Student Performance Guide. Student Performance Guide. Student Performance Guide
|
|
|
- Bruno Gilmore
- 5 years ago
- Views:
Transcription
1 LESSON 8-2 Collecting and Processing Specimens for Parasite Examination Student Performance Guide LESSON 8-3 Microscopic Methods for Student Performance Guide LESSON 8-4 Preparing and Staining Smears for Blood Parasites Student Performance Guide
2 Student Performance Guide LESSON 8-2 Collecting and Processing Specimens for Parasite Examination Name Date INSTRUCTIONS 1. Practice the procedures for collecting and processing specimens for parasite examination, following the step-by-step procedure. 2. Demonstrate your understanding of this lesson by: a. Completing a written examination successfully, and b. Performing the procedures for collecting and processing specimens for parasite examination satisfactorily for the instructor. All steps must be completed as listed on the instructor s Performance Check Sheet. MATERIALS AND EQUIPMENT gloves hand disinfectant 10% chlorine bleach solution wooden tongue depressors wooden applicator sticks microscope slides clear cellophane tape xylene or toluene microscope biohazard disposal container sharps disposal container leakproof vials for transporting specimens 10% formalin PVA (or other appropriate) fixative fecal specimens (students may bring pet specimens for practice) fecal specimen containers atlas of parasitic morphology containing illustrations PROCEDURE Record in the comment section any problems encountered while practicing the procedure (or have a fellow student or the instructor evaluate your performance). S = Satisfactory U = Unsatisfactory You must: S U Comments 1. Instruct a patient in the proper procedure for collecting a fecal specimen (steps 1a c): a. Give patient a fecal specimen container with lid and label b. Explain the fecal collection procedure to the patient, emphasizing the following precautions: (1) Specimen must not be contaminated with urine or water (2) Outer surface of specimen container must not be contaminated (3) Container must be labeled with the patient s name, date, and time of specimen collection
3 You must: S U Comments (4) Unpreserved specimens must be transported to a laboratory immediately following collection (within two hours) (5) Specimens should not be collected until at least a week after ingesting interfering substances such as oily laxatives, barium or radiopaque contrast media, or antidiarrheal medications c. If immediate transport to a laboratory is not possible, instruct the patient in the proper way to preserve the specimen, following the instructions in step Prepare a fecal specimen for transport (steps 2a i) a. Wash your hands and put on gloves b. Obtain a fecal specimen, fixative vials, and transport containers. Check label to be sure required patient information is present (name, date, time of collection) c. Open container and observe consistency of specimen. Record as watery, loose, soft, or formed d. Obtain a vial of 10% formalin and one of PVA fixative e. Use disposable applicators to obtain a portion of specimen approximately one-third the volume of the fixative f. Place specimen in vial and mix thoroughly using applicator. Discard applicator(s) into biohazard container. Label vial with patient information g. Repeat procedure (steps 2e f) with the second vial of fixative, using clean applicators h. Disinfect work area with surface disinfectant i. Remove and discard gloves in biohazard container and wash your hands with hand disinfectant 3. Prepare a cellophane tape perianal swab (steps 3a e) a. Obtain tape, clean microscope slide, and paper tab b. Attach a four-to-five-inch section of tape to one end of the microscope slide s back c. Fold the tape around the end of the slide and smooth down over the slide top, so the tape adheres to the slide, leaving a small portion free at the end d. Attach a small paper tab to the free end of the tape to use as a label and lifting tab e. Store the prepared slide in a cool, dust-free location 4. Demonstrate the use of the cellophane tape swab (steps 4a k) a. Wash your hands and put on gloves b. Obtain a clean wooden tongue depressor c. Obtain previously prepared cellophane tape slide (step 3) and label the tab d. Place the cellophane tape swab near one end of the depressor, tape side up e. Lift the cellophane tape and form a loop around the end of the tongue depressor, sticky side of tape to the outside f. Explain how to obtain the pinworm specimen by touching sticky surface of the tape to perianal region
4 You must: S U Comments g. Remove the tape from the tongue depressor and smooth back into place on the microscope slide, being careful not to touch the sticky surface h. Discard the tongue depressor in biohazard container (if actually used for specimen collection) i. Place the slide on a microscope stage and demonstrate how to observe for eggs using the low-power (10X) objective j. Clear glue from tape by placing a drop of xylene or toluene under the tape if desired (follow proper precautions in handling these chemicals) k. Remove the slide from the microscope stage and discard in biohazard sharps container 5. Discard specimen in appropriate biohazard container, as directed by instructor 6. Discard used materials appropriately 7. Return supplies to storage 8. Disinfect work area with surface disinfectant 9. Remove and discard gloves in biohazard container and wash your hands with hand disinfectant Evaluator Comments: Evaluator Date
5 Student Performance Guide LESSON 8-3 Microscopic Methods for Detecting Intestinal Parasites Name Date INSTRUCTIONS 1. Practice preparing fecal specimens for microscopic examination for intestinal parasites, following the step-by-step procedure. 2. Demonstrate your understanding of this lesson by: a. Completing a written examination successfully, and b. Preparing fecal specimens for microscopic examination for intestinal parasites satisfactorily for the instructor. All steps must be completed as listed on the instructor s Performance Check Sheet. MATERIALS AND EQUIPMENT gloves hand disinfectant surface disinfectant (10% chlorine bleach solution) absorbent lab paper puncture-proof container for sharps biohazard containers wooden applicator sticks fecal specimens preserved in 10% formalin and in PVA (specimens from pets may be used) fume hood Materials for Wet Mounts: coverglasses, 22 mm 2 glass microscope slides, 2 x 3 inch preferable saline (0.85% NaCl) iodine solution for fecal wet mounts (Lugol s, Dobell and O Connors, or D Antoni s) dropping pipets D Antoni s Iodine: Add 1.0 g potassium iodide (KI) and 1.5 g iodine crystals to 100 ml distilled water in a dark bottle. Shake well and filter daily before use. Materials for Preparing Fecal Smear for Staining: 35 C incubator or laboratory oven microscope slides, 1 x 3 inch wooden applicator sticks Materials for Formalin-Ethyl Acetate Sedimentation (or a commercial stool concentration kit): conical centrifuge tubes, 15 ml capacity (preferably disposable) 10% formalin ethyl acetate cotton swabs gauze or cheesecloth plastic funnel clinical centrifuge capable of spinning 15 ml conical tubes at 500 xg wooden applicator sticks Optional: Preserved fecal specimens containing parasites, atlases, diagrams, stained microscope slides, photographs, or videos of intestinal parasites
