Safety Office -- Laboratory Inspection Form
|
|
|
- Reynold McCormick
- 5 years ago
- Views:
Transcription
1 RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual inspection required for renewal of protocol(s) PI: Building: Rooms: Date of Inspection: Inspectors: Item # Item Yes No N/A Comments 1.0 SIGNAGE 1.1 Laboratory Door(s) have up-to-date postings of applicable signs (hazards, safety, animals, etc.). 1.2 Hazard signs (Chemical, Bio, Acid, Flammable, etc.) are posted as appropriate on refrigerators, freezers, storage areas. 2.0 DOCUMENTATION & TRAINING 2.1 Laboratory Protocols are easily available to staff 2.2 All personnel know how to access the Scott & White Bloodborne Pathogen Exposure Control Plan 2.3 All personnel know how to access MSDS 2.4 An up-to-date inventory of chemicals is on hand 2.5 All personnel have taken a laboratory safety course within the past year
2 2.6 All personnel who work with human blood, tissues or OPIM have taken the Bloodborne Pathogen Training within the past year 2.7 All personnel have had radiation safety training, if applicable 2.8 If laboratory is designated as BSL-2, personnel have had specific training 2.9 All persons entering the laboratory are informed of potential hazards 2.10 All personnel are provided medical surveillance and offered appropriate immunizations for agents handled or potentially present in the laboratory 3.0 CHEMICAL SAFETY 3.1 All chemicals are labeled with the full chemical name and are in English 3.2 Chemical containers are in good condition 3.3 Legacy/obsolete chemicals (inherited/unused for 10+ years) are disposed of properly 3.4 Chemicals are stored by compatibility (flammables stored separately from oxidizers, etc.) 3.5 Chemicals are stored in appropriate locations (flammables are in a flammables cabinet, etc) 3.6 Shelves, cabinets and counter tops are stable and in good condition for chemical storage 3.7 Chemicals are not stored on the floor Special Chemical Hazards 3.8 Hazardous chemicals are not disposed of in the general waste or down the sanitary sewer 3.9 Chemicals are stored in such a way as to prevent release to the envrionment 3.10 Volatiles are stored in an explosion-proof refrigerator 3.11 Special chemical hazards are stored appropriately and in compatibility groups (i.e., carcinogens, teratogens, acetyl cholinesterase inhibitors, pyrophoric compounds, etc.) 3.12 Written procedures are in place for use of acutely hazardous chemicals (carcinogens, reproductive hazards, etc.) 2
3 3.13 Alternatives to mercury are used, if possible (including non-mercury containing thermometers) 3.14 Chemical fume hoods are available for use with relevant hazards. Fume hoods need to be certified on an annual basis and must be used appropriately. DEA Controlled Substances 3.15 Federal DEA License is available 3.16 Texas Department of Public Safety License is available 3.17 DEA-regulated items are stored in an immobile, locked container 3.18 Lab has proper record keeping of stock, usage and disposal of DEA-regulated items 3.19 Expired controlled substances are disposed of properly Compressed Gas Cylinders 3.20 Gas cylinders are tagged as empty or full 3.21 Gas cylinders are labeled for contents 3.22 Cylinders are secured to a stationary surface by a chain link or strap 3.23 Cylinders are capped when not in use and have a pressure regulator when in use 4.0 BIOLOGICAL SAFETY General Biosafety 4.1 Laboratory supervisor must enforce policies that control access to the laboratory 4.2 Laboratory personnel understand and practice universal precautions 4.3 Laboratory personnel must wash their hands after working with potentially hazardous material and before leaving the laboratory 4.4 Laboratory personnel are up-to-date with any required immunizations 4.5 Eating, drinking, smoking, applying cosmetics, handling contact lenses and storing food for human consumption is not permitted in the laboratory. 3
4 4.6 Appropriate PPE is available for workers to use and is utilized. No shorts or open-toed shoes are permitted in the laboratory. Sharps 4.7 The laboratory minimizes the usage of sharps (needles, syringes, scalpels, razor blades) 4.8 Needles are not re-capped 4.9 Policies for the safe handling of sharps have been developed and implemented 4.10 Sharps containers are readily available and utilized. Sharps containers are never allowed to fully fill and are disposed of when ¾ full Sharps containers associated with biohazardous materials are disposed of through biohazard waste 4.12 Broken glass is not handled directly, but is removed using other implements such as a brush and dustpan, tongs, or forceps Glassware is disposed of in broken glass boxes containers lined with plastic liners Glassware associated with biohazardous materials is discarded through biohazard waste PPE 4.15 Gloves are appropriate for the hazards in the laboratory and worn when required Gloves are changed when integrity is compromised, when contaminated, or when otherwise necessary 4.17 Safety glasses with side protection, meeting ANSI Z87.1 are available and worn when appropriate 4.18 Goggles are available and used when there is a potential for splash or splatters 4.19 Face shields are available and used when needed 4.20 Lab coats and/or other protective clothing is worn to minimize exposure to hazardous material (gowns, shoe covers, masks, respirators) 4
