BD Disposal Solutions Product Catalog
|
|
|
- Emil Sharp
- 5 years ago
- Views:
Transcription
1 Product Catalog Recognizing that human health and a healthy environment are inseparable Sharps Pharmaceutical Chemotherapy RCRA Accessories
2 Driven by Sustainability We re working smarter to evolve BD Sharps Disposal Solutions So we can bring you high-quality products with a sharp focus on a greener, sustainable future BD is always searching for ways to be a better environmental steward while helping our customers. That s why our BD Recykleen products are made from post-industrial and/or post-consumer recycled materials. We continue to overcome obstacles and strive to maximize the recycled content in each of our products. We are committed to moving the entire line in this direction, one step at a time. Did You Know... Pounds Percentage of total units sold that contain recycled material % 70% 8 25% 6 15% 9 % 10 % 20% % 1 % 2 % % 2
3 Evolution in the BD is committed to a greener, sustainable future. BD Recykleen Brand Direction Products made to suit your clinical needs and the environment Our goals high quality, less environmental impact We strive to use as much recycled content as possible, while maintaining the products you know and trust, in order to deliver high-quality, clinically proven disposal solutions with a sharp focus on sustainability. In line with our long term goals, standard sharps collectors may also contain recycled material. offers: Improved products that are best suited to your clinical needs 100% Products that incorporate more recycled content than before Counterbalanced door for hands-free disposal Large opening for easy, safe disposal 70% VAT locks for secure assembly (Visual, Audible, Tactile) Handles designed for safe transport Practical considerations for greener BD Recykleen products Supply Availability of recycled resin sufficient to meet the market demand Performance Same stringent performance requirements for recycled resin as those for virgin plastic 3
4 Table of Contents Waste Stream Segregation...5 How to Choose the Right Collector...6 Patient Room Sharps Collectors...7 Extra-Large Sharps Collectors...8 Nestable Sharps Collectors...9 Sharps Collectors...10 Phlebotomy Sharps Collectors Pharmaceutical Sharps Collectors...11 Chemotherapy Sharps Collectors RCRA Hazardous Waste Collectors Non-DOT...13 Cabinets and Accessories Trolleys...15 Look for these symbols throughout the catalog to easily identify key features and benefits: SECURITY HANDS FREE SLIDE TOP DUAL OPEN ABSORBENT LOW TEMP HIGH TEMP 4
5 Waste Stream Segregation offers collectors to meet the disposal needs of the four categories of waste generated in medical settings. There are two main categories of waste: regulated medical waste and hazardous waste. Regulated medical waste includes Sharps / Infectious Waste, Pharmaceutical (non-hazardous) and Chemotherapy waste. The hazardous waste category is for the collection of RCRA (U.S. EPA Resource Conservation and Recovery Act) waste. This table indicates general guidelines for proper waste stream segregation. Regulated Medical Waste Hazardous Waste Sharps Non-Hazardous Pharmaceutical Trace Chemotherapy Hazardous RCRA Needles Syringes Pipettes Glass slides Sharp instruments Other single-use sharps ( Do not include any pharmaceuticals in the sharps container) Partial creams, ointments Partial vials, ampules Partially filled syringes Discontinued IVs with non-hazardous pharmaceuticals Trace chemotherapy waste Gowns Masks Gloves IV tubing Trace chemotherapy sharps Vials (empty / trace) Syringes / needles (empty / trace) IVs (empty / trace) Hazardous compatible RCRA pharm waste * P-, U-, D-listed RCRA pharmaceutical waste (e.g., drugs that are reactive, ignitable, corrosive or toxic) Non-RCRA bulk chemo Hazardous aerosol pharm waste * Pressurized inhalers * * Message or label will indicate Autoclave Incineration Grinder / Shredder Landfill LOW TEMP Landfill HIGH TEMP Lined hazardous waste landfill 5
