by. All rights reserved. No part of this manual may be reproduced or transmitted
Instructor Guide Georgia Department of Technical and Adult Education Decontamination and Infection Control Copyright October 2002 by. All rights reserved. No part of this manual may be reproduced or transmitted
Material Safety Data Sheet Volara Type A All Colors, All Densities
 MSDS No: 0100 Validation Date: 04/30/09 Revision No: 4 Material Safety Data Sheet Volara Type A All Colors, All Densities Volara Type A Derivatives: AB, AO, AL, AA, AC, AF, AFL, AFR, AFX, AG, AGF, AGL,
MSDS No: 0100 Validation Date: 04/30/09 Revision No: 4 Material Safety Data Sheet Volara Type A All Colors, All Densities Volara Type A Derivatives: AB, AO, AL, AA, AC, AF, AFL, AFR, AFX, AG, AGF, AGL,
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET Product Name: SILK OIL OF MOROCCO ARGAN VOLUMIZING SHAMPOO SECTION 1: IDENTIFICATION OF THE MATERIAL & SUPPLIER 1.1 Supplier Name: Totally Hair Direct Pty Ltd 1.2 Address: PO
MATERIAL SAFETY DATA SHEET Product Name: SILK OIL OF MOROCCO ARGAN VOLUMIZING SHAMPOO SECTION 1: IDENTIFICATION OF THE MATERIAL & SUPPLIER 1.1 Supplier Name: Totally Hair Direct Pty Ltd 1.2 Address: PO
CARE AND MAINTENANCE
 CARE AND MAINTENANCE CARE AND MAINTENANCE Vicostone Quartz surfaces are a high quality, solid, non-porous product that is resistant to scratches, heat, and stains. However, Vicostone Quartz surfaces are
CARE AND MAINTENANCE CARE AND MAINTENANCE Vicostone Quartz surfaces are a high quality, solid, non-porous product that is resistant to scratches, heat, and stains. However, Vicostone Quartz surfaces are
Vasari LLC products as listed above are composed of the following materials:
 Material Safety Data Sheet Manufacturer : Vasari Plaster & Stucco llc 624 E. Haley St. Santa Barbara, CA, 93103 Telephone: (805) 845-2497 Prepared 12-03-09 by Vasari Plaster & Stucco LLC 24 hour emergency
Material Safety Data Sheet Manufacturer : Vasari Plaster & Stucco llc 624 E. Haley St. Santa Barbara, CA, 93103 Telephone: (805) 845-2497 Prepared 12-03-09 by Vasari Plaster & Stucco LLC 24 hour emergency
SAFETY DATA SHEET Regulation (EC) No 1907/2006 ATP EASY2, DISPERSE INK MAGENTA
 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product information Product name : Product code : 60 INKU 0012 Use : Digital printing ink Company : Telephone : Telefax : E-mail
1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product information Product name : Product code : 60 INKU 0012 Use : Digital printing ink Company : Telephone : Telefax : E-mail
Material Safety Data Sheet
 SECTION 1: PRODUCT AND COMPANY IDENTIFICATION Product Name Sku# BWK 704-8 Company LAGASSE, Inc. One Parkway North Deerfield, IL USA 60015 Telephone: 800-989-7847 Prepared By Product Development KIK Classic
SECTION 1: PRODUCT AND COMPANY IDENTIFICATION Product Name Sku# BWK 704-8 Company LAGASSE, Inc. One Parkway North Deerfield, IL USA 60015 Telephone: 800-989-7847 Prepared By Product Development KIK Classic
SECTION 1 : IDENTIFICATION. Emergency Phone Number (702) Distributor, Importer, or Other Responsible Party Name Address.
 SAFETY DATA SHEET : Dynamite Page 1 of 7 SECTION 1 : IDENTIFICATION Product Identifier Used on the Label Other Means of Identification (Part Number, Common Name, Synonyms) Recommended Use of the Chemical
SAFETY DATA SHEET : Dynamite Page 1 of 7 SECTION 1 : IDENTIFICATION Product Identifier Used on the Label Other Means of Identification (Part Number, Common Name, Synonyms) Recommended Use of the Chemical
SAFETY DATA SHEET. Anasept Antimicrobial Skin and wound Gel. Revision date: 06/16/2017 Revision number: 02
 1. PRODUCT NAME AND COMPANY IDENTIFICATION: GHS product identifier: Product name: Other mean of identification Product code(s): UN-Number: Synonyms: Anasept Antimicrobial Skin and Wound Gel 5015G, 5003G
1. PRODUCT NAME AND COMPANY IDENTIFICATION: GHS product identifier: Product name: Other mean of identification Product code(s): UN-Number: Synonyms: Anasept Antimicrobial Skin and Wound Gel 5015G, 5003G
Household Chemicals. J. W. Hill and D. K. Kolb, Chemistry for Changing Times, Chapter 17.
 Household Chemicals J. W. Hill and D. K. Kolb, Chemistry for Changing Times, Chapter 17. Soap and Detergent http://www.sdahq.org/ Soap and Detergent Old way of making soap: Animal fats or vegetable oil
Household Chemicals J. W. Hill and D. K. Kolb, Chemistry for Changing Times, Chapter 17. Soap and Detergent http://www.sdahq.org/ Soap and Detergent Old way of making soap: Animal fats or vegetable oil
For BALKAMP Inc. by The NOCO Company, Glenwillow, OH % by WT CAS No. Ingredient PEL PPM TLV PPM TLV MG/M3 UEL
 Section 1 Identification. Product name: NOCO NCP-2 Battery Corrosion Preventative Brush-On Compound (4 oz.) Recommended Use: Brush on battery post to fight battery corrosion and prolong battery life. Product
Section 1 Identification. Product name: NOCO NCP-2 Battery Corrosion Preventative Brush-On Compound (4 oz.) Recommended Use: Brush on battery post to fight battery corrosion and prolong battery life. Product
Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None. Emergency telephone number: CHEMTREC
 1. Product and Company Identification Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None Recommended Use: Uses Advised Against: No information available Supplier information: Garcoa
1. Product and Company Identification Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None Recommended Use: Uses Advised Against: No information available Supplier information: Garcoa
SAFETY DATA SHEET SILK OIL OF MOROCCO ARGAN ANTI-FRIZZ CREAM
 SAFETY DATA SHEET Product Name: SILK OIL OF MOROCCO ARGAN ANTI-FRIZZ CREAM SECTION 1: IDENTIFICATION OF THE MATERIAL & SUPPLIER 1.1 Supplier Name: Totally Hair Direct Pty Ltd 1.2 Address: PO Box 2146 Redcliffe
SAFETY DATA SHEET Product Name: SILK OIL OF MOROCCO ARGAN ANTI-FRIZZ CREAM SECTION 1: IDENTIFICATION OF THE MATERIAL & SUPPLIER 1.1 Supplier Name: Totally Hair Direct Pty Ltd 1.2 Address: PO Box 2146 Redcliffe
