For the use only of Registered Medical Practitioners or a Hospital or a Laboratory DERMOCALM LOTION. Calamine and Light Liquid Paraffin Lotion
|
|
|
- Arnold Morris
- 5 years ago
- Views:
Transcription
1 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory DERMOCALM LOTION and Light Liquid Paraffin Lotion QUALITATIVE AND QUANTITATIVE COMPOSITION * IP Light Liquid Paraffin IP in a lotion base 8.0 % w/w 10.0 % w/w * is Zinc Oxide with a small proportion of ferric oxide. PHARMACEUTICAL FORM Lotion. CLINICAL PARTICULARS Therapeutic Indications DERMOCALM LOTION is indicated for use as an anti-pruritic and smoothening agent. Posology and Method of Administration Adults For Topical/cutaneous use. Dermocalm lotion to be applied gently on the affected area of the skin. Apply on the affected zone as many times as required. Children Elderly Renal impairment Hepatic impairment 1
2 Contraindications DERMOCALM LOTION is contraindicated in hypersensitivity to any of the ingredients of this product. Special warnings and Special Precautions for Use X-ray examination: should not be applied onto the skin before an X-ray examination because it may affect the outcome of the radiograph. Light liquid paraffin should be used with caution in patients with a known hypersensitivity or allergy to light liquid paraffin or to any of the excipients in the preparation. Discontinue use, if skin irritation occurs. Avoid entry of Dermocalm lotion in the eyes or mouth. Interaction with Other Medicaments and Other Forms of Interaction Benzyl penicillin Zinc oxide is incompatible with benzyl penicillin. Fatty acids Zinc oxide reacts slowly with fatty acid in oils and fats to produce the corresponding fatty acid esters. No drug interaction studies have been conducted with cutaneous light liquid paraffin. Considering the low level of systemic absorption, drug interactions are unlikely to occur. Pregnancy and Lactation Fertility Pregnancy and Lactation The safety of DERMOCALM LOTION during pregnancy and lactation has not been established. No adverse effects have been reported with calamine. No effects of light liquid paraffin during pregnancy are anticipated, since systemic exposure to light liquid paraffin is expected to be low. It is not known if light liquid paraffin is excreted in human milk. Risk to the infant is likely to be low since systemic exposure is low. 2
3 Patients should be advised to ensure that any residual product is fully washed off the breast prior to breast feeding. Effects on Ability to Drive and Use Machines Undesirable effects Light liquid paraffin Adverse drug reactions (ADR) are listed below by MedDRA system organ class and by frequency. Frequencies are defined as: very common ( 1/10), common ( 1/100 to <1/10), uncommon ( 1/1,000 to <1/100), rare ( 1/10,000 to <1/1,000) and very rare (<1/10,000), including isolated reports. Post marketing data: Skin and subcutaneous Tissue Disorders Rare: Application site reactions including application site irritation, rash, erythema, pruritus. Immune System Disorders Rare: Application site hypersensitivity reactions including application site dermatitis. Overdose Symptoms and signs There are no relevant data available for calamine. The product is intended for cutaneous use only Accidental ingestion of light liquid paraffin may cause gastrointestinal irritation with nausea, vomiting and diarrhea. Treatment No special procedures or anti-dote is likely to be necessary. In case of overuse wash off with soap and hot water. In case of accidental ingestion, management should be as clinically indicated or as recommended by the national poisons centre, where available. PHARMACOLOGICAL PROPERTIES Pharmacodynamic Properties Pharmacotherapeutic Group 3