6 PROCEDURE Record in the comment section any problems encountered while practicing the procedure (or have a fellow student or the instructor evaluate your performance). S = Satisfactory U = Unsatisfactory You must: S U Comments 1. Assemble equipment and materials 2. Wash your hands and put on gloves 3. Prepare a work area by placing absorbent lab paper on counter or in fume hood 4. Demonstrate the procedure for preparing a fecal smear for staining, following steps 4a d a. Obtain PVA-fixed fecal specimen and two 1 x 3 inch microscope slides b. Use applicator stick to mix specimen and remove a portion c. Spread specimen evenly on slide by rolling stick across slide or smearing in a zig-zag fashion (see Figure 8-11). Smear should cover one-third to one-half the length of the slide and should extend from the slide s top to bottom edges. Prepare a second slide from the same specimen d. Label slides and place them in 35 C incubator to dry for at least four hours, preferably overnight 5. Demonstrate the procedure for preparing saline and iodine wet mounts from a formalin-fixed fecal specimen, following steps 5a i a. Obtain a formalin-fixed specimen b. Place a 2 x 3 inch glass slide on work area c. Place one drop of saline on left half of slide and one drop of iodine solution on right half of slide d. Use applicator stick to mix specimen e. Remove a small portion of specimen with applicator stick and mix in with saline drop. Place a coverglass over the drop f. Remove another small portion of fecal specimen and mix in with iodine drop. Place a coverglass over the drop. Discard applicator in biohazard container g. Place slide over newsprint and check thickness of wet mounts (letters should be readable through the specimen) h. Optional: Place slide on microscope stage and scan specimens with the low-power and high-power objectives. Use visual aids such as atlases, diagrams, or photographs to help recognize parasitic forms i. Discard slide in biohazard sharps container
7 You must: S U Comments 6. Demonstrate the procedure for formalin-ethyl acetate sedimentation, following steps 6a-k, or skip to step 7 a. Obtain a formalin-fixed fecal specimen b. Mix the specimen and strain 5mL through wet gauze into a conical centrifuge tube c. Add saline or formalin to a volume of 15 ml and centrifuge the tube at 500 xg for 10 min d. Decant supernatant into a container of disinfectant, leaving approximately 1mL in tube e. Add 9 ml of 10% formalin to tube and mix using applicator stick f. Add 4 ml ethyl acetate, cap tube securely, and shake it vigorously by inversion for one-half minute g. Centrifuge tube for ten minutes at 500 xg h. Use a wooden applicator to ring the debris plug i. Pour off the top layers of ethyl acetate, debris, and formalin (see Figure 8-10), leaving ml of sediment in tube j. Use a cotton swab to remove debris from the inside of tube. Add a few drops of 10% formalin to resuspend sediment k. Mix sediment with applicator stick and prepare saline and iodine wet mounts from portions of sediment, as in step 5b i 7. Discard all contaminated materials in appropriate biohazard containers 8. Discard or store preserved specimens, as directed by instructor. 9. Disinfect work area and reusable supplies with 10% chlorine bleach solution 10. Remove and discard gloves in biohazard container and wash your hands with hand disinfectant Evaluator Comments: Evaluator Date
8 Student Performance Guide LESSON 8-4 Preparing and Staining Smears for Blood Parasites Name Date INSTRUCTIONS 1. Practice preparing and staining smears for blood parasites, following the step-by-step procedure. 2. Demonstrate your understanding of this lesson by: a. Completing a written examination successfully, and b. Performing the procedure for preparing and staining smears for blood parasites satisfactorily for the instructor. All steps must be completed as listed on the instructor s Performance Check Sheet. MATERIALS AND EQUIPMENT gloves hand disinfectant surface disinfectant biohazard container puncture-proof container for sharp objects acrylic safety shield or face protection Materials for capillary puncture clean glass slides absolute methanol stock Giemsa stain staining jars (Coplin jars) phosphate-buffered water, ph 7.2 Recipe for Giemsa buffer (phosphate-buffered water): 39 ml M NaH 2 PO 4 61 ml M Na 2 HPO ml distilled water check ph. Should be microscope immersion oil lens paper slide box Optional visual aids: commercially prepared stained slides of Plasmodium and Babesia; commercially prepared stained slides of filarial worms, such as dog heartworm (Dirofilaria sp.); charts, Kodachrome slides, and figures showing morphology of blood parasites Note: Consult manufacturer s instructions accompanying Giemsa stain for recommended optimal dilution and staining time. PROCEDURE Record in the comment section any problems encountered while practicing the procedure (or have a fellow student or the instructor evaluate the performance). S = Satisfactory U = Unsatisfactory You must: S U Comments 1. Wash your hands and put on gloves 2. Assemble equipment and materials for capillary puncture 3. Perform a capillary puncture (as described in Lesson 2-2) 4. Wipe away the first drop of blood