5 4.21 If respirators are required for work with hazardous agents, personnel are appropriately trained and fittested on an annual basis Facilities and Equipment 4.22 Laboratories have doors for controlled access 4.23 Laboratory has a sink for hand washing 4.24 Laboratory furniture must be stable and capable of supporting anticipated loads from equipment and supplies 4.25 Bench tops must be impervious to water and resistant to heat, organic solvents, etc. They must be non-porous and able to be decontaminated 4.26 Chairs or stools in the laboratory must be covered with a non-porous material that can be cleaned and decontaminated 4.27 An effective pest management program must be in place for BSL-2 laboratories 4.28 Appropriate biological safety cabinets are available for use with hazardous agents and any procedures that may produce splashes or aerosols 4.29 Biological safety cabinets are up-to-date and certified on an annual basis 4.30 Personnel have been instructed on the proper usage of biological safety cabinets and can demonstrate this 4.31 Surfaces within the biological safety cabinets are disinfected prior to and after use, with appropriate disinfectant solutions 4.32 Bunsen burners and/or open flames are not used in biosafety cabinets 4.33 Centrifuges used with biohazardous material must utilize cups to prevent aerosol generation. If microcentrifugation of biohazardous material is conducted, it should be performed within a biological safety cabinet Vacuum lines must be protected with in-line HEPA filters. Liquid disinfectant traps should be utilized to prevent contamination 5
6 4.35 No mouth pipetting is permitted. Mechanical pipettors must be used at all times Eye wash stations are available. These must be ANSI-approved stations and be located no more than 10 seconds travel time from the work space. 5.0 EMERGENCY PROCEDURES 5.1 The laboratory has a written emergency plan. 5.2 Personnel know how to report accidents, injuries and exposures 5.3 Eye wash stations are tested on a weekly basis by laboratory personnel (documented). Eye washes are free of obstruction and protective caps are in place. 5.4 A safety shower is available 5.5 Personnel understand spill procedures for biological hazards and a spill kit is available in the laboratory. 5.6 Procedures are in place for notification of the Safety Office and other responsible personnel. 6.0 FIRE SAFETY 6.1 The correct type of fire extinguisher is present in the laboratory 6.2 All personnel have had fire extinguisher education or training by either: 1) Attending a hands-on training; 2) Viewing a Fire Extinguisher training video; or 3) Reading Fire Educational material 6.3 An annual inspection of the fire extinguisher is conducted 6.4 The extinguisher is not obstructed 6.5 Fire alarm pull stations are not obstructed 7.0 ANIMAL USE 7.1 Are animals brought into the lab space for procedures? 7.2 If yes to 7.1, is there IACUC approval to bring animals into the lab space? 7.3 Are personnel educated on hazards of working with animals, including allergies? 6
7 7.4 Are any perfusion experiments with formaldehyde performed in the lab space? 7.5 Are personnel monitored for formaldehyde exposure? 7.6 Is isoflurane used in the lab space? 7.7 If yes to 7.5, do you use an isoflurane vaporizer? 7.8 If yes to 7.6, when was the vaporizer last certified? 8.0 LABORATORY BIOLOGICAL WASTE DISPOSAL/DECONTAMINATION 8.1 Is an autoclave available for decontamination? 8.2 Is biohazardous waste placed into proper bags (orange or red bags that have the biohazard symbol)? 8.3 Are the biohazardous waste containers labeled with the biohazard symbol and the word biohazard? 8.4 Biohazard containers are closed except when adding waste 8.5 Biohazards are not put in regular trash or flushed down the sink 8.6 Biohazard waste is not mixed with chemical waste 8.7 Biohazard waste is disposed of through the MedSharps program? 8.8 Biohazard waste for disposal is properly packed and sealed in leak-proof containers and properly stored before pickup? 8.9 Regular trash is not placed into biohazardous waste 8.10 Biohazardous sharps containers are packaged properly into biohazard waste containers Liquid biohazardous waste is decontaminated using approved dinsinfectants prior to placing in biohazardous waste containers 8.12 Equipment must be decontaminated before repair, maintenance or removal from the laboratory Item # Item Yes No N/A Comments 9.0 SHIPPING 9.1 Does the laboratory ship biohazardous material? 9.2 If yes, has a laboratory representative undergone 7
8 required shipping training for shipping infectious and biological substances in the past 2 years? 9.3 If yes to 9.2, please list the person here: Name: Training Date: 8
Roosevelt Biosafety Training. Created 10/2015
 Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
List any references used for the procedure design (research publications, etc.):
 Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Provide a brief description of the procedure and infectious organisms used:
 Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Biological Safety Training
 Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Bloodborne Pathogens Exposure Control Plan. December 2003
 Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Biosafety Self-Audit Checklist
 Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
The following standard practices, safety equipment, and facility requirements apply to BSL-1:
 Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
BSL-2 Emergency Plan
 BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
General Lab Safety Rules and Practices SOP-GLSRP-01
 Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee
 Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Brazosport College Life Science Laboratory Safety Rules and Regulations
 Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
BIOLOGICAL SAFETY INSPECTION CHECKLIST
 BIOLOGICAL SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
BIOLOGICAL SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # Approved: 10/3/18
 Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN
 FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
Self-Inspection 2018 Biosafety Containment Level 2 Requirements To be verified at an Inspection by Biohazard Committee Members