4 : Emollients and protectives; ATC Code: D02AB. Light Liquid Paraffin: Dermatologicals, other emollients and protectives; ATC code: D02AX. Mechanism of Action/ Pharmacodynamic effects has a mild astringent antipruritic and soothing action on the skin. has astringent effects in the skin or mucous membranes by coagulating protein. The protein precipitates which forms a protective coat, allowing new tissue to regenerate underneath. It is commonly used to reduce the extent of weeping in dermatitis and to check oozing and discharge in other mild disorders. Light Liquid Paraffin Light liquid paraffin exerts an emollient effect by forming an occlusive film on the stratum corneum. Pharmacokinetic Properties is unlikely to be absorbed through the skin. Light Liquid Paraffin There have been no studies to investigate the pharmacokinetics of cutaneously applied light liquid paraffin. It is chemically and biologically inert; systemic uptake is considered to be minimal (<1%). Preclinical safety data PHARMACEUTICAL PARTICULARS List of excipients Aloe Vera gel, Glycerin, Bentonite, Brij S2-SO-(TH), Brij-S21-FL-(TH), Silicone Oil 350 cs, Dow corning 1403 fluid, Magnesium Aluminium Silicate, Perfume composition 12447, Citric Acid Monohydrate, Methylparaben, Propylparaben. Incompatibilities Shelf life 4
5 The expiry date is indicated on the label and packaging. Special Precautions for Storage Store at a temperature not exceeding 30 C. Do not freeze Keep out of reach of children. Nature and Specifications of Container Bottle in a carton. All pack presentations may not be marketed in the country. Instruction for Use/Handling: For external use only. Shake the bottle before use. There are no other special requirements for use or handling of this product. For further information please contact: GlaxoSmithKline Pharmaceuticals Limited Registered Office Dr Annie Besant Road, Worli, Mumbai India. Trade marks are owned by or licensed to the GSK group of companies Version: DER/PI/IN/2018/ 02 dated 05 Sep 2018 Adapted from NCDS v 02 dated 08-June-2012 and Light Liquid Paraffin GDS 01 dated 24-September
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM EMOLLIENT. Light Liquid Paraffin Emollient
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM EMOLLIENT Light Liquid Paraffin Emollient QUALITATIVE AND QUANTITATIVE COMPOSITION Light Liquid Paraffin IP Base
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM EMOLLIENT Light Liquid Paraffin Emollient QUALITATIVE AND QUANTITATIVE COMPOSITION Light Liquid Paraffin IP Base
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL CREAMY WASH. Benzoyl Peroxide Creamy Wash 4%
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL CREAMY WASH Benzoyl Peroxide Creamy Wash 4% QUALITATIVE AND QUANTITATIVE COMPOSITION Hydrous Benzoyl Peroxide
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL CREAMY WASH Benzoyl Peroxide Creamy Wash 4% QUALITATIVE AND QUANTITATIVE COMPOSITION Hydrous Benzoyl Peroxide
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL HYDROPHASE. Benzoyl Peroxide Cream IP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL HYDROPHASE Benzoyl Peroxide Cream IP QUALITATIVE AND QUANTITATIVE COMPOSITION Hydrous Benzoyl Peroxide IP equivalent
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BREVOXYL HYDROPHASE Benzoyl Peroxide Cream IP QUALITATIVE AND QUANTITATIVE COMPOSITION Hydrous Benzoyl Peroxide IP equivalent
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory NEDAX -5 LOTION. Permethrin Lotion 5% w/w
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory NEDAX -5 LOTION Permethrin Lotion 5% w/w QUALITATIVE AND QUANTITATIVE COMPOSITION Permethrin 5% w/w in a aqueous base
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory NEDAX -5 LOTION Permethrin Lotion 5% w/w QUALITATIVE AND QUANTITATIVE COMPOSITION Permethrin 5% w/w in a aqueous base
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Product Summary 1. Name of the Medicinal Product Bactroban 2% Nasal Ointment 2. Qualitative and Quantitative Composition Each gram of nasal ointment contains mupirocin
SUMMARY OF PRODUCT CHARACTERISTICS Product Summary 1. Name of the Medicinal Product Bactroban 2% Nasal Ointment 2. Qualitative and Quantitative Composition Each gram of nasal ointment contains mupirocin
POLYTAR Plus Liquid PRODUCT INFORMATION. Polytar Plus Liquid medicated scalp cleanser, contains coal tar solution.
 NAME OF THE MEDICINE POLYTAR Plus Liquid PRODUCT INFORMATION Polytar Plus Liquid medicated scalp cleanser, contains coal tar solution. DESCRIPTION Polytar Plus Liquid contains coal tar solution 4% w/w.