9 You must: S U Comments 5. Apply a small drop of blood to a clean glass slide and use a clean spreader slide to form a thin blood film (as in Lesson 2-8). Make duplicate smears and set slides aside to air-dry 6. Immerse dried thin smears in absolute methanol for thirty to sixty seconds and air-dry. 7. Make a thick smear by holding a clean slide under the patient s finger and allowing one or two large drops of blood to fall on the center of the slide 8. Spread the blood evenly into a dime-sized circle to form the thick smear, using slight pressure against the fingertip (or use the corner of a clean glass slide to spread the blood). Make duplicate smears 9. Check the thickness of the thick smears by laying the slides on printed material. The print should be readable through the blood film 10. Place the slides on a flat surface in a dust-free place and allow them to air-dry at room temperature for several hours or overnight. DO NOT FIX. 11. Have patient apply pressure to puncture site with sterile gauze when satisfactory smears have been obtained. (At this point, if staining is to be done another day, work area may be cleaned, gloves removed, and hands washed, steps Reglove before handling smears for staining procedure) 12. Stain smears by immersing slides in a freshly prepared 1:50 dilution of Giemsa stain for fifty minutes. (Be sure thin smear has been fixed and thick smear has dried for several hours) 13. Rinse stained smears in buffered water: rinse thin smear one to two minutes; rinse thick smear three to five minutes 14. Place rinsed slides in slide rack and allow to air-dry 15. Place thin smear on microscope stage and observe cells using oil-immersion objective. Observe quality of stain: RBCs should be pinkish, WBC nuclei should be blue-purple. Examine RBCs for stained intracellular parasitic inclusions. (Refer to charts, figures, or commercially prepared slides, if available) 16. Remove thin smear from microscope stage and place thick smear in position 17. Examine thick smear using oil-immersion objective. WBCs and platelets should be visible, but RBCs should have been destroyed in the staining process. If parasites were present in the blood specimen, they will be stained. (Refer to charts, figures, or commercially prepared slides, if available) 18. Remove slide from microscope stage 19. Clean oil from microscope objective with lens paper
10 You must: S U Comments 20. Place slides in covered slide box 21. Discard capillary puncture materials in appropriate biohazard containers 22. Clean equipment and return to proper storage 23. Clean work area with surface disinfectant 24. Remove and discard gloves in biohazard container 25. Wash your hands with hand disinfectant Evaluator Comments: Evaluator Date
Student Performance Guide. Student Performance Guide. Student Performance Guide. Student Performance Guide. LESSON 3-3 Bleeding Time
 LESSON 3-3 Bleeding Time Student Performance Guide LESSON 3-4 Prothrombin Time Student Performance Guide LESSON 3-5 Activated Partial Thromboplastin Time Student Performance Guide LESSON 3-6 Rapid Tests
LESSON 3-3 Bleeding Time Student Performance Guide LESSON 3-4 Prothrombin Time Student Performance Guide LESSON 3-5 Activated Partial Thromboplastin Time Student Performance Guide LESSON 3-6 Rapid Tests
PREPARATION OF BLOOD FILMS FOR MALARIA DETECTION
 PREPARATION OF BLOOD FILMS FOR MALARIA DETECTION Materials for Preparation of Malaria Smears: Clean and wrapped slides Sterile lancets 70% ethanol and water Absorbent cotton wool Surgical gloves Lint-free
PREPARATION OF BLOOD FILMS FOR MALARIA DETECTION Materials for Preparation of Malaria Smears: Clean and wrapped slides Sterile lancets 70% ethanol and water Absorbent cotton wool Surgical gloves Lint-free
Staining of the clinical material or the bacteria from colonies on laboratory media provide a direct visualization of the morphology of the organisms
 COMMON STAINING PROCEDURES Staining of the clinical material or the bacteria from colonies on laboratory media provide a direct visualization of the morphology of the organisms as well as their reactions
COMMON STAINING PROCEDURES Staining of the clinical material or the bacteria from colonies on laboratory media provide a direct visualization of the morphology of the organisms as well as their reactions
Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System. Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe
 Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Biohazardous Waste. 1. Solid Biohazardous Waste (non-sharps) Storage
 Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
for Stool Examination Issued by: LABORATORY MANAGER Original Date: March 13, 2000 Approved by: Laboratory Director Hematoxylin Stain
 Section: Page 28 Policy # MI\PAR\05\06\v01 Page 1 of 5 Subject Title: Laboratory Procedures for Stool Examination Issued by: LABORATORY MANAGER Original Date: March 13, 2000 Approved by: Laboratory Director
Section: Page 28 Policy # MI\PAR\05\06\v01 Page 1 of 5 Subject Title: Laboratory Procedures for Stool Examination Issued by: LABORATORY MANAGER Original Date: March 13, 2000 Approved by: Laboratory Director
Laboratory technique and preparations
 Laboratory technique and preparations Bio 381 written by : Hind Alzaylaee Alshareef_ Maryam Alzayn Alshareef 9/17/2012 graduated cylinder Funnel Flask beaker Dropping bottle Watch glass Petri dish Reagent
Laboratory technique and preparations Bio 381 written by : Hind Alzaylaee Alshareef_ Maryam Alzayn Alshareef 9/17/2012 graduated cylinder Funnel Flask beaker Dropping bottle Watch glass Petri dish Reagent
COLLECTION INSTRUCTIONS - SAMPLES (VARIOUS TYPES)
 COLLECTION INSTRUCTIONS - SAMPLES (VARIOUS TYPES) You need to complete the necessary forms for each sample collected. To avoid any possible contamination, please ensure that gloves are worn during the
COLLECTION INSTRUCTIONS - SAMPLES (VARIOUS TYPES) You need to complete the necessary forms for each sample collected. To avoid any possible contamination, please ensure that gloves are worn during the
#74 - CHANGING A MOIST TO DRY DRESSING (TEST)
 #74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
#74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
LAB 5 Blood Collection
 LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
Surgical Gown. Tongue Depressor. A disposable gown worn by medical staff during surgery. A thin, flat, wooden stick rounded at both ends
 Tongue Depressor A thin, flat, wooden stick rounded at both ends Accidentally dropped on the floor by the doctor 16 Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon
Tongue Depressor A thin, flat, wooden stick rounded at both ends Accidentally dropped on the floor by the doctor 16 Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon
Bacterial smear and Staining
 Practical Microbiology 18-22/11/2018 University of Sulaimani college of Pharmacy Year2 Lab. 4: Bacterial smear and Staining Before staining and observing a microbe under a microscope, a smear must be prepared.