 Self-nspection 2018 To be verified at an nspection by Biohazard Committee Members Containment requirements of the Canadian Biosafety Standard, 2 nd Edition, 2015, published by the Public Health gency of
Self-nspection 2018 To be verified at an nspection by Biohazard Committee Members Containment requirements of the Canadian Biosafety Standard, 2 nd Edition, 2015, published by the Public Health gency of
CCS Administrative Procedure T Biosafety for Laboratory Settings
 CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
Standard Operating Procedure for Biosafety Cabinet Use
 NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
Safety Rules for Laboratory
 Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Case Western Reserve University Department of Environmental Health & Safety
 Case Western Reserve University Department of Environmental Health & Safety Laboratory Specific Supplement: CWRU Exposure Control Plan for Biohazards (including Bloodborne Pathogens) All laboratories at
Case Western Reserve University Department of Environmental Health & Safety Laboratory Specific Supplement: CWRU Exposure Control Plan for Biohazards (including Bloodborne Pathogens) All laboratories at
Emergency Procedures Specific Biological Spill Clean-Up Guidelines
 Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
A ppendix 15 WUStL Bloodborne Pathogens Exposure Control Plan Research Laboratory-Specific Work Practices
 Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Biohazardous Waste. 1. Solid Biohazardous Waste (non-sharps) Storage
 Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach
 Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Bloodborne Pathogens Exposure Control Plan
 Bloodborne Pathogens Exposure Control Plan Environmental Health, Safety, and Risk Management Department Box 6113, SFA Station Nacogdoches, Texas 75962-6113 January 2011 Revised May 2017 APPLICABILITY These
Bloodborne Pathogens Exposure Control Plan Environmental Health, Safety, and Risk Management Department Box 6113, SFA Station Nacogdoches, Texas 75962-6113 January 2011 Revised May 2017 APPLICABILITY These
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS
 Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
Introduction. BSL Level 1-4 is also different from Risk Group 1-4 as described earlier but is very much related to each other.
 LABORATORY BIOSAFETY CONTAINMENT LEVEL Introduction Laboratory Biosafety Containment Level or often known as Bio Safety Level (BSL) is referred to the containment level of the laboratory setting (including
LABORATORY BIOSAFETY CONTAINMENT LEVEL Introduction Laboratory Biosafety Containment Level or often known as Bio Safety Level (BSL) is referred to the containment level of the laboratory setting (including
Standard Operating Procedures
 Standard Operating Procedures Laboratory Specific Chemical: Formaldehyde Please fill out the form completely. Print a copy and insert into your Chemical Hygiene Plan. Department: Date when SOP was written:
Standard Operating Procedures Laboratory Specific Chemical: Formaldehyde Please fill out the form completely. Print a copy and insert into your Chemical Hygiene Plan. Department: Date when SOP was written:
BSL2 Exposure Control Plan: Human or Non Human Primate Materials
 Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Disposal of Biohazard Wastes
 4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
LABORATORY SAFETY SERIES: The OSHA Formaldehyde Standard
 ERI Safety Videos Videos for Safety Meetings 2963 LABORATORY SAFETY SERIES: The OSHA Formaldehyde Standard Leader s Guide Marcom Group Ltd. INTRODUCTION TO THE PROGRAM Structure and Organization Information
ERI Safety Videos Videos for Safety Meetings 2963 LABORATORY SAFETY SERIES: The OSHA Formaldehyde Standard Leader s Guide Marcom Group Ltd. INTRODUCTION TO THE PROGRAM Structure and Organization Information
Building/Lab Room No(s): Biosafety Containment level: BSL Click here to enter text.
 Click here to enter text. Laboratory Biosafety Manual Building/Lab Room No(s): Biosafety Containment level: BSL Click here to enter text. Date: Click here to enter text. Expires One year from the above
Click here to enter text. Laboratory Biosafety Manual Building/Lab Room No(s): Biosafety Containment level: BSL Click here to enter text. Date: Click here to enter text. Expires One year from the above
University Of Florida. Bloodborne Pathogen Program. Standard Operating Procedures
 University Of Florida Bloodborne Pathogen Program Standard Operating Procedures Revised February 9, 2011 Updated (annually) BBP Standard Operating Procedures Page 1 of 13 University Of Florida Bloodborne
University Of Florida Bloodborne Pathogen Program Standard Operating Procedures Revised February 9, 2011 Updated (annually) BBP Standard Operating Procedures Page 1 of 13 University Of Florida Bloodborne
SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
STANDARD: Laboratory Safety Effective: March 20, 2018
 University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
Welcome to the Hazard Communication Course
 Welcome to the Hazard Communication Course THE GLOSSARY A glossary is included in the Resources section on the home page of this course and on the OH&S website. These terms will be on the quiz. THE HAZARD
Welcome to the Hazard Communication Course THE GLOSSARY A glossary is included in the Resources section on the home page of this course and on the OH&S website. These terms will be on the quiz. THE HAZARD
Appendix C. Infectious Waste Guidelines
 Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
Emergency Response and Biohazard Exposure Control Plan IBC Approved: 10/3/18
 Institutional Biosafety Committee Emergency Response and IBC Approved: 10/3/18 Table of Contents I. PURPOSE... 3 II. DEFINITIONS... 3 III. RESPONSIBILTIES... 4 IV. BIOHAZARDOUS SPILL EMERGENCY PREPAREDNESSS...