6 How to Choose the Right Collector Waste Stream Type Capacity Available in BD Recykleen Access Opening Size (inches) Sample Products that Fit in Collector Sharps Patient Room Phlebotomy Nestable 5.4 qt with Counterbalanced Door 2.0 x gal with Counterbalanced Door 2.0 x gal with Counterbalanced Door 2.0 x 8.0 Blood collection tubes and collection assembly, syringes up to 60 ml with needle attached 1 qt 2.5 x 1.2 Open (305635) 1 ml - 10 ml syringes, blood collection 1.5 qt 1.5 x 1.5 Open (305487) tubes and assembly, slides, pipettes 8 qt 14 qt 6 gal 2.5 x 1.75 Dia Open ( only); 1.4 x 1.0 Dia Petals (305344, ) 2.5 Dia Open (305646); 4.22 Dia Petals (305456); 1.8 x 1.1 Dia Petals (305464); 1.0 Dia Petals (305480) 4.22 Dia Open (305457); 1.0 Dia Petals (305465) 1 ml - 20 ml syringes with needle, blood collection tubes, slides, pipettes 1 ml - 60 ml syringes with needle, blood collection assembly, blood collection tubes, slides, pipettes slides, urine collection assembly, blood culture bottles, blood collection assembly 1.4 qt 1.8 Dia Petals 3.3 qt 1.8 Dia Petals 6.9 qt 1.8 Dia Petals Small syringes, needles, pipettes, slides 8.2 qt 1.8 Dia Petals 5 gal 4.2 Dia Open slides, urine collection assembly, blood culture bottles, blood collection assembly 9 gal Hinge Top 9.0 x 10.0 All items mentioned above plus small laparoscopic instruments, phlebotomy 9 gal Slide Top 5.0 x 7.5 procedure bottles, dialysis catheters 17 gal Hinge Top 10.5 x 12.5 Medical devices, spinal tap devices, 19 gal Slide Top 6.0 x 8.5 laparoscopy instrumentation Non-Hazardous Pharmaceutical Patient Room 5.4 qt with Counterbalanced Door 2.0 x gal with Counterbalanced Door 2.0 x 8.0 small IV bags 50 ml ml, medication vials, ampules small IV bags 50 ml ml 3 gal 4.2 Dia Open (305633) All syringe sizes and bags up to 250 ml 9 gal Hinge Top 9.0 x gal Slide Top 5.0 x gal Hinge Top 10.5 x gal Slide Top 6.0 x 8.5 All syringe sizes, all IV bag sizes with tubing attached, all medication vials, ampules, TPN bags, IV glass bottles Trace Chemotherapy Hazardous RCRA 3 gal 4.22 Dia Open (305076) 5 gal 4.2 Dia Open (305493) 5 gal Screw Cap 4.5 Dia Open (305578) 9 gal Hinge Top 9.0 x gal Slide Top 5.0 x gal Hinge Top 10.5 x gal Slide Top 6.0 x Dia Open (305066) 3 gal 1.0 Dia Petal (305075) 5 gal 4.2 Dia Open (305067) 9 gal Hinge Top 9.0 x gal Slide Top 5.0 x gal Hinge Top 10.5 x gal Slide Top 6.0 x 8.5 slides, urine collection assembly, blood culture bottles, blood collection assembly All syringe sizes, all IV bag sizes with tubing attached, all medication vials, ampules, TPN bags, IV glass bottles Smaller size of solid RCRA hazardous materials plus liquids if contained in a leak resistant container Solid RCRA hazardous materials plus liquids if contained in a leak resistant container 6
7 Patient Room Sharps Collectors BD Patient Room Collectors are available in BD Recykleen brand and BD standard brand and come in a variety of sizes and colors. All BD Patient Room Collectors have visible fill-level indicators and handles for safe transportation. Key product features include: Compatible with locking cabinets to provide security and prevent unwanted access to collector contents Counterbalanced door facilitates hands-free disposal and is designed to reduce the potential of sharps injuries Designed to improve tamper resistance by preventing hand access to the contents Available in BD Recykleen brand % 15% recycled material HANDS FREE Patient / Exam Room Reference # Product Size BD Recykleen Sharps Collectors Product Dimensions ( H x W x D inches ) Qty Case Compatible with gal BD Recykleen Counterbalanced Door Pearl 16.6 x 10.7 x , , , gal BD Recykleen Counterbalanced Door Red 16.6 x 10.7 x , qt Counterbalanced Door Red 12 x 12 x qt Counterbalanced Door Clear 12 x 12 x qt Counterbalanced Door Red 12 x 12 x qt Counterbalanced Door Clear 12 x 12 x gal Counterbalanced Door Pearl x x gal Counterbalanced Door Red x x gal Counterbalanced Door Red 16.6 x 10.7 x qt Counterbalanced Side Entry Red 11.7 x 11.6 x qt Counterbalanced Side Entry Pearl 11.7 x 11.6 x qt Counterbalanced Side Entry Pearl 11.7 x 11.6 x , , , , , , , ,
BD Disposal Solutions Product Catalog
 Product Catalog Recognizing that human health and a healthy environment are inseparable Sharps Pharmaceutical Chemotherapy RCRA Accessories The Story Table of Contents Waste Stream Segregation...4 Being
Product Catalog Recognizing that human health and a healthy environment are inseparable Sharps Pharmaceutical Chemotherapy RCRA Accessories The Story Table of Contents Waste Stream Segregation...4 Being
PREVENT. Sharps Containers. If you are not completely satisfied with any McKesson Brands product, you may return it for a full refund or credit.