Practical Microbiology 18-22/11/2018 University of Sulaimani college of Pharmacy Year2 Lab. 4: Bacterial smear and Staining Before staining and observing a microbe under a microscope, a smear must be prepared.
Standard Laboratory Practice for Consumer Applied Pet Stain and Odor Removal Chemical Evaluation on Pile Yarn Floor Coverings
 P.O. Box 2048 Dalton Georgia 30722-2048 706.278.3176 carpet-rug.org CRI Test Method - 116 Technical Bulletin Standard Laboratory Practice for Consumer Applied Pet Stain and Odor Removal Chemical Evaluation
P.O. Box 2048 Dalton Georgia 30722-2048 706.278.3176 carpet-rug.org CRI Test Method - 116 Technical Bulletin Standard Laboratory Practice for Consumer Applied Pet Stain and Odor Removal Chemical Evaluation
LAB 3 CHARACTERIZING YOUR UNKNOWN BACTERIA AND USING MORE COMPLEX STAINS. Part I: Isolating Your Unknown Bacteria and Describing Colony Morphology
 LAB 3 CHARACTERIZING YOUR UNKNOWN BACTERIA AND USING MORE COMPLEX STAINS Objectives In this lab you will learn how to: - describe bacteria on the basis of colony and cell morphology - isolate bacterial
LAB 3 CHARACTERIZING YOUR UNKNOWN BACTERIA AND USING MORE COMPLEX STAINS Objectives In this lab you will learn how to: - describe bacteria on the basis of colony and cell morphology - isolate bacterial
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
Stains and Solutions Used in Hematology and Cytology
 Stains and Solutions Used in Hematology and Cytology A APPENDIX Acid-Fast Stain Commercially prepared acid-fast stains are available 1. Ziehl Neelsen carbolfuchsin: Dissolve 3.0 g basic fuchsin in 100
Stains and Solutions Used in Hematology and Cytology A APPENDIX Acid-Fast Stain Commercially prepared acid-fast stains are available 1. Ziehl Neelsen carbolfuchsin: Dissolve 3.0 g basic fuchsin in 100
PROTOCOL FOR SCIENCE EQUIPMENT USAGE WITH EMPHASIS ON BS240/242: MICROORGANISMS AND THEIR HUMAN HOSTS Updated July 2015
 PROTOCOL FOR SCIENCE EQUIPMENT USAGE WITH EMPHASIS ON BS240/242: MICROORGANISMS AND THEIR HUMAN HOSTS Updated July 2015 Personnel Maija Bluma (573-651-2069; mbluma@semo.edu) is the Microbiology Preparation
PROTOCOL FOR SCIENCE EQUIPMENT USAGE WITH EMPHASIS ON BS240/242: MICROORGANISMS AND THEIR HUMAN HOSTS Updated July 2015 Personnel Maija Bluma (573-651-2069; mbluma@semo.edu) is the Microbiology Preparation
Updated by S. McNew, March Deborah Jung Microbiology Preparation Technician
 Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers
 Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Introductory Chemistry
 Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
BSL-2 Emergency Plan
 BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
Basic Microbiology and Immunology Practical Course
 Basic Microbiology and Immunology Practical Course 2 Lab # 2: Colouring the microorganisms Rules that must be followed to maintain an aseptic zone 3 For most bacterial cultures, you will use a sterile
Basic Microbiology and Immunology Practical Course 2 Lab # 2: Colouring the microorganisms Rules that must be followed to maintain an aseptic zone 3 For most bacterial cultures, you will use a sterile
Brazosport College Life Science Laboratory Safety Rules and Regulations
 Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Emergency Procedures Specific Biological Spill Clean-Up Guidelines
 Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
: In order to study tissues with a microscope they must be preserved (fixed)- fixation Following fixation, blocks of tissue must be cut into thin
 : In order to study tissues with a microscope they must be preserved (fixed)- fixation Following fixation, blocks of tissue must be cut into thin sections.-microtomy Other techniques involve dehydration
: In order to study tissues with a microscope they must be preserved (fixed)- fixation Following fixation, blocks of tissue must be cut into thin sections.-microtomy Other techniques involve dehydration
Mt. San Antonio College: Spring 2018 MICR 22 Lab Orientation. Welcome to the Microbiology 22 Laboratory!
 Mt. San Antonio College: Spring 2018 MICR 22 Lab Orientation Welcome to the Microbiology 22 Laboratory! Laboratory Objectives: To teach concepts of microbiological techniques using critically selected
Mt. San Antonio College: Spring 2018 MICR 22 Lab Orientation Welcome to the Microbiology 22 Laboratory! Laboratory Objectives: To teach concepts of microbiological techniques using critically selected
Tape Strip. Year Group: BVSc4 + Document number: CSL_P03
 Year Group: BVSc4 + Document number: CSL_P03 Equipment list: Equipment for this station: Sellotape Glass microscope slides Wax marker crayon Gloves Diff-Quik booklet ( CSL_L06 Diff-Quik Staining ) Diff-Quik
Year Group: BVSc4 + Document number: CSL_P03 Equipment list: Equipment for this station: Sellotape Glass microscope slides Wax marker crayon Gloves Diff-Quik booklet ( CSL_L06 Diff-Quik Staining ) Diff-Quik
MANICURE. Before preparing client for manicure, look for any nail disorders or infections like:
 Practical 1 MANICURE Objectives At the end of this session, you will be able to: clean and shape the nails using manicure tools clean and massage the hands apply nail polish Tools, Equipments and Materials
Practical 1 MANICURE Objectives At the end of this session, you will be able to: clean and shape the nails using manicure tools clean and massage the hands apply nail polish Tools, Equipments and Materials
DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling Kuala Lumpur, Malaysia. Tel : Fax :
 DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling 57000 Kuala Lumpur, Malaysia. Tel : 03-9057 1316 Fax : 03-90578925 sales@dzcmarketing.com WhatsApp +6018-666 8794 MEDICAL DISPOSABLE LIST
DZC Marketing 15, Jalan Radin Anum 2, Bandar Baru Sri Petaling 57000 Kuala Lumpur, Malaysia. Tel : 03-9057 1316 Fax : 03-90578925 sales@dzcmarketing.com WhatsApp +6018-666 8794 MEDICAL DISPOSABLE LIST
Comparative Proteomics Kit II: Western Blot Module Quick Guide
 Comparative Proteomics Kit II: Western Blot Module Quick Guide Lesson 1 1 Label one 1.5 ml fliptop micro tube for each of five fish samples. Also label one screw-cap micro tube for each fish sample. 2.