Institutional Biosafety Committee Emergency Response and IBC Approved: 10/3/18 Table of Contents I. PURPOSE... 3 II. DEFINITIONS... 3 III. RESPONSIBILTIES... 4 IV. BIOHAZARDOUS SPILL EMERGENCY PREPAREDNESSS...
Regulated Medical Waste. Be sure to sign in!
 Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Package Leaks. OH&S Biosafety Emergency Response Document. Examine outer packaging. Leaks or evidence of leaks. No evidence of leaks
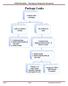 Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT
 UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
Type of Application (Check One) New Protocol Revised Protocol Project Duration Start Date: End Date:
 Page 1 of 11 INSTITUTIONAL BIOSAFETY COMMITTEE Winston-Salem State University Application for the Use of Biohazardous Materials, Recombinant DNA and Infectious Agents 1. APPLICANT INFORMATION Assigned
Page 1 of 11 INSTITUTIONAL BIOSAFETY COMMITTEE Winston-Salem State University Application for the Use of Biohazardous Materials, Recombinant DNA and Infectious Agents 1. APPLICANT INFORMATION Assigned
Updated by S. McNew, March Deborah Jung Microbiology Preparation Technician
 Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET
 TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
Laboratory Orientation. Biological Screening
 Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
GENERAL CHEMISTRY LABORATORY SAFETY
 GENERAL CHEMISTRY LABORATORY SAFETY 1. Read the experiment before coming to Keyes 405. The more prepared you are, the safer and more efficient you will be in lab. 2. Think about what you need to wear to
GENERAL CHEMISTRY LABORATORY SAFETY 1. Read the experiment before coming to Keyes 405. The more prepared you are, the safer and more efficient you will be in lab. 2. Think about what you need to wear to
Disposal of Biological Waste
 Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
The Aim Of Biosafety Training Is To Increase Your Ability To Recognize And Reduce Hazards In a BSL1 Lab
 The Aim Of Biosafety Training Is To Increase Your Ability To Recognize And Reduce Hazards In a BSL1 Lab Think before you do anything What could possibly happen? What is the worst thing that could happen?
The Aim Of Biosafety Training Is To Increase Your Ability To Recognize And Reduce Hazards In a BSL1 Lab Think before you do anything What could possibly happen? What is the worst thing that could happen?
Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene
 Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene Presented By: Biological Safety http://biosafety.utk.edu Training Overview: This training is designed to: Orient new personnel
Standard Microbiological Practices: Basic Biosafety Principles & Lab Hygiene Presented By: Biological Safety http://biosafety.utk.edu Training Overview: This training is designed to: Orient new personnel
OSHA: Occupational Safety and Health Administration PPE Personal protective equipment
 Bloodborne Pathogens University of Tennessee Safety Program HM-010 Document Contact: EHS Date effective: March 15, 2011 Revision Date: October 2, 2017 Purpose The purpose of this written program is to
Bloodborne Pathogens University of Tennessee Safety Program HM-010 Document Contact: EHS Date effective: March 15, 2011 Revision Date: October 2, 2017 Purpose The purpose of this written program is to
INFECTION PREVENTION AND CONTROL PLAN (IPCP)
 INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
ECU Radiation, Biosafety and Hazardous Substances Committee
 Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Section 4 Procedures for Biohazard Control
 Page 4-1 Section 4 Procedures for Biohazard Control Contents SECTION 4 PROCEDURES FOR BIOHAZARD CONTROL... 4-1 A. FACILITY REQUIREMENTS... 4-3 1. BSL-1 Laboratory Facilities... 4-3 2. BSL-2 Laboratory
Page 4-1 Section 4 Procedures for Biohazard Control Contents SECTION 4 PROCEDURES FOR BIOHAZARD CONTROL... 4-1 A. FACILITY REQUIREMENTS... 4-3 1. BSL-1 Laboratory Facilities... 4-3 2. BSL-2 Laboratory
Handling and Disposing of Needles
 Guidance Document UBC-RMS-OHS-GDL 14-008 Effective date: June 4, 2014 Review date: June 4, 2014 Supersedes: N/A 1. SCOPE Handling and Disposing of Needles This guidance document on Handling and Disposing
Guidance Document UBC-RMS-OHS-GDL 14-008 Effective date: June 4, 2014 Review date: June 4, 2014 Supersedes: N/A 1. SCOPE Handling and Disposing of Needles This guidance document on Handling and Disposing
THUNDER BAY REGIONAL RESEARCH INSTITUTE STATEMENT of POLICY and PROCEDURE Manual: Joint Health & Safety SPP No. JH Section: Issued: May 6, 2011
 Page: 1 of 6 POLICY This policy outlines the safety, access approval process, and training requirements, for the Thunder Bay Regional Research Institute s (TBRRI) Machine Shop, located within TBRRI s facilities
Page: 1 of 6 POLICY This policy outlines the safety, access approval process, and training requirements, for the Thunder Bay Regional Research Institute s (TBRRI) Machine Shop, located within TBRRI s facilities
Infection Control 101
 Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
GENERAL LAB SAFETY RULES. The following is a small list of the rules that MUST be followed in the lab!