 Sharps Containers McKesson offers a full line of sharps containers and accessories that feature OSHA-compliant safety features and excellent performance. The line was created to help protect our customers
Sharps Containers McKesson offers a full line of sharps containers and accessories that feature OSHA-compliant safety features and excellent performance. The line was created to help protect our customers
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
BSL-2 Emergency Plan
 BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
Biohazardous Waste. 1. Solid Biohazardous Waste (non-sharps) Storage
 Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
For Pricing and Ordering Please Call
 For Pricing and Ordering Please Call 800.616.5319 Needles and Syringes Prefilled I.V. Flush Syringes, Blood Collection and Sharps Disposal Products 1-800-962-9888 1-800-962-9888 3 Table of Contents Needles
For Pricing and Ordering Please Call 800.616.5319 Needles and Syringes Prefilled I.V. Flush Syringes, Blood Collection and Sharps Disposal Products 1-800-962-9888 1-800-962-9888 3 Table of Contents Needles
Safe Sharps Disposal. Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program
 Safe Sharps Disposal Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program How to Safely Dispose of Household Sharps Millions of people use
Safe Sharps Disposal Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program How to Safely Dispose of Household Sharps Millions of people use
Surgical Gown. Tongue Depressor. A disposable gown worn by medical staff during surgery. A thin, flat, wooden stick rounded at both ends
 Tongue Depressor A thin, flat, wooden stick rounded at both ends Accidentally dropped on the floor by the doctor 16 Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon
Tongue Depressor A thin, flat, wooden stick rounded at both ends Accidentally dropped on the floor by the doctor 16 Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon
Safe Handling and Disposal of Sharps. Reference Guide
 Safe Handling and Disposal of Sharps Reference Guide Safe Handling and Disposal of Syringes and other Sharps All staff involved in the administration of a drug or other substance should be trained in the
Safe Handling and Disposal of Sharps Reference Guide Safe Handling and Disposal of Syringes and other Sharps All staff involved in the administration of a drug or other substance should be trained in the
SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling Kuala Lumpur, Malaysia. Tel : Fax :
 DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling 57000 Kuala Lumpur, Malaysia. Tel : 03-9057 1316 Fax : 03-90578925 sales@dzcmarketing.com WhatsApp +6018-666 8794 MEDICAL DISPOSABLE LIST
DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling 57000 Kuala Lumpur, Malaysia. Tel : 03-9057 1316 Fax : 03-90578925 sales@dzcmarketing.com WhatsApp +6018-666 8794 MEDICAL DISPOSABLE LIST
Safety Rules for Laboratory
 Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Infection Control 101
 Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Disposal of Biohazard Wastes
 4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
UPEI Waste Disposal Protocol
 UPEI Waste Disposal Protocol Purpose: The purpose of this document is to ensure that waste is disposed of properly and safely in order to ensure the safety of all who handle waste. Waste Pretreatment:
UPEI Waste Disposal Protocol Purpose: The purpose of this document is to ensure that waste is disposed of properly and safely in order to ensure the safety of all who handle waste. Waste Pretreatment:
Infection Prevention Guidelines. Safe Use, Handling & Disposal of Sharps
 Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
TARLETON STATE UNIVERSITY Biohazardous Waste Program
 TARLETON STATE UNIVERSITY Biohazardous Waste Program Office of Risk Management and Safety June 2012 1. GENERAL The following information is provided to assist in developing requirements, guidelines and
TARLETON STATE UNIVERSITY Biohazardous Waste Program Office of Risk Management and Safety June 2012 1. GENERAL The following information is provided to assist in developing requirements, guidelines and
Medical Waste Management Plan
 Medical Waste Management Plan Safety Services - Biosafety University of California, Davis Version 1.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website:
Medical Waste Management Plan Safety Services - Biosafety University of California, Davis Version 1.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website:
Handling and Disposing of Needles
 Guidance Document UBC-RMS-OHS-GDL 14-008 Effective date: June 4, 2014 Review date: June 4, 2014 Supersedes: N/A 1. SCOPE Handling and Disposing of Needles This guidance document on Handling and Disposing
Guidance Document UBC-RMS-OHS-GDL 14-008 Effective date: June 4, 2014 Review date: June 4, 2014 Supersedes: N/A 1. SCOPE Handling and Disposing of Needles This guidance document on Handling and Disposing
Biological Safety Training
 Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Roosevelt Biosafety Training. Created 10/2015
 Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN
 FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
ECU Radiation, Biosafety and Hazardous Substances Committee
 Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION
 State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
Biosafety Self-Audit Checklist
 Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Regulated Medical Waste. Be sure to sign in!
 Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
MEDICAL WASTE MANAGEMENT PLAN
 MEDICAL WASTE MANAGEMENT PLAN University of California, Davis Version 2.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website: safetyservices.ucdavis.edu
MEDICAL WASTE MANAGEMENT PLAN University of California, Davis Version 2.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website: safetyservices.ucdavis.edu
TARLETON STATE UNIVERSITY Biohazardous Waste Program
 TARLETON STATE UNIVERSITY Biohazardous Waste Program Program Name: Biohazardous Waste Department Name: TSU Risk Management & Compliance Doc. No.: BIOS-04-L2-S0-CH0-001 Rev. No.: 2 Concurrence and Approval
TARLETON STATE UNIVERSITY Biohazardous Waste Program Program Name: Biohazardous Waste Department Name: TSU Risk Management & Compliance Doc. No.: BIOS-04-L2-S0-CH0-001 Rev. No.: 2 Concurrence and Approval
West Tennessee Healthcare Part 3
 West Tennessee Healthcare Part 3 Hazardous Pharmaceutical Waste Program: RCRA Waste Why are we concerned about hazardous pharmaceutical waste? EPA has determined that certain pharmaceutical waste is hazardous
West Tennessee Healthcare Part 3 Hazardous Pharmaceutical Waste Program: RCRA Waste Why are we concerned about hazardous pharmaceutical waste? EPA has determined that certain pharmaceutical waste is hazardous
Appendix L: Accessing/Deaccessing Implanted Central Venous Access Port
 Effective Date: 03/01/2008 Page 1 of 5 Recommendations for Use Insertion Considerations Implanted Port Dressing Access/ Reaccess An implanted port is strongly recommended for patients in whom more than
Effective Date: 03/01/2008 Page 1 of 5 Recommendations for Use Insertion Considerations Implanted Port Dressing Access/ Reaccess An implanted port is strongly recommended for patients in whom more than
General Lab Safety Rules and Practices SOP-GLSRP-01
 Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene
 Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene Presented By: Biological Safety http://biosafety.utk.edu Training Overview: This training is designed to: Orient new personnel
Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene Presented By: Biological Safety http://biosafety.utk.edu Training Overview: This training is designed to: Orient new personnel
Who is Bovie Animal Health?