Comparative Proteomics Kit II: Western Blot Module Quick Guide Lesson 1 1 Label one 1.5 ml fliptop micro tube for each of five fish samples. Also label one screw-cap micro tube for each fish sample. 2.
EXERCISE 8C - Lab Procedures
 EXERCISE 8C - Lab Procedures SAFETY WARNING: Acrylamide in the unpolymerized form is a skin irritant and a potential neurotoxin. Fortunately, the acrylamide in your gels is polymerized, so it should not
EXERCISE 8C - Lab Procedures SAFETY WARNING: Acrylamide in the unpolymerized form is a skin irritant and a potential neurotoxin. Fortunately, the acrylamide in your gels is polymerized, so it should not
Roosevelt Biosafety Training. Created 10/2015
 Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
VisPRO 5 Minutes Protein Stain Kit
 Manual VisPRO 5 Minutes Protein Stain Kit VP01-125/VP01-500/VP05-125/VP05-500 V2.0 Store at room temperature For Research Use Only Introduction VisPRO 5 Minutes Protein Stain Kit (1 nanogram grade) provides
Manual VisPRO 5 Minutes Protein Stain Kit VP01-125/VP01-500/VP05-125/VP05-500 V2.0 Store at room temperature For Research Use Only Introduction VisPRO 5 Minutes Protein Stain Kit (1 nanogram grade) provides
Safety Office -- Laboratory Inspection Form
 RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
Procedure 19 Changing A Clean Dressing. Procedure 20 Applying A Bandage. Procedure 21 Applying A Sterile Dressing
 Chapter 5 Wound Care Procedure 19 Changing A Clean Dressing Procedure 20 Applying A Bandage Procedure 21 Applying A Sterile Dressing Procedure 22 Applying A Dressing Around A Drain Procedure 23 Changing
Chapter 5 Wound Care Procedure 19 Changing A Clean Dressing Procedure 20 Applying A Bandage Procedure 21 Applying A Sterile Dressing Procedure 22 Applying A Dressing Around A Drain Procedure 23 Changing
SPECIMEN COLLECTION. A Study Guide
 SPECIMEN COLLECTION A Study Guide Expectations At the UAP Marathon the UAP will be expected to verbalize the process of performing a peripheral blood draw and demonstrating the correct collection of a
SPECIMEN COLLECTION A Study Guide Expectations At the UAP Marathon the UAP will be expected to verbalize the process of performing a peripheral blood draw and demonstrating the correct collection of a
Biomedical Laboratory Science
 Biomedical Laboratory Science!! " " New for 2015-2016 The 70% mastery for each skill has been added to the rating sheet so that competitors and advisors know what score is required for mastery. Rating
Biomedical Laboratory Science!! " " New for 2015-2016 The 70% mastery for each skill has been added to the rating sheet so that competitors and advisors know what score is required for mastery. Rating
Bloodborne Pathogens Exposure Control Plan. December 2003
 Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Lab Six:- Medical Microbiology Prepared by: Luma J. Witwit. Staining
 Staining Even with the microscope, bacteria are difficult to see unless they are treated in a way that increases contrast between the organisms and their background. The most common method to increase
Staining Even with the microscope, bacteria are difficult to see unless they are treated in a way that increases contrast between the organisms and their background. The most common method to increase
LYMPHOCYTE ISOLATION
 Blood tubes to be processed for Lymphocyte Isolation and/or Lymphocyte Transformation should be placed in the rack located on top of the black cabinet and stored at room temperature. A. WARM WASH MEDIUM
Blood tubes to be processed for Lymphocyte Isolation and/or Lymphocyte Transformation should be placed in the rack located on top of the black cabinet and stored at room temperature. A. WARM WASH MEDIUM
Safety Rules for Laboratory
 Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
PET Barrier Test PET- R- 02
 PET Barrier Test PET- R- 02 The following protocol is designed to provide a procedure for identifying and quantifying residual amounts of three barrier materials, EVOH, MXD6 nylon, and epoxy diamine, in
PET Barrier Test PET- R- 02 The following protocol is designed to provide a procedure for identifying and quantifying residual amounts of three barrier materials, EVOH, MXD6 nylon, and epoxy diamine, in
x. ANNUAL REVIEW SIGNATURE SHEET
 x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
Sterilization A Training Module
 Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
STUDENT LABORATORY PACKET
 L5 Elodea-Onion-Cheek-Cell_Size Page 1 of 7 STUDENT LABORATORY PACKET Student s Full Name Lab #5: Elodea, Onion, Cheek Cells-Cell Size Lab Instructor Date Points Microscope # OBJECTIVES: a. to examine
L5 Elodea-Onion-Cheek-Cell_Size Page 1 of 7 STUDENT LABORATORY PACKET Student s Full Name Lab #5: Elodea, Onion, Cheek Cells-Cell Size Lab Instructor Date Points Microscope # OBJECTIVES: a. to examine
ANALYSIS OF FINGERPRINTS, LIPSTICK 2 ND HAIR
 ANALYSIS OF FINGERPRINTS, LIPSTICK 2 ND HAIR LAB FORENSICS.3 From Sourcebook, National Science Foundation, 1997 INTRODUCTION PART A. OBTAINING A FINGERPRINT Black ink stamp pad Tissue paper 4 x 4 cm Card
ANALYSIS OF FINGERPRINTS, LIPSTICK 2 ND HAIR LAB FORENSICS.3 From Sourcebook, National Science Foundation, 1997 INTRODUCTION PART A. OBTAINING A FINGERPRINT Black ink stamp pad Tissue paper 4 x 4 cm Card
Infection Control 101
 Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