 LABORATORY SAFETY GENERAL LAB SAFETY RULES The following is a small list of the rules that MUST be followed in the lab! General Lab Safety Rules: 1. Wear Goggles and Gloves when instructed. - Safety goggles
LABORATORY SAFETY GENERAL LAB SAFETY RULES The following is a small list of the rules that MUST be followed in the lab! General Lab Safety Rules: 1. Wear Goggles and Gloves when instructed. - Safety goggles
Standard Operating Procedures
 Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
Spring 2005 Pollution Prevention Workshop For Healthcare
 Spring 2005 Pollution Prevention Workshop For Healthcare Regulated Medical Waste Compliance Issues Daniel Salzler ADEQ Solid Waste Inspection & Compliance Unit Arizona Solid Waste Rules Arizona Administrative
Spring 2005 Pollution Prevention Workshop For Healthcare Regulated Medical Waste Compliance Issues Daniel Salzler ADEQ Solid Waste Inspection & Compliance Unit Arizona Solid Waste Rules Arizona Administrative
x. ANNUAL REVIEW SIGNATURE SHEET
 x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
Standard Operating Procedure: Hydrofluoric Acid. Copyright Drexel University Health and Safety
 Standard Operating Procedure: Hydrofluoric Acid Copyright Drexel University Health and Safety Hydrofluoric Acid (HF) Purpose of Training The Purpose of this training session is to develop safe working,
Standard Operating Procedure: Hydrofluoric Acid Copyright Drexel University Health and Safety Hydrofluoric Acid (HF) Purpose of Training The Purpose of this training session is to develop safe working,
PUBLIC HEALTH DEPARTMENT
 ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
MATERIAL SAFETY DATA SHEET SECTION 1 - PRODUCT INFORMATION SUPERPLASTICIZER SECTION 2 - HAZARD IDENTIFICATION
 MATERIAL SAFETY DATA SHEET SUPERPLASTICIZER SECTION 1 - PRODUCT INFORMATION PRODUCT NAME: SUPERPLASTICIZER CHEMICAL NAME: Sodium salt Naphthalene sulfonate formaldehyde condensate CAS NUMBER: 9084-06-4
MATERIAL SAFETY DATA SHEET SUPERPLASTICIZER SECTION 1 - PRODUCT INFORMATION PRODUCT NAME: SUPERPLASTICIZER CHEMICAL NAME: Sodium salt Naphthalene sulfonate formaldehyde condensate CAS NUMBER: 9084-06-4
Bloodborne Pathogens Exposure Control Plan
 Bloodborne Pathogens Exposure Control Plan Document History Version Date Comments 0.2 January, 2018 Program Review Foreword This written program is site specific to UVa Facilities Management and is in
Bloodborne Pathogens Exposure Control Plan Document History Version Date Comments 0.2 January, 2018 Program Review Foreword This written program is site specific to UVa Facilities Management and is in
DIVISION OF MATHEMATICS, SCIENCE & ENGINEERING
 DIVISION OF MATHEMATICS, SCIENCE & ENGINEERING Formaldehyde Safety Program Updated March, 2014 A. PURPOSE 1. The purpose of this program is to protect employees from the hazards associated with formaldehyde
DIVISION OF MATHEMATICS, SCIENCE & ENGINEERING Formaldehyde Safety Program Updated March, 2014 A. PURPOSE 1. The purpose of this program is to protect employees from the hazards associated with formaldehyde
Annual Associate Safety Module Hazardous Materials
 Annual Associate Safety Module Hazardous Materials Working Safely with Hazardous Materials What are Hazardous Materials? Any material or chemical that causes acute health problems, death or chronic illness.
Annual Associate Safety Module Hazardous Materials Working Safely with Hazardous Materials What are Hazardous Materials? Any material or chemical that causes acute health problems, death or chronic illness.