 Who is Bovie Animal Health? The Animal Health team at Bovie is dedicated to selling first class Electrosurgery products and offering affordable solutions for use in veterinarian s exam room, surgery suite,
Who is Bovie Animal Health? The Animal Health team at Bovie is dedicated to selling first class Electrosurgery products and offering affordable solutions for use in veterinarian s exam room, surgery suite,
Figure A. Figure B To prevent premature activation of the needle safety guard, do not touch the NEEDLE GUARD ACTIVATION CLIPS at any time during use.
 INSTRUCTIONS FOR USE STELARA (stel ar a) (ustekinumab) injection, for subcutaneous use Instructions for injecting STELARA using a prefilled syringe. Read this Instructions for Use before you start using
INSTRUCTIONS FOR USE STELARA (stel ar a) (ustekinumab) injection, for subcutaneous use Instructions for injecting STELARA using a prefilled syringe. Read this Instructions for Use before you start using
Lenis Needle-free Safety Syringe Device User Manual
 Lenis Needle-free Safety Syringe Device User Manual 1 Table of Contents Welcome.3 Lenis Kit Components.4 Instructions 5-9 Maintenance and Care..10 Troubleshooting. 11 Warranty.12 Precautions 13 Return
Lenis Needle-free Safety Syringe Device User Manual 1 Table of Contents Welcome.3 Lenis Kit Components.4 Instructions 5-9 Maintenance and Care..10 Troubleshooting. 11 Warranty.12 Precautions 13 Return
Spring 2005 Pollution Prevention Workshop For Healthcare
 Spring 2005 Pollution Prevention Workshop For Healthcare Regulated Medical Waste Compliance Issues Daniel Salzler ADEQ Solid Waste Inspection & Compliance Unit Arizona Solid Waste Rules Arizona Administrative
Spring 2005 Pollution Prevention Workshop For Healthcare Regulated Medical Waste Compliance Issues Daniel Salzler ADEQ Solid Waste Inspection & Compliance Unit Arizona Solid Waste Rules Arizona Administrative
STANDARD: Laboratory Safety Effective: March 20, 2018
 University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # Approved: 10/3/18
 Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Instructions for Use. (dulaglutide) injection, for subcutaneous use. 1.5 mg/0.5 ml Single-Dose Pen. once weekly. Unfold and lay flat
 1 BREAK SEAL Instructions for Use TRULICITY (Trū-li-si-tee) BREAK SEAL (dulaglutide) injection, for subcutaneous use 1.5 mg/0.5 ml Single-Dose Pen once weekly Unfold and lay flat Read both sides for full
1 BREAK SEAL Instructions for Use TRULICITY (Trū-li-si-tee) BREAK SEAL (dulaglutide) injection, for subcutaneous use 1.5 mg/0.5 ml Single-Dose Pen once weekly Unfold and lay flat Read both sides for full
Package Leaks. OH&S Biosafety Emergency Response Document. Examine outer packaging. Leaks or evidence of leaks. No evidence of leaks
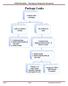 Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System. Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe
 Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Instructions For Use
 Instructions For Use 1 subcutaneous injection Welcome This guide contains information on how to use Rebif Rebidose, a pre assembled, single use autoinjector that administers one dose of Rebif (interferon
Instructions For Use 1 subcutaneous injection Welcome This guide contains information on how to use Rebif Rebidose, a pre assembled, single use autoinjector that administers one dose of Rebif (interferon
Laboratory Orientation. Biological Screening
 Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Product Name: Duro(R) All-Purpose Spray Adhesive Page 1 of 5
 Product Name: Duro(R) All-Purpose Spray Adhesive Page 1 of 5 Duro(R) All-Purpose Spray Adhesive 81088 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Part No.: MS-H Product Type: Aerosol Spray Adhesive
Product Name: Duro(R) All-Purpose Spray Adhesive Page 1 of 5 Duro(R) All-Purpose Spray Adhesive 81088 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Part No.: MS-H Product Type: Aerosol Spray Adhesive
Treatment Systemic Therapy. Your Guide to Subcutaneous Chemotherapy Self Injection
 Treatment Systemic Therapy Your Guide to Subcutaneous Chemotherapy Self Injection Table of Contents 1 Gather your supplies 2 Setting up for your self-injection 3 Preparing the injection 5 Choosing and
Treatment Systemic Therapy Your Guide to Subcutaneous Chemotherapy Self Injection Table of Contents 1 Gather your supplies 2 Setting up for your self-injection 3 Preparing the injection 5 Choosing and
Student Performance Guide. Student Performance Guide. Student Performance Guide. Student Performance Guide. LESSON 3-3 Bleeding Time
 LESSON 3-3 Bleeding Time Student Performance Guide LESSON 3-4 Prothrombin Time Student Performance Guide LESSON 3-5 Activated Partial Thromboplastin Time Student Performance Guide LESSON 3-6 Rapid Tests