KERATIN CONTAMINATION
 KERATIN CONTAMINATION Keratin contamination is almost always observed as a background protein. Wear only nitrile gloves and rinse with HPLC grade water all trays, containers and surfaces that contact the
KERATIN CONTAMINATION Keratin contamination is almost always observed as a background protein. Wear only nitrile gloves and rinse with HPLC grade water all trays, containers and surfaces that contact the
General Lab Safety Rules and Practices SOP-GLSRP-01
 Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Biomedical Laboratory Science
 Biomedical Laboratory Science New for 2018-2019 At ILC, photo ID must be presented prior to competing in each round. Biotechnology: Science for the New Millennium Edition 2 has been released. For information
Biomedical Laboratory Science New for 2018-2019 At ILC, photo ID must be presented prior to competing in each round. Biotechnology: Science for the New Millennium Edition 2 has been released. For information
PharmChek Drugs of Abuse Sweat Patch Training Manual
 PharmChek Drugs of Abuse Sweat Patch Training Manual For the Application, Removal, Specimen collection, and Transport of the PharmChek Drugs of Abuse Patch For Professional Use Only Revised November, 2016
PharmChek Drugs of Abuse Sweat Patch Training Manual For the Application, Removal, Specimen collection, and Transport of the PharmChek Drugs of Abuse Patch For Professional Use Only Revised November, 2016
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN
 FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
Tips On Proper Instrument Cleaning, Handling and Maintenance!
 304 304 Rinsing Tips On Proper Instrument Cleaning, Handling and Maintenance! *Immediately after instrument use thoroughly rinse off all blood, tissue and other fluids. *Using filtered water to rinse and
304 304 Rinsing Tips On Proper Instrument Cleaning, Handling and Maintenance! *Immediately after instrument use thoroughly rinse off all blood, tissue and other fluids. *Using filtered water to rinse and
Optiblot Non-Reducing Electrophoresis Kit
 Optiblot Non-Reducing Electrophoresis Kit Instructions for Use For the use in non-reducing SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table
Optiblot Non-Reducing Electrophoresis Kit Instructions for Use For the use in non-reducing SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table
Regulated Medical Waste. Be sure to sign in!
 Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Optiblot SDS-PAGE Gel
 Optiblot SDS-PAGE Gel Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table of Contents 1. Introduction 3
Optiblot SDS-PAGE Gel Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table of Contents 1. Introduction 3
STANDARDIZED CLINICAL PROTOCOLS. Sterilization Protocols. Aravind Eye Care System 1, Anna Nagar, Madurai , Tamilnadu, India
 STANDARDIZED CLINICAL PROTOCOLS Sterilization Protocols Aravind Eye Care System 1, Anna Nagar, Madurai - 625 020, Tamilnadu, India i.exe Standardized Sterilization Protocol STANDARDIZED STERILIZATION PROCEDURE
STANDARDIZED CLINICAL PROTOCOLS Sterilization Protocols Aravind Eye Care System 1, Anna Nagar, Madurai - 625 020, Tamilnadu, India i.exe Standardized Sterilization Protocol STANDARDIZED STERILIZATION PROCEDURE
EXPERIMENT. Bacterial Morphology and Staining Techniques
 EXPERIMENT Bacterial Morphology and Staining Techniques Hands-On Labs, Inc. Version 42-0240-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before
EXPERIMENT Bacterial Morphology and Staining Techniques Hands-On Labs, Inc. Version 42-0240-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before
UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach
 Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
PROTOCOLS FOR ANATOMY/MICROMORPHOLOGY
 PROTOCOLS FOR ANATOMY/MICROMORPHOLOGY General dissection of spikelets... 2 Hand sections and epidermal scrapes of bamboo leaves... 2 Clearing and staining of intact plant organs... 4 Scanning electron
PROTOCOLS FOR ANATOMY/MICROMORPHOLOGY General dissection of spikelets... 2 Hand sections and epidermal scrapes of bamboo leaves... 2 Clearing and staining of intact plant organs... 4 Scanning electron
PURPOSE: To define the responsibilities for obtaining nipple and breast milk cultures.
 1 PURPOSE: To define the responsibilities for obtaining nipple and breast milk cultures. RESPONSIBILITY STATEMENT: Staff may obtain nipple and breast milk culture with a physician order. SUPPORTIVE DATA:
1 PURPOSE: To define the responsibilities for obtaining nipple and breast milk cultures. RESPONSIBILITY STATEMENT: Staff may obtain nipple and breast milk culture with a physician order. SUPPORTIVE DATA:
Package Leaks. OH&S Biosafety Emergency Response Document. Examine outer packaging. Leaks or evidence of leaks. No evidence of leaks
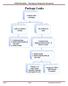 Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Optiblot SDS-PAGE Gel
 Optiblot SDS-PAGE Gel Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table of Contents 1. Introduction 3
Optiblot SDS-PAGE Gel Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. 1 Table of Contents 1. Introduction 3
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # Approved: 10/3/18
 Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
What is infection control?
 Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET
 TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
POP ACRYLIC NAILS. user guide
 POP ACRYLIC NAILS user guide POP ACRYLIC NaILS Pop Acrylic Nails make professional results even quicker and easier to achieve. The traditional method of applying acrylic nails can be difficult to master.
POP ACRYLIC NAILS user guide POP ACRYLIC NaILS Pop Acrylic Nails make professional results even quicker and easier to achieve. The traditional method of applying acrylic nails can be difficult to master.
Franklin Regional School District SCIENCE MIDDLE SCHOOL Bid #16 Vendor Specifications
 SCIENCE MIDDLE SCHOOL Bid #16 Quotation number: (Number you want reflected on the Purchase Order) Company: Address: Telephone: Email: Date: Authorized name (print): Authorized Signature: 1 GLASSWARE/TUBING/CYLINDERS
SCIENCE MIDDLE SCHOOL Bid #16 Quotation number: (Number you want reflected on the Purchase Order) Company: Address: Telephone: Email: Date: Authorized name (print): Authorized Signature: 1 GLASSWARE/TUBING/CYLINDERS
Standard Operating Procedure for Biosafety Cabinet Use
 NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
Disposal of Biological Waste
 Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
PHYSICAL EVIDENCE. Something that shows or proves, or that gives reason for believing; proof or indication (Webster s New World Dictionary;1995) tox
 PHYSICAL EVIDENCE Something that shows or proves, or that gives reason for believing; proof or indication (Webster s New World Dictionary;1995) swabs shirt debris hair Patient tox fibers pattern DNA Skin
PHYSICAL EVIDENCE Something that shows or proves, or that gives reason for believing; proof or indication (Webster s New World Dictionary;1995) swabs shirt debris hair Patient tox fibers pattern DNA Skin
Acid Or Alkali? Testing With Cabbage
 Acid Or Alkali? Testing With Cabbage Topic Using vegetables as an acid/base indicator Introduction Forensic scientists need to discover if someone has tampered with liquids (e.g., cosmetics, cleaning products,
Acid Or Alkali? Testing With Cabbage Topic Using vegetables as an acid/base indicator Introduction Forensic scientists need to discover if someone has tampered with liquids (e.g., cosmetics, cleaning products,
ECU Radiation, Biosafety and Hazardous Substances Committee
 Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Parasitology PARASITOLOGY FIXATIVES, REAGENTS & STAINS FIXATIVES, REAGENTS & STAINS. Hymenolepis nana Paragonimus species Diphyllobothrium latum
 FIXATIVES, REAGENTS & STAINS Hymenolepis nana Paragonimus species Diphyllobothrium latum FIXATIVES, REAGENTS & STAINS Clonorchis Hymenolepis diminuta Trichuris trichiura Taenia species Hookworm Fasciola
FIXATIVES, REAGENTS & STAINS Hymenolepis nana Paragonimus species Diphyllobothrium latum FIXATIVES, REAGENTS & STAINS Clonorchis Hymenolepis diminuta Trichuris trichiura Taenia species Hookworm Fasciola
Lab Safety & Volume Measurement. Virtual Science University
 Lab Safety & Volume Measurement Virtual Science University 1 TEK & Objective Lab Safety & Volume Measurement Texas Science TEK B.1(A) Demonstrate safe practices during field and laboratory investigations.
Lab Safety & Volume Measurement Virtual Science University 1 TEK & Objective Lab Safety & Volume Measurement Texas Science TEK B.1(A) Demonstrate safe practices during field and laboratory investigations.
Optiblot SDS-PAGE Gel
 Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. www.abcam.com 1 Table of Contents Optiblot SDS-PAGE Gel 1. Introduction
Instructions for Use For the use in SDS-PAGE with precast gels This product is for research use only and is not intended for diagnostic use. www.abcam.com 1 Table of Contents Optiblot SDS-PAGE Gel 1. Introduction
BSL2 Exposure Control Plan: Human or Non Human Primate Materials
 Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Table 6: Detailed Infection Prevention and Control Procedures for Tattooing and Micropigmentation. Use During Tattooing
 FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
ab Trichrome Stain (Connective Tissue Stain)
 Version 3 Last updated 12 February 2019 ab150686 Trichrome Stain (Connective Tissue Stain) For the histological visualization of collagenous connective tissue fibers in tissue sections. This product is
Version 3 Last updated 12 February 2019 ab150686 Trichrome Stain (Connective Tissue Stain) For the histological visualization of collagenous connective tissue fibers in tissue sections. This product is
Competency. Method of Instruction Codes. Yes / No / RDN. Yes No RDN. Yes No RDN. Yes No RDN. Yes No RDN. Yes No RDN. Yes No RDN.