Introductory Chemistry
 Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
Deadly Bloodborne Diseases
 What and Why This Refresher Blood Borne Pathogens on-line training is offered for all returning employees of Harnett County Schools who have previously completed the Initial BBP training video. This is
What and Why This Refresher Blood Borne Pathogens on-line training is offered for all returning employees of Harnett County Schools who have previously completed the Initial BBP training video. This is
San Bernardino Valley College. Blood Borne Pathogens. Exposure Control Program
 San Bernardino Valley College Blood Borne Pathogens Exposure Control Program December 7, 2009 I. PURPOSE The Blood Borne Pathogens Exposure Control Program (BBP) has been developed by San Bernardino Valley
San Bernardino Valley College Blood Borne Pathogens Exposure Control Program December 7, 2009 I. PURPOSE The Blood Borne Pathogens Exposure Control Program (BBP) has been developed by San Bernardino Valley
Phenol Safety Training. ENVIRONMENTAL Health & Safety and Risk Management Support
 Phenol Safety Training ENVIRONMENTAL Health & Safety and Risk Management Support Overview Properties of Phenol Routes of Exposure and Health Effects Protecting Yourself Handling and Storage Spills and
Phenol Safety Training ENVIRONMENTAL Health & Safety and Risk Management Support Overview Properties of Phenol Routes of Exposure and Health Effects Protecting Yourself Handling and Storage Spills and
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College
 Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860. Effective Date: August 31, 2006
 COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
M A T E R I A L S A F E T Y D A T A
 NFPA 0 1 0 HMIS HEALTH 1 FLAMMABILITY 0 REACTIVITY 0 PERSONAL PROTECTION J 1. Product And Company Identification Supplier Manufacturer Building 20 Building 20 505 Blue Ball Road 505 Blue Ball Road Elkton,
NFPA 0 1 0 HMIS HEALTH 1 FLAMMABILITY 0 REACTIVITY 0 PERSONAL PROTECTION J 1. Product And Company Identification Supplier Manufacturer Building 20 Building 20 505 Blue Ball Road 505 Blue Ball Road Elkton,
Bloodborne Pathogens
 Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
GECDSB WHMIS Training 2017
 GECDSB WHMIS Training 2017 1.1 GECDSB Welcome to Greater Essex County District School Board WHMIS training session. WHMIS is governed by Ontario Regulations 860 and 168, both made under the Occupational
GECDSB WHMIS Training 2017 1.1 GECDSB Welcome to Greater Essex County District School Board WHMIS training session. WHMIS is governed by Ontario Regulations 860 and 168, both made under the Occupational
2006 COURSE TITLE: HAZARDOUS MATERIALS SAFETY
 COURSE INTRODUCTION Hazardous substances are routinely used at work. However, that does not mean they are without risk. Some substances harm you right away. Others may cause health problems that show up
COURSE INTRODUCTION Hazardous substances are routinely used at work. However, that does not mean they are without risk. Some substances harm you right away. Others may cause health problems that show up
J. B. Jewitt Co., Inc. Homestead Finishing Products 1935 West 96 th Street Unit Q Cleveland OH Telephone: (216)
 J. B. Jewitt Co., Inc. Homestead Finishing Products 1935 West 96 th Street Unit Q Cleveland OH 44102-2673 Telephone: (216) 631-5309 1 Emergency Assistance For emergency assistance involving this product
J. B. Jewitt Co., Inc. Homestead Finishing Products 1935 West 96 th Street Unit Q Cleveland OH 44102-2673 Telephone: (216) 631-5309 1 Emergency Assistance For emergency assistance involving this product
Sterilization A Training Module
 Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
Chemistry Lab Safety Handout PSI Chemistry
 Chemistry Lab Safety Handout PSI Chemistry Name Chemistry is exciting! Each day in the laboratory you are given the opportunity to confront the unknown and to understand it. You can wonder how things work,
Chemistry Lab Safety Handout PSI Chemistry Name Chemistry is exciting! Each day in the laboratory you are given the opportunity to confront the unknown and to understand it. You can wonder how things work,
MATERIAL SAFETY DATA SHEET
 Page: 1 of 6 Ref. No: 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: GENERAL USE: Water clarifier MANUFACTURER Asepsis, Inc. P.O. Box 537 Avondale Estates, GA 30002 Customer SERVICE: (800) 959-7946
Page: 1 of 6 Ref. No: 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: GENERAL USE: Water clarifier MANUFACTURER Asepsis, Inc. P.O. Box 537 Avondale Estates, GA 30002 Customer SERVICE: (800) 959-7946