LESSON 3-3 Bleeding Time Student Performance Guide LESSON 3-4 Prothrombin Time Student Performance Guide LESSON 3-5 Activated Partial Thromboplastin Time Student Performance Guide LESSON 3-6 Rapid Tests
Brazosport College Life Science Laboratory Safety Rules and Regulations
 Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml)
 Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml) Important Information The device is a single-dose disposable pen. It
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml) Important Information The device is a single-dose disposable pen. It
Original Date:
 Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
Disposal of Biological Waste
 Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
The following standard practices, safety equipment, and facility requirements apply to BSL-1:
 Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
INFECTION PREVENTION AND CONTROL SAFE USE AND DISPOSAL OF SHARPS
 INFECTION PREVENTION AND CONTROL SAFE USE AND DISPOSAL OF SHARPS Policy title Safe Use and Disposal of Sharps Infection Control and Prevention (IPC) Policy CL05C reference Policy category Clinical Relevant
INFECTION PREVENTION AND CONTROL SAFE USE AND DISPOSAL OF SHARPS Policy title Safe Use and Disposal of Sharps Infection Control and Prevention (IPC) Policy CL05C reference Policy category Clinical Relevant
Annual Associate Safety Module Hazardous Materials
 Annual Associate Safety Module Hazardous Materials Working Safely with Hazardous Materials What are Hazardous Materials? Any material or chemical that causes acute health problems, death or chronic illness.
Annual Associate Safety Module Hazardous Materials Working Safely with Hazardous Materials What are Hazardous Materials? Any material or chemical that causes acute health problems, death or chronic illness.
Safety Office -- Laboratory Inspection Form
 RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
ISO Sharps injury protection Requirements and test methods Sharps containers
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 23907 First edition 2012-09-01 Sharps injury protection Requirements and test methods Sharps containers Protection contre les blessures par perforants
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 23907 First edition 2012-09-01 Sharps injury protection Requirements and test methods Sharps containers Protection contre les blessures par perforants
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza Cincinnati, OH 45202 MATERIAL SAFETY DATA SHEET MSDS#: FH/C/2006/BCWT-6TCNFS Supersedes: 10/06 Issue Date: 11/06 Identity: Disinfecting Liquid
Page 1 of 5 Procter & Gamble CPG TN 6 2 Procter & Gamble Plaza Cincinnati, OH 45202 MATERIAL SAFETY DATA SHEET MSDS#: FH/C/2006/BCWT-6TCNFS Supersedes: 10/06 Issue Date: 11/06 Identity: Disinfecting Liquid
Table 6: Detailed Infection Prevention and Control Procedures for Tattooing and Micropigmentation. Use During Tattooing
 FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy
 The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy This policy applies to ALL sharps injuries where any hazardous substance (including, toxins, chemicals and human pathogens)
The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy This policy applies to ALL sharps injuries where any hazardous substance (including, toxins, chemicals and human pathogens)
Standard Operating Procedures
 Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
SPECIMEN COLLECTION. A Study Guide
 SPECIMEN COLLECTION A Study Guide Expectations At the UAP Marathon the UAP will be expected to verbalize the process of performing a peripheral blood draw and demonstrating the correct collection of a
SPECIMEN COLLECTION A Study Guide Expectations At the UAP Marathon the UAP will be expected to verbalize the process of performing a peripheral blood draw and demonstrating the correct collection of a
Henry Schein, Inc. 135 Duryea Road Melville, NY Henry Schein Bacitracin Zinc Ointment First Aid Antibiotic
 SAFETY DATA SHEET Henry Schein, Inc. Henry Schein, Inc. 135 Duryea Road Melville, NY 11747 800-472-4346 TRANSPORTATION EMERGENCY CALL CHEMTREC... : (800) 424-9300 INTERNATIONAL... : (703) 527-3887 Section
SAFETY DATA SHEET Henry Schein, Inc. Henry Schein, Inc. 135 Duryea Road Melville, NY 11747 800-472-4346 TRANSPORTATION EMERGENCY CALL CHEMTREC... : (800) 424-9300 INTERNATIONAL... : (703) 527-3887 Section
A ppendix 15 WUStL Bloodborne Pathogens Exposure Control Plan Research Laboratory-Specific Work Practices
 Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach
 Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Updated by S. McNew, March Deborah Jung Microbiology Preparation Technician
 Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
Imres can offer solutions for all types of health programmes in the international relief market PROVIDING GLOBAL ACCESS TO QUALITY MEDICAL CARE
 MEDICAL CONSUMABLES PROVIDING GLOBAL ACCESS TO QUALITY MEDICAL CARE P R O D U C T G U I D E Imres can offer solutions for all types of health programmes in the international relief market OUR RANGE OF
MEDICAL CONSUMABLES PROVIDING GLOBAL ACCESS TO QUALITY MEDICAL CARE P R O D U C T G U I D E Imres can offer solutions for all types of health programmes in the international relief market OUR RANGE OF
Uline. SECTION 1 : CHEMICAL PRODUCT AND COMPANY IDENTIFICATION MSDS Name: 3M 4004, 4008, 4026 AND 4032 DOUBLE COATED URETHANE FOAM TAPE
 Uline 3M 4004, 4008, 4026 AND 4032 DOUBLE COATED URETHANE FOAM TAPE Manufacturer MSDS Number: 07-0796-8 NFPA 1 2 0 SECTION 1 : CHEMICAL PRODUCT AND COMPANY IDENTIFICATION MSDS Name: 3M 4004, 4008, 4026
Uline 3M 4004, 4008, 4026 AND 4032 DOUBLE COATED URETHANE FOAM TAPE Manufacturer MSDS Number: 07-0796-8 NFPA 1 2 0 SECTION 1 : CHEMICAL PRODUCT AND COMPANY IDENTIFICATION MSDS Name: 3M 4004, 4008, 4026
Standard Operating Procedure for disposal of biological waste
 Standard Operating Procedure for disposal of biological waste Effective date: 22 nd March 2018 Review due date: 19 th March 2018 Original Author Name: Rebecca Toone Position: Technician Date: 29.07.2013
Standard Operating Procedure for disposal of biological waste Effective date: 22 nd March 2018 Review due date: 19 th March 2018 Original Author Name: Rebecca Toone Position: Technician Date: 29.07.2013
Migraine Attack Abortive Treatment Medication Overuse Protocol Treatment Refractory Cluster Headache Treatment
 D i h y d r o e r g o t a m i n e ( D H E ) S u b c u t a n e o u s I n j e c t i o n G u i d e Migraine Attack Abortive Treatment Medication Overuse Protocol Treatment Refractory Cluster Headache Treatment
D i h y d r o e r g o t a m i n e ( D H E ) S u b c u t a n e o u s I n j e c t i o n G u i d e Migraine Attack Abortive Treatment Medication Overuse Protocol Treatment Refractory Cluster Headache Treatment
KERN HEALTH SYSTEMS POLICIES AND PROCEDURES 2.21-P
 Page 1 of5 RESPONSIBLE DEPARTMENT HEAD: Director of Quality Improvement, Health Education and Disease Management Review Date 08/29/97 08/00 01102 08/2005 OS/2010 Effective Date 09/01103 11118/05 DS/;ZOhtJ
Page 1 of5 RESPONSIBLE DEPARTMENT HEAD: Director of Quality Improvement, Health Education and Disease Management Review Date 08/29/97 08/00 01102 08/2005 OS/2010 Effective Date 09/01103 11118/05 DS/;ZOhtJ
SOFT CARE SENSITIVE FOAM
 SAFETY DATA SHEET SOFT CARE SENSITIVE FOAM Version Number: 5 Preparation date: 2017-03-01 1. IDENTIFICATION Product name: SOFT CARE SENSITIVE FOAM Product Code: 100907880 SDS #: MS0301557 Recommended use:
SAFETY DATA SHEET SOFT CARE SENSITIVE FOAM Version Number: 5 Preparation date: 2017-03-01 1. IDENTIFICATION Product name: SOFT CARE SENSITIVE FOAM Product Code: 100907880 SDS #: MS0301557 Recommended use:
SAFETY DATA SHEET PROUDS OFF OINTMENT SECTION 1: IDENTIFICATION. Basic copper, Copper sulfate basic, Fixed copper
 SAFETY DATA SHEET PROUDS OFF OINTMENT SECTION 1: IDENTIFICATION Product Identifier: Synonyms: Recommended Use: Restrictions on Use: Manufactured for: Distributed by: Prouds Off Ointment Basic copper, Copper
SAFETY DATA SHEET PROUDS OFF OINTMENT SECTION 1: IDENTIFICATION Product Identifier: Synonyms: Recommended Use: Restrictions on Use: Manufactured for: Distributed by: Prouds Off Ointment Basic copper, Copper
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS
 SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT
 UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
CCS Administrative Procedure T Biosafety for Laboratory Settings
 CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
Soft Care Deluxe Foam
 SAFETY DATA SHEET Soft Care Deluxe Foam Version Number: 6 Preparation date: 2017-03-01 1. IDENTIFICATION Product name: Soft Care Deluxe Foam Product Code: 100907879 SDS #: MS0301546 Recommended use: HandSoap
SAFETY DATA SHEET Soft Care Deluxe Foam Version Number: 6 Preparation date: 2017-03-01 1. IDENTIFICATION Product name: Soft Care Deluxe Foam Product Code: 100907879 SDS #: MS0301546 Recommended use: HandSoap
Appendix C. Infectious Waste Guidelines
 Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
SAFETY DATA SHEETS. This SDS packet was issued with item:
 SAFETY DATA SHEETS This SDS packet was issued with item: 078080905 The safety data sheets (SDS) in this packet apply to the individual products listed below. Please refer to invoice for specific item number(s).