 JOHNS HOPKINS MEDICAL INSTITUTIONS Competency Validation Tool BIOLOGICAL PPE ENHANCED CONTACT AND DROPLET PRECAUTIONS (POWERED AIR PURIFYING RESPIRATOR (PAPR) OPTION) Employee Name: Role: Unit: Date: DESIRED
JOHNS HOPKINS MEDICAL INSTITUTIONS Competency Validation Tool BIOLOGICAL PPE ENHANCED CONTACT AND DROPLET PRECAUTIONS (POWERED AIR PURIFYING RESPIRATOR (PAPR) OPTION) Employee Name: Role: Unit: Date: DESIRED
under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC
 Page 1 of 7 under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC New Miana Pura East, Roras Road, Sialkot - Pakistan Tel: +92-52-3560135 Fax: +92-52-3563647 E-mail: info@longstoneintl.com
Page 1 of 7 under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC New Miana Pura East, Roras Road, Sialkot - Pakistan Tel: +92-52-3560135 Fax: +92-52-3563647 E-mail: info@longstoneintl.com
Laboratory Orientation. Biological Screening
 Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
ENHANCED CONTACT AND DROPLET PRECAUTIONS PPE CHECKLIST. Trained Observer: Unit: Date: [ ] the floor. 2 Engage Trained Observer and Assistant [ ]
![ENHANCED CONTACT AND DROPLET PRECAUTIONS PPE CHECKLIST. Trained Observer: Unit: Date: [ ] the floor. 2 Engage Trained Observer and Assistant [ ] ENHANCED CONTACT AND DROPLET PRECAUTIONS PPE CHECKLIST. Trained Observer: Unit: Date: [ ] the floor. 2 Engage Trained Observer and Assistant [ ]](/thumbs/78/77108755.jpg) Page 1 of 5 ENHANCED CONTACT AND DROPLET PRECAUTIONS PPE CHECKLIST Trained Observer: Unit: Date: Healthcare Worker: Assistant: Instructions: Check box after completion of each step. Donning of HEALTHCARE
Page 1 of 5 ENHANCED CONTACT AND DROPLET PRECAUTIONS PPE CHECKLIST Trained Observer: Unit: Date: Healthcare Worker: Assistant: Instructions: Check box after completion of each step. Donning of HEALTHCARE
Provide a brief description of the procedure and infectious organisms used:
 Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Exercise 6-C STAINING OF MICROORGANISMS ACID-FAST STAIN
 Exercise 6-C STAINING OF MICROORGANISMS ACID-FAST STAIN Introduction The acid-fast stain is a differential stain that separates bacteria on the basis of the lipid content of their cell walls. Bacteria
Exercise 6-C STAINING OF MICROORGANISMS ACID-FAST STAIN Introduction The acid-fast stain is a differential stain that separates bacteria on the basis of the lipid content of their cell walls. Bacteria
Biosafety Self-Audit Checklist
 Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
CHM111 Lab Physical Separations Grading Rubric
 CHM111 Lab Physical Separations Grading Rubric Name Team Name Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste disposal
CHM111 Lab Physical Separations Grading Rubric Name Team Name Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste disposal
Training Manual. For the Application, Removal and Transport of the PharmChek Drugs of Abuse Patch. (The Sweat Patch is For Professional Use Only)
 PharmChek Drugs of Abuse Sweat Patch Training Manual For the Application, Removal and Transport of the PharmChek Drugs of Abuse Patch (The Sweat Patch is For Professional Use Only) Revised February 2015
PharmChek Drugs of Abuse Sweat Patch Training Manual For the Application, Removal and Transport of the PharmChek Drugs of Abuse Patch (The Sweat Patch is For Professional Use Only) Revised February 2015
STANDARD: Laboratory Safety Effective: March 20, 2018
 University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
Experiment #3. Physical Separations Candy Chromatography
 Experiment #3. Physical Separations Candy Chromatography Goals 1. To physically separate and identify dyes in candy by comparison to commercial food dyes using paper chromatography. 2. To become familiar
Experiment #3. Physical Separations Candy Chromatography Goals 1. To physically separate and identify dyes in candy by comparison to commercial food dyes using paper chromatography. 2. To become familiar
Ventricular Assist Device (VAD) Exit Site Care Guidelines
 Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
Dextrose 25%solution or equivalent. Dextrose 5%+normal saline.9% solution Adhesive fabric tape,stretchable,easily cut roll
 Item no. Item description Unit Qty.. 3. 4.. 6. 7. 8. 9. 0... 3. 4.. 6. 7. 8. 9. 0... 3. 4.. 6. 7. 8. 9. 30. 3. 3. 33. 34. 3. 36. 37. 38. 39. 40. 4. 4. Dextrose %solution or equivalent Dextrose %solution
Item no. Item description Unit Qty.. 3. 4.. 6. 7. 8. 9. 0... 3. 4.. 6. 7. 8. 9. 0... 3. 4.. 6. 7. 8. 9. 30. 3. 3. 33. 34. 3. 36. 37. 38. 39. 40. 4. 4. Dextrose %solution or equivalent Dextrose %solution
TRANSGENDER HEALTH Injection Guide
 TRANSGENDER HEALTH Injection Guide 1of the Southern Finger Lakes This information in this booklet has been adapted with permission from a handbook created by Fenway Health. Fenwayhealth.org The instructions
TRANSGENDER HEALTH Injection Guide 1of the Southern Finger Lakes This information in this booklet has been adapted with permission from a handbook created by Fenway Health. Fenwayhealth.org The instructions
SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
The Effectiveness of Water, Sodium Hypochlorite Bleach, and Peroxyacetic Acid (PAA) in Eradicating Listeria monocytogenes
 The Effectiveness of Water, Sodium Hypochlorite Bleach, and Peroxyacetic Acid (PAA) in Eradicating Listeria monocytogenes from the Surface of Cantaloupes Background Tina Rodrigues, B.S. Jonathan Howarth
The Effectiveness of Water, Sodium Hypochlorite Bleach, and Peroxyacetic Acid (PAA) in Eradicating Listeria monocytogenes from the Surface of Cantaloupes Background Tina Rodrigues, B.S. Jonathan Howarth
Slide 1. Slide 2. Slide 3 EVIDENCE COLLECTION. Evidence Collection 101. Physical Evidence
 Slide 1 EVIDENCE COLLECTION YWCA - Oklahoma City Created by Shannon Liew, RN, BSN, SANE-A Slide 2 Evidence Collection 101 Two crime scenes in sexual assault cases: Place of the assault Patient s body The
Slide 1 EVIDENCE COLLECTION YWCA - Oklahoma City Created by Shannon Liew, RN, BSN, SANE-A Slide 2 Evidence Collection 101 Two crime scenes in sexual assault cases: Place of the assault Patient s body The
Glycoprotein Staining Kit
 193PR 02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glycoprotein Staining Kit With Rapidstain for Enhanced Glycoprotein & Non Glycoproteins
193PR 02 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Glycoprotein Staining Kit With Rapidstain for Enhanced Glycoprotein & Non Glycoproteins
Biological Safety Training
 Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION
 State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860. Effective Date: August 31, 2006
 COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