MATERIAL SAFETY DATA SHEET
 Effective Date: 9/7/27 Revision Number: SECTION 1. MATERIAL AND MANUFACTURER IDENTIFICATION Manufacturer: Prime Enterprises Inc. Address: 16363 N.W. 49 th Avenue City: Miami State: FL Zip Code: 3314 Phone:
Effective Date: 9/7/27 Revision Number: SECTION 1. MATERIAL AND MANUFACTURER IDENTIFICATION Manufacturer: Prime Enterprises Inc. Address: 16363 N.W. 49 th Avenue City: Miami State: FL Zip Code: 3314 Phone:
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET SECTION 1: PRODUCT and COMPANY IDENTIFICATION Arch Chemicals, Inc. 501 Merritt 7 PO Box 5204 Issue Date: 12/16/98 Norwalk, CT 06856-5204 MSDS No.: 150000020273 For MSDS Questions:
MATERIAL SAFETY DATA SHEET SECTION 1: PRODUCT and COMPANY IDENTIFICATION Arch Chemicals, Inc. 501 Merritt 7 PO Box 5204 Issue Date: 12/16/98 Norwalk, CT 06856-5204 MSDS No.: 150000020273 For MSDS Questions:
METHODS OF IMPLEMENTATION AND CONTROL
 Universal Precautions: METHODS OF IMPLEMENTATION AND CONTROL All employees will utilize universal precautions (MIOSHA Rule 325.70005) Rule 5. Universal precautions shall be observed to prevent contact
Universal Precautions: METHODS OF IMPLEMENTATION AND CONTROL All employees will utilize universal precautions (MIOSHA Rule 325.70005) Rule 5. Universal precautions shall be observed to prevent contact
REQUEST FOR QUOTE. Community Initiatives Bureau. Biohazardous Cleaning Service
 REQUEST FOR QUOTE Community Initiatives Bureau Biohazardous Cleaning Service December 7, 2017 OVERVIEW The Boston Public Health Commission (BPHC) protects the health protects the health of Bostonians and
REQUEST FOR QUOTE Community Initiatives Bureau Biohazardous Cleaning Service December 7, 2017 OVERVIEW The Boston Public Health Commission (BPHC) protects the health protects the health of Bostonians and
BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN
 BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection Prevention
BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection Prevention
COMPANY IDENTITY: Jon Don Inc. SDS DATE: 05/15/2015 PRODUCT IDENTITY: #5 Wood Cleaner SAFETY DATA SHEET 1. IDENTIFICATION 2. HAZARD IDENTIFICATION
 SAFETY DATA SHEET 1. IDENTIFICATION Product Name: Recommended Use: Supplier: #5 WOOD CLEANER Solvent emulsion cleaner Jon Don Inc. 400 Medinah Road Roselle, IL 60172 Phone: 630-893-4747 Emergency Phone:
SAFETY DATA SHEET 1. IDENTIFICATION Product Name: Recommended Use: Supplier: #5 WOOD CLEANER Solvent emulsion cleaner Jon Don Inc. 400 Medinah Road Roselle, IL 60172 Phone: 630-893-4747 Emergency Phone:
PRESENTS WHMIS AND THE SAFE HANDLING OF HAZARDOUS MATERIALS
 PRESENTS WHMIS AND THE SAFE HANDLING OF HAZARDOUS MATERIALS CESafety 1 WHMIS CESafety 2 What is WHMIS? WHMIS is a Canada-wide system designed to give employers and workers information about hazardous materials
PRESENTS WHMIS AND THE SAFE HANDLING OF HAZARDOUS MATERIALS CESafety 1 WHMIS CESafety 2 What is WHMIS? WHMIS is a Canada-wide system designed to give employers and workers information about hazardous materials
Independently. Read the Lab Safety handout given to you with your groups. Take turns.
 Lab Safety Independently Read the Lab Safety handout given to you with your groups. Take turns. 1. Cut out the lab safety rules and glue it on page one (left side) of your notebook. 2. Add the title Lab
Lab Safety Independently Read the Lab Safety handout given to you with your groups. Take turns. 1. Cut out the lab safety rules and glue it on page one (left side) of your notebook. 2. Add the title Lab
Hazard Communication Program
 ILIULIUK FAMILY AND HEALTH SERVICES Hazard Communication Program Reviewed April 2015 TABLE OF CONTENTS SECTION ONE: Staff and General Information Overview 1 I. Introduction II. Purposes III. Responsibility
ILIULIUK FAMILY AND HEALTH SERVICES Hazard Communication Program Reviewed April 2015 TABLE OF CONTENTS SECTION ONE: Staff and General Information Overview 1 I. Introduction II. Purposes III. Responsibility
Date Printed: 12/27/2007 Date Updated: 02/03/2006 Version 2.9
 SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 12/27/2007 Date Updated: 02/03/2006 Version 2.9 Section 1 - Product and Company Information Product Name Product Number Brand BOUIN'S SOLUTION HT10132
SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 12/27/2007 Date Updated: 02/03/2006 Version 2.9 Section 1 - Product and Company Information Product Name Product Number Brand BOUIN'S SOLUTION HT10132
MATERIAL SAFETY DATA SHEET