SAFETY DATA SHEETS This SDS packet was issued with item: 078080905 The safety data sheets (SDS) in this packet apply to the individual products listed below. Please refer to invoice for specific item number(s).
Gum Remover. Version Number: 1 Preparation date: IDENTIFICATION. Uses other than those identified are not recommended
 SAFETY DATA SHEET Version Number: 1 Preparation date: 2014-05-19 1. IDENTIFICATION Product name: Product Code: 95628817 SDS #: MS0800327 Recommended use: Uses advised against: Uses other than those identified
SAFETY DATA SHEET Version Number: 1 Preparation date: 2014-05-19 1. IDENTIFICATION Product name: Product Code: 95628817 SDS #: MS0800327 Recommended use: Uses advised against: Uses other than those identified
VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers
 Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
DOVE Moisturizing Gentle Hand Cleaner
 SAFETY DATA SHEET DOVE Moisturizing Gentle Hand Cleaner Version Number: 3 Preparation date: 2017-05-30 1. IDENTIFICATION Product name: DOVE Moisturizing Gentle Hand Cleaner Product Code: 2979401 SDS #:
SAFETY DATA SHEET DOVE Moisturizing Gentle Hand Cleaner Version Number: 3 Preparation date: 2017-05-30 1. IDENTIFICATION Product name: DOVE Moisturizing Gentle Hand Cleaner Product Code: 2979401 SDS #:
Dove Hair & Body Wash
 SAFETY DATA SHEET Dove Hair & Body Wash Version Number: 3 Preparation date: 2017-05-30 1. IDENTIFICATION Product name: Dove Hair & Body Wash Product Code: 100948782 SDS #: MS0801010 Recommended use: Personalcareproduct
SAFETY DATA SHEET Dove Hair & Body Wash Version Number: 3 Preparation date: 2017-05-30 1. IDENTIFICATION Product name: Dove Hair & Body Wash Product Code: 100948782 SDS #: MS0801010 Recommended use: Personalcareproduct
MATERIAL SAFETY DATA SHEET
 1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER VASELINE BODY MOISTURISERS Product Name/s: Variant/s Manufacturer s Product Codes Vaseline Dry Skin 20249840 25mL 61061794 35mL 61038808 75mL 20249848 225mL
1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER VASELINE BODY MOISTURISERS Product Name/s: Variant/s Manufacturer s Product Codes Vaseline Dry Skin 20249840 25mL 61061794 35mL 61038808 75mL 20249848 225mL
SDS. GHS Safety Data Sheet. Blistex Inc. Blistex Lip Balm - Regular PRODUCT AND COMPANY IDENTIFICATION. Manufacturer
 of 5 PRODUCT AND COMPANY IDENTIFICATION Manufacturer 800 Swift Drive Oak Brook, IL 60523 Contact: Phone: Fax: Email: Web: Environmental, Health and Safety Coordinator (630) 57-2870 (630) 57-4265 msds@blistex.com
of 5 PRODUCT AND COMPANY IDENTIFICATION Manufacturer 800 Swift Drive Oak Brook, IL 60523 Contact: Phone: Fax: Email: Web: Environmental, Health and Safety Coordinator (630) 57-2870 (630) 57-4265 msds@blistex.com
Annual Associate Safety Module. Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan
 Annual Associate Safety Module Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan Since you work in a healthcare facility, you may have potential exposure to blood or body fluids.
Annual Associate Safety Module Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan Since you work in a healthcare facility, you may have potential exposure to blood or body fluids.