NAME OF THE MEDICINE POLYTAR Plus Liquid PRODUCT INFORMATION Polytar Plus Liquid medicated scalp cleanser, contains coal tar solution. DESCRIPTION Polytar Plus Liquid contains coal tar solution 4% w/w.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Drapolene Cream 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Drapolene contains: Benzalkonium chloride solution 0.02% w/w Equivalent to
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Drapolene Cream 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Drapolene contains: Benzalkonium chloride solution 0.02% w/w Equivalent to
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BACTROBAN CREAM. Mupirocin Cream USP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BACTROBAN CREAM Mupirocin Cream USP QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin Calcium USP equivalent to Mupirocin
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory BACTROBAN CREAM Mupirocin Cream USP QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin Calcium USP equivalent to Mupirocin
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory T-BACT OINTMENT. Mupirocin Ointment IP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory T-BACT OINTMENT Mupirocin Ointment IP QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin IP 2.0% w/w in a non-greasy base
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory T-BACT OINTMENT Mupirocin Ointment IP QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin IP 2.0% w/w in a non-greasy base
Each gram of mupirocin ointment contains 20 mg of mupirocin (2% w/w) as the active ingredient.
 AUSTRALIAN PRODUCT INFORMATION APO-Mupirocin, 20 mg/g (2% w/w) Ointment 1. NAME OF THE MEDICINE Mupirocin 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of mupirocin ointment contains 20 mg of mupirocin
AUSTRALIAN PRODUCT INFORMATION APO-Mupirocin, 20 mg/g (2% w/w) Ointment 1. NAME OF THE MEDICINE Mupirocin 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of mupirocin ointment contains 20 mg of mupirocin
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Duac Once Daily 10 mg/g + 50 mg/g Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g of gel contains: 10 mg clindamycin as clindamycin
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Duac Once Daily 10 mg/g + 50 mg/g Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g of gel contains: 10 mg clindamycin as clindamycin
BACTROBAN CREAM QUALITATIVE AND QUANTITATIVE COMPOSITION
 BACTROBAN CREAM Mupirocin calcium QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin calcium equivalent to 2% w/w mupirocin free acid. PHARMACEUTICAL FORM White cream for topical administration in a multi-use
BACTROBAN CREAM Mupirocin calcium QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin calcium equivalent to 2% w/w mupirocin free acid. PHARMACEUTICAL FORM White cream for topical administration in a multi-use
Package leaflet: Information for the user. ZORAC 0.05% gel ZORAC 0.1% gel. Tazarotene
 Package leaflet: Information for the user ZORAC 0.05% gel ZORAC 0.1% gel Tazarotene Read all of this leaflet carefully before you start using this medicine because it contains important information for
Package leaflet: Information for the user ZORAC 0.05% gel ZORAC 0.1% gel Tazarotene Read all of this leaflet carefully before you start using this medicine because it contains important information for
DAKTARIN. Miconazole, Miconazole nitrate CREAM, POWDER, SPRAY POWDER, LOTION, TINCTURE PRODUCT INFORMATION
 DAKTARIN Miconazole, Miconazole nitrate CREAM, POWDER, SPRAY POWDER, LOTION, TINCTURE PRODUCT INFORMATION Description Miconazole is 1-[2,4-dichloro-beta-(2,4-dichlorobenzyloxy)phenethyl]imidazole derivative
DAKTARIN Miconazole, Miconazole nitrate CREAM, POWDER, SPRAY POWDER, LOTION, TINCTURE PRODUCT INFORMATION Description Miconazole is 1-[2,4-dichloro-beta-(2,4-dichlorobenzyloxy)phenethyl]imidazole derivative
BACTROBAN OINTMENT. Mupirocin free acid. Bacterial skin infections, e.g. impetigo, folliculitis and furunculosis.
 BACTROBAN OINTMENT Mupirocin free acid QUALITATIVE AND QUANTITATIVE COMPOSITION 2% w/w mupirocin free acid in a white, translucent, water soluble, polyethylene glycol base. PHARMACEUTICAL FORM Ointment.
BACTROBAN OINTMENT Mupirocin free acid QUALITATIVE AND QUANTITATIVE COMPOSITION 2% w/w mupirocin free acid in a white, translucent, water soluble, polyethylene glycol base. PHARMACEUTICAL FORM Ointment.