 ASPHALT ORGANIC BASE SHEET 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION 400 Old Reading Pike, Ste 304 Pottstown, PA 19464 Tel: 610-579-9075 Fax: 610-323-0115 www.durapax.com PRODUCT NAME: 43 Lb. Organic
ASPHALT ORGANIC BASE SHEET 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION 400 Old Reading Pike, Ste 304 Pottstown, PA 19464 Tel: 610-579-9075 Fax: 610-323-0115 www.durapax.com PRODUCT NAME: 43 Lb. Organic
Working at Biosafety Level 2 (BSL-2)
 Originator: 1.0 Purpose Department of Environmental Health and Safety The purpose of this document is to enhance safety at U of L by ensuring that everyone with potential exposure to infectious agents
Originator: 1.0 Purpose Department of Environmental Health and Safety The purpose of this document is to enhance safety at U of L by ensuring that everyone with potential exposure to infectious agents
SAFETY DATA SHEET. SSS Bella Pearlescent Lotion Hand Soap
 SAFETY DATA SHEET SSS Bella Pearlescent Lotion Hand Soap Date Prepared : 05/14/2015 SDS No : Triple S_Bella 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: SSS Bella Pearlescent Lotion Hand Soap GENERAL
SAFETY DATA SHEET SSS Bella Pearlescent Lotion Hand Soap Date Prepared : 05/14/2015 SDS No : Triple S_Bella 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: SSS Bella Pearlescent Lotion Hand Soap GENERAL
SAFETY DATA SHEET. Majestic Fresh & Clean Triple Action. MANUFACTURER 24 HR. EMERGENCY TELEPHONE NUMBERS MISCO Products Corporation
 SAFETY DATA SHEET Majestic Fresh & Clean Triple Action Date Issued : 11/19/2014 SDS No : I75 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: Majestic Fresh & Clean Triple Action GENERAL USE: Remove
SAFETY DATA SHEET Majestic Fresh & Clean Triple Action Date Issued : 11/19/2014 SDS No : I75 1. PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: Majestic Fresh & Clean Triple Action GENERAL USE: Remove
Bloodborne Pathogens: Exposure In The Workplace Employee Handbook
 Bloodborne Pathogens: Exposure In The Workplace Employee Handbook Introduction There s a danger in the workplace that s not even visible to the naked eye, yet it could change your life forever if you re
Bloodborne Pathogens: Exposure In The Workplace Employee Handbook Introduction There s a danger in the workplace that s not even visible to the naked eye, yet it could change your life forever if you re
OSHA - Hazard Communication
 OSHA - Hazard Communication 1 Hazard Communication Employee Right to Know 2 Components of OSHA s Hazard Communication Standard To ensure that employers and employees know of the hazards associated with
OSHA - Hazard Communication 1 Hazard Communication Employee Right to Know 2 Components of OSHA s Hazard Communication Standard To ensure that employers and employees know of the hazards associated with
Acid Or Alkali? Testing With Cabbage
 Acid Or Alkali? Testing With Cabbage Topic Using vegetables as an acid/base indicator Introduction Forensic scientists need to discover if someone has tampered with liquids (e.g., cosmetics, cleaning products,
Acid Or Alkali? Testing With Cabbage Topic Using vegetables as an acid/base indicator Introduction Forensic scientists need to discover if someone has tampered with liquids (e.g., cosmetics, cleaning products,
"RIGHT-TO-KNOW FOR BUILDING AND CONSTRUCTION COMPANIES"
 MAJOR PROGRAM POINTS "RIGHT-TO-KNOW FOR BUILDING AND CONSTRUCTION COMPANIES" Part of the "CONSTRUCTION SAFETY KIT" Series Quality Safety and Health Products, for Today...and Tomorrow OUTLINE OF MAJOR PROGRAM
MAJOR PROGRAM POINTS "RIGHT-TO-KNOW FOR BUILDING AND CONSTRUCTION COMPANIES" Part of the "CONSTRUCTION SAFETY KIT" Series Quality Safety and Health Products, for Today...and Tomorrow OUTLINE OF MAJOR PROGRAM
M A T E R I A L S A F E T Y D A T A
 NFPA 0 1 0 HMIS HEALTH 1 FLAMMABILITY 0 REACTIVITY 0 PERSONAL PROTECTION E 1. Product And Company Identification Supplier Manufacturer Building 20 Building 20 505 Blue Ball Road 505 Blue Ball Road Elkton,
NFPA 0 1 0 HMIS HEALTH 1 FLAMMABILITY 0 REACTIVITY 0 PERSONAL PROTECTION E 1. Product And Company Identification Supplier Manufacturer Building 20 Building 20 505 Blue Ball Road 505 Blue Ball Road Elkton,
Warning.: OVEREXPOSURE TO THIS PRODUCT MAY CAUSE IRRITATION TO THE EYES AND SKIN.
 MATERIAL SAFETY DATA SHEET ORGANIC DYESTUFFS CORPORATION P.O. Box 14258 East Providence, Rhode Island 02914-0258 Telephone No. (401) 434-3300 Trade Name...: ORCOSOLVE BLUE AP Chemical Name: ANTHRAQUINONE
MATERIAL SAFETY DATA SHEET ORGANIC DYESTUFFS CORPORATION P.O. Box 14258 East Providence, Rhode Island 02914-0258 Telephone No. (401) 434-3300 Trade Name...: ORCOSOLVE BLUE AP Chemical Name: ANTHRAQUINONE