LAB 5 Blood Collection
 LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
NOT CLASSIFIED AS HAZARDOUS ACCORDING TO CRITERIA OF NOHSC. International Beauty Supplies. 6e Narabang Way, Belrose NSW, Australia 2085
 1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address SOIN DU CORPS CYOSLIM LOTION CRYOTONIQUE SB81324 BODY SKIN CARE International Beauty Supplies
1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address SOIN DU CORPS CYOSLIM LOTION CRYOTONIQUE SB81324 BODY SKIN CARE International Beauty Supplies
Medical Waste Manual. California State University, Chico
 California State University, Chico The Department of Environmental Health and Safety March 2017 TABLE OF CONTENTS Section Page 1.0 Introduction... 1-1 2.0 Containment, Storage, and Disposal of Biohazardous
California State University, Chico The Department of Environmental Health and Safety March 2017 TABLE OF CONTENTS Section Page 1.0 Introduction... 1-1 2.0 Containment, Storage, and Disposal of Biohazardous
WHMIS. Material Safety Data Sheet. Product Information VP-TECH SECTION 1: MATERIAL IDENTIFICATION SECTION 2: HAZARDOUS INGREDIENTS
 Page 1 of 7 WHMIS Material Safety Data Sheet Product Information VP-TECH SECTION 1: MATERIAL IDENTIFICATION CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Emergency: Chemtrek Telephone: 1-800-424-9300 General
Page 1 of 7 WHMIS Material Safety Data Sheet Product Information VP-TECH SECTION 1: MATERIAL IDENTIFICATION CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Emergency: Chemtrek Telephone: 1-800-424-9300 General
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS
 Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
x. ANNUAL REVIEW SIGNATURE SHEET
 x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
SAFETY DATA SHEET Consumer Product
 Page 1 of 5 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: GENERAL USE: Hair Care PRODUCT DESCRIPTION: CHEMICAL FAMILY: Personal care GENERIC NAME: Hair conditioner MANUFACTURER DISTRIBUTOR Revlon
Page 1 of 5 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: GENERAL USE: Hair Care PRODUCT DESCRIPTION: CHEMICAL FAMILY: Personal care GENERIC NAME: Hair conditioner MANUFACTURER DISTRIBUTOR Revlon
TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee
 Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Bench-top Biohazard Bags. CAtALoG No. SIzE DESCRIPtIoN thickness
 Bench-top Biohazard Bags Easy Open, Dispose Used Tips and Tubes Bright red, autoclavable bags feature one long flap for easy opening. Their size allows the bag to be inverted over your hand to safely pick
Bench-top Biohazard Bags Easy Open, Dispose Used Tips and Tubes Bright red, autoclavable bags feature one long flap for easy opening. Their size allows the bag to be inverted over your hand to safely pick
Material Safety Data Sheet This MSDS is prepared in accordance with OSHA 29 CFR
 Material Safety Data Sheet This MSDS is prepared in accordance with OSHA 29 CFR 1910.1200 CLASS B-2: Flammable liquid with a flash point lower than 7.8 C (100 F). Class: Flammable liquid having a flash
Material Safety Data Sheet This MSDS is prepared in accordance with OSHA 29 CFR 1910.1200 CLASS B-2: Flammable liquid with a flash point lower than 7.8 C (100 F). Class: Flammable liquid having a flash
NOT CLASSIFIED AS HAZARDOUS ACCORDING TO CRITERIA OF NOHSC. International Beauty Supplies. 6e Narabang Way, Belrose NSW, Australia 2085
 1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address Mango Exfoliating Scrub BFCM04MS, BFCM08MS, BFCM20MS, BFCM1GMS Removes Dead Skin Cells International
1. IDENTIFICATION OF THE PREPARATION AND THE COMPANY Product name Product code Product use Company Address Mango Exfoliating Scrub BFCM04MS, BFCM08MS, BFCM20MS, BFCM1GMS Removes Dead Skin Cells International
J e w e l l e r y R e p a i r ( 1 2 A )
 8 8 2 4 J e w e l l e r y R e p a i r ( 1 2 A ) 40S/40E/40M A Jewellery/Metalsmithing Course 8 8 2 4 : J e w e l l e r y R e p a i r ( 1 2 A ) 4 0 S / 4 0 E / 4 0 M Course Description This course is intended
8 8 2 4 J e w e l l e r y R e p a i r ( 1 2 A ) 40S/40E/40M A Jewellery/Metalsmithing Course 8 8 2 4 : J e w e l l e r y R e p a i r ( 1 2 A ) 4 0 S / 4 0 E / 4 0 M Course Description This course is intended
INJECTING EFFICIENCY FROM SET-UP TO CLEAN-UP.
 INJECTING EFFICIENCY FROM SET-UP TO CLEAN-UP. I-OPS Instrument Delivery System. A new best practice for your practice. Tradition and Innovation Since 1898, visionary thinking and a fascination with technology
INJECTING EFFICIENCY FROM SET-UP TO CLEAN-UP. I-OPS Instrument Delivery System. A new best practice for your practice. Tradition and Innovation Since 1898, visionary thinking and a fascination with technology
BSL2 Exposure Control Plan: Human or Non Human Primate Materials
 Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
MATERIAL SAFETY DATA SHEET 7/27/2012
 MATERIAL SAFETY DATA SHEET 7/27/2012 MANUFACTURER OR DISTRIBUTOR: Speedball Art Products Co. P.O. Box 5157 2301 Speedball Road Statesville, North Carolina 28677 INFORMATION TELEPHONE NUMBER: 704-978-4166
MATERIAL SAFETY DATA SHEET 7/27/2012 MANUFACTURER OR DISTRIBUTOR: Speedball Art Products Co. P.O. Box 5157 2301 Speedball Road Statesville, North Carolina 28677 INFORMATION TELEPHONE NUMBER: 704-978-4166
Canon Laser Consumable Return Program
 Canon Laser Consumable Return Program Canon s Overview on the Environment KYOSEI Living and working together for the common good. We at Canon are guided by the philosophy of Kyosei living and working
Canon Laser Consumable Return Program Canon s Overview on the Environment KYOSEI Living and working together for the common good. We at Canon are guided by the philosophy of Kyosei living and working