New Zealand Datasheet
 New Zealand Datasheet 1 PRODUCT NAME LOCERYL 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Amorolfine Nail Lacquer 5% 3 PHARMACEUTICAL FORM LOCERYL nail lacquer is a clear colourless liquid. 4 CLINICAL PARTICULARS
New Zealand Datasheet 1 PRODUCT NAME LOCERYL 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Amorolfine Nail Lacquer 5% 3 PHARMACEUTICAL FORM LOCERYL nail lacquer is a clear colourless liquid. 4 CLINICAL PARTICULARS
4.3. Contraindications Hypersensitivity to the active substances or to any of the excipients listed in section 6.1.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Tactuo 0.1% / 2.5% gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g of gel contains: Adapalene 1 mg (0.1 %) Benzoyl Peroxide 25 mg
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Tactuo 0.1% / 2.5% gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g of gel contains: Adapalene 1 mg (0.1 %) Benzoyl Peroxide 25 mg
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Epiduo 0.3% / 2.5% gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of gel contains: adapalene 3 mg (0.3%) benzoyl peroxide 25
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Epiduo 0.3% / 2.5% gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of gel contains: adapalene 3 mg (0.3%) benzoyl peroxide 25
The solution contains isopropyl alcohol 50% v/v, propylene glycol, and water.
 Cleocin T (clindamycin phosphate topical solution, USP) (clindamycin phosphate topical gel) (clindamycin phosphate topical lotion) For External Use DESCRIPTION CLEOCIN T Topical Solution and CLEOCIN T
Cleocin T (clindamycin phosphate topical solution, USP) (clindamycin phosphate topical gel) (clindamycin phosphate topical lotion) For External Use DESCRIPTION CLEOCIN T Topical Solution and CLEOCIN T
DUAL ACTION LOGO. Quinoderm 10% / 0.5% w/w Cream Benzoyl Peroxide, hydrous / Potassium Hydroxyquinoline Sulfate
 DUAL ACTION LOGO Quinoderm 10% / 0.5% w/w Cream Benzoyl Peroxide, hydrous / Potassium Hydroxyquinoline Sulfate Read all of this leaflet carefully before you start using this medicine because it contains
DUAL ACTION LOGO Quinoderm 10% / 0.5% w/w Cream Benzoyl Peroxide, hydrous / Potassium Hydroxyquinoline Sulfate Read all of this leaflet carefully before you start using this medicine because it contains
New Zealand Datasheet
 New Zealand Datasheet 1 PRODUCT NAME Epiduo 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Adapalene 0.1% / benzoyl peroxide 2.5% gel 3 PHARMACEUTICAL FORM Epiduo is a white to very pale yellow opaque gel
New Zealand Datasheet 1 PRODUCT NAME Epiduo 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Adapalene 0.1% / benzoyl peroxide 2.5% gel 3 PHARMACEUTICAL FORM Epiduo is a white to very pale yellow opaque gel
Rheumon Gel 1 g of gel contains 50 mg of etofenamate. For use in adults.
 New Zealand Consumer Medicine Information Rheumon Gel 1 g of gel contains 50 mg of etofenamate. For use in adults. Read all of this leaflet carefully before you start using this medicine. This medicine
New Zealand Consumer Medicine Information Rheumon Gel 1 g of gel contains 50 mg of etofenamate. For use in adults. Read all of this leaflet carefully before you start using this medicine. This medicine
PRODUCT INFORMATION BREVOXYL CREAM
 NAME F THE MEDICINE Brevoxyl contains benzoyl peroxide. PRDUCT INFRMATIN BREVXYL CREAM DESCRIPTIN Benzoyl peroxide is a white powder with the following chemical structure: The compound has anti-bacterial
NAME F THE MEDICINE Brevoxyl contains benzoyl peroxide. PRDUCT INFRMATIN BREVXYL CREAM DESCRIPTIN Benzoyl peroxide is a white powder with the following chemical structure: The compound has anti-bacterial
Your Manufacturer. of Pharmaceuticals. Licensing & Contract manufacturing
 Your Manufacturer of Pharmaceuticals Licensing & Contract manufacturing Delicate and healthy skin of Your baby Hascosan... as delicate as Your baby s skin Hascosan Baby bath lotion Bath lotion is used
Your Manufacturer of Pharmaceuticals Licensing & Contract manufacturing Delicate and healthy skin of Your baby Hascosan... as delicate as Your baby s skin Hascosan Baby bath lotion Bath lotion is used
ACETOCAUSTIN 0,5 ml, Cutaneous solution
 PACKAGE LEAFLET: INFORMATION FOR THE USER ACETOCAUSTIN 0,5 ml, Cutaneous solution MONOCHLOROACETIC ACID This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet
PACKAGE LEAFLET: INFORMATION FOR THE USER ACETOCAUSTIN 0,5 ml, Cutaneous solution MONOCHLOROACETIC ACID This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet
MUPINASE Ointment/ Cream (Mupirocin)
 Published on: 10 Jul 2014 MUPINASE Ointment/ Cream (Mupirocin) Composition MUPINASE Ointment Mupirocin, USP.2.0% w/w Ointment base..q. s. MUPINASE Cream Mupirocin, USP.2.0% w/w Dosage Forms Ointment and
Published on: 10 Jul 2014 MUPINASE Ointment/ Cream (Mupirocin) Composition MUPINASE Ointment Mupirocin, USP.2.0% w/w Ointment base..q. s. MUPINASE Cream Mupirocin, USP.2.0% w/w Dosage Forms Ointment and
New Zealand Datasheet. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Adapalene 0.1% topical gel
 New Zealand Datasheet 1 PRODUCT NAME DIFFERIN 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Adapalene 0.1% topical gel 3 PHARMACEUTICAL FORM DIFFERIN topical gel is a smooth white gel containing 1 mg/g adapalene.
New Zealand Datasheet 1 PRODUCT NAME DIFFERIN 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Adapalene 0.1% topical gel 3 PHARMACEUTICAL FORM DIFFERIN topical gel is a smooth white gel containing 1 mg/g adapalene.
PACKAGE LEAFLET: INFORMATION FOR THE USER. CURANAIL 5%, medicated nail lacquer Amorolfine
 Medicinal product name Outline PACKAGE LEAFLET: INFORMATION FOR THE USER CURANAIL 5%, medicated nail lacquer Amorolfine Read all of this leaflet carefully before you start using this medicine because it
Medicinal product name Outline PACKAGE LEAFLET: INFORMATION FOR THE USER CURANAIL 5%, medicated nail lacquer Amorolfine Read all of this leaflet carefully before you start using this medicine because it
Package leaflet: Information for the user. Acnatac 10 mg/g mg/g gel clindamycin and tretinoin
 Package leaflet: Information for the user Acnatac 10 mg/g + 0.25 mg/g gel clindamycin and tretinoin Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user Acnatac 10 mg/g + 0.25 mg/g gel clindamycin and tretinoin Read all of this leaflet carefully before you start using this medicine because it contains important
EpiCeram Topical therapeutic Skin Barrier Emulsion
 EpiCeram Topical therapeutic Skin Barrier Emulsion PEDIAPHARM INC. Date of preparation: August 31, 2010 Summary Product Information: EpiCeram Skin Barrier Emulsion is a steroid-free, fragrance - free,
EpiCeram Topical therapeutic Skin Barrier Emulsion PEDIAPHARM INC. Date of preparation: August 31, 2010 Summary Product Information: EpiCeram Skin Barrier Emulsion is a steroid-free, fragrance - free,
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION. NIX Dermal Cream. Permethrin Cream, 5% w/w. Topical Scabicidal Agent
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION NIX Dermal Cream Permethrin Cream, 5% w/w Topical Scabicidal Agent GlaxoSmithKline Consumer Healthcare Inc. 7333 Mississauga Road Mississauga,
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION NIX Dermal Cream Permethrin Cream, 5% w/w Topical Scabicidal Agent GlaxoSmithKline Consumer Healthcare Inc. 7333 Mississauga Road Mississauga,
Scabies. Dr. Ghassan Salah
 Scabies Dr. Ghassan Salah Scabies Scabies is a skin infestation caused by a mite Causes: The female scabies mite (Sarcoptes scabei ) burrows through the stratum corneum of the skin and lays its eggs just
Scabies Dr. Ghassan Salah Scabies Scabies is a skin infestation caused by a mite Causes: The female scabies mite (Sarcoptes scabei ) burrows through the stratum corneum of the skin and lays its eggs just
PATIENT INFORMATION LEAFLET. Hydromol HC Intensive Hydrocortisone and urea
 PATIENT INFORMATION LEAFLET Hydromol HC Intensive Hydrocortisone and urea Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again. If you
PATIENT INFORMATION LEAFLET Hydromol HC Intensive Hydrocortisone and urea Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again. If you
EMOLLIENTS. NEW 500g NEW. pump. Up to 37% Wider choice, greater savings! cost savings. without compromising on quality or patient care!
 CREAMS BATH ADDITIVES SOAP SUBSTITUTES OINTMENT GEL Up to 37% cost savings without compromising on quality or patient care! Wider choice, greater savings! emollients A wider choice with greater savings!
CREAMS BATH ADDITIVES SOAP SUBSTITUTES OINTMENT GEL Up to 37% cost savings without compromising on quality or patient care! Wider choice, greater savings! emollients A wider choice with greater savings!
Package leaflet: Information for the user Rogaine 50 mg/g, cutaneous foam. minoxidil
 Package leaflet: Information for the user Rogaine 50 mg/g, cutaneous foam minoxidil Read all of this leaflet carefully before you start using this medicine because it contains important information for
Package leaflet: Information for the user Rogaine 50 mg/g, cutaneous foam minoxidil Read all of this leaflet carefully before you start using this medicine because it contains important information for
Prescribing Information. phisohex (hexachlorophene) Bottles of 150 ml, 450 ml. Pharmaceutical standard: USP. Antibacterial Skin Cleanser
 Prescribing Information phisohex (hexachlorophene) Bottles of 150 ml, 450 ml Pharmaceutical standard: USP Antibacterial Skin Cleanser sanofi-aventis Canada, Inc. 2150 St. Elzear Blvd. West Laval, Quebec
Prescribing Information phisohex (hexachlorophene) Bottles of 150 ml, 450 ml Pharmaceutical standard: USP Antibacterial Skin Cleanser sanofi-aventis Canada, Inc. 2150 St. Elzear Blvd. West Laval, Quebec
PACKAGE LEAFLET. EN Elastoplast Heat Plaster 11 mg January
 PACKAGE LEAFLET EN Elastoplast Heat Plaster 11 mg January 2013 1 Package leaflet: Information for the user Elastoplast Heat Plaster11 mg medicated Plaster Soft extract of cayenne pepper (equiv. to 11 mg
PACKAGE LEAFLET EN Elastoplast Heat Plaster 11 mg January 2013 1 Package leaflet: Information for the user Elastoplast Heat Plaster11 mg medicated Plaster Soft extract of cayenne pepper (equiv. to 11 mg
Package leaflet: Information for the patient. Clindamycin ABR 150 mg capsules, hard. Clindamycin ABR 300 mg capsules, hard
 Package leaflet: Information for the patient Clindamycin ABR 150 mg capsules, hard Clindamycin ABR 300 mg capsules, hard Clindamycin ABR 600 mg capsules, hard Clindamycin Read all of this leaflet carefully
Package leaflet: Information for the patient Clindamycin ABR 150 mg capsules, hard Clindamycin ABR 300 mg capsules, hard Clindamycin ABR 600 mg capsules, hard Clindamycin Read all of this leaflet carefully
Eumovate Cream clobetasone butyrate
 Package leaflet: Information for the user Eumovate Cream clobetasone butyrate Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
Package leaflet: Information for the user Eumovate Cream clobetasone butyrate Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
Although clindamycin phosphate is inactive in vitro, rapid in vivo hydrolysis converts this compound to the
 APPROVED PACKAGE INSERT SCHEDULING STATUS:S4 PROPRIETARY NAME AND DOSAGE FORM: DALACIN T ( Solution) DALACIN T (Lotion) COMPOSITION: DALACIN T Solution contains the following per ml : Clindamycin phosphate
APPROVED PACKAGE INSERT SCHEDULING STATUS:S4 PROPRIETARY NAME AND DOSAGE FORM: DALACIN T ( Solution) DALACIN T (Lotion) COMPOSITION: DALACIN T Solution contains the following per ml : Clindamycin phosphate
Occlusal 26%w/w cutaneous solution salicylic acid
 Occlusal 26%w/w cutaneous solution salicylic acid Information for Patients Read all of this leaflet carefully because it contains important information for you. This medicine is available without prescription.
Occlusal 26%w/w cutaneous solution salicylic acid Information for Patients Read all of this leaflet carefully because it contains important information for you. This medicine is available without prescription.
PATIENT INFORMATION LEAFLET. Alphaderm 1% & 10% w/w Cream hydrocortisone and urea
 PATIENT INFORMATION LEAFLET Alphaderm 1% & 10% w/w Cream hydrocortisone and urea Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again.
PATIENT INFORMATION LEAFLET Alphaderm 1% & 10% w/w Cream hydrocortisone and urea Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again.
Consumer Medicine Information. Topical Cream Clotrimazole 10 mg/g & Hydrocortisone 10 mg/g
 CANESTEN PLUS Topical Cream Clotrimazole 10 mg/g & Hydrocortisone 10 mg/g What is in this Leaflet This leaflet answers some common questions about CANESTEN PLUS Clotrimazole and Hydrocortisone Cream. It
CANESTEN PLUS Topical Cream Clotrimazole 10 mg/g & Hydrocortisone 10 mg/g What is in this Leaflet This leaflet answers some common questions about CANESTEN PLUS Clotrimazole and Hydrocortisone Cream. It
PACKAGE LEAFLET: INFORMATION FOR THE USER. Isotrex Gel isotretinoin 0.05% w/w
 PACKAGE LEAFLET: INFORMATION FOR THE USER Isotrex Gel isotretinoin 0.05% w/w Read all of this leaflet carefully before you start using this medicine because it contains important information for you. -
PACKAGE LEAFLET: INFORMATION FOR THE USER Isotrex Gel isotretinoin 0.05% w/w Read all of this leaflet carefully before you start using this medicine because it contains important information for you. -
Safety Data Sheet Version Extra Volume Shampoo For All Hair Types
 1. IDENTIFICATION OF THE PRODUCT AND OF THE COMPANY Product name : Product code : MO-EVS250 Use of the product : Personal hair care product Company Office : Moroccanoil Israel Ltd. 16 Moshe Levi. St. Kennedy
1. IDENTIFICATION OF THE PRODUCT AND OF THE COMPANY Product name : Product code : MO-EVS250 Use of the product : Personal hair care product Company Office : Moroccanoil Israel Ltd. 16 Moshe Levi. St. Kennedy
EPIDUO GEL PRODUCT INFORMATION
 EPIDUO GEL PRODUCT INFORMATION NAME OF THE MEDICINE EPIDUO Topical gel: 0.1% adapalene + 2.5% benzoyl peroxide Common Names: Adapalene and benzoyl peroxide Chemical Name : - Adapalene: 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic
EPIDUO GEL PRODUCT INFORMATION NAME OF THE MEDICINE EPIDUO Topical gel: 0.1% adapalene + 2.5% benzoyl peroxide Common Names: Adapalene and benzoyl peroxide Chemical Name : - Adapalene: 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic
Package Leaflet: Information for the User
 GSK Logo Package Leaflet: Information for the User Betnovate Lotion betamethasone valerate Read all of this leaflet carefully before you start using this medicine because it contains important information
GSK Logo Package Leaflet: Information for the User Betnovate Lotion betamethasone valerate Read all of this leaflet carefully before you start using this medicine because it contains important information
HOW DOES DERMA SHIELD WORK?
 about Derma Shield HOW DOES DERMA SHIELD WORK? Derma Shield has specially formulated ingredients which form a chemical molecular bond with the epithelial or top layer of the skin. The mesh-like bond actually
about Derma Shield HOW DOES DERMA SHIELD WORK? Derma Shield has specially formulated ingredients which form a chemical molecular bond with the epithelial or top layer of the skin. The mesh-like bond actually
ISOTREX GEL Isotretinoin 0.05% w/w
 ISOTREX GEL Isotretinoin 0.05% w/w Consumer Medicine Information What is in this leaflet Please read this leaflet carefully before you use Isotrex Gel. This leaflet answers some common questions about
ISOTREX GEL Isotretinoin 0.05% w/w Consumer Medicine Information What is in this leaflet Please read this leaflet carefully before you use Isotrex Gel. This leaflet answers some common questions about
Hydroform Cream 1% Hydrocortisone (microfine) 1% w/w and clioquinol 1% w/ w
 Hydroform Cream 1% Hydrocortisone (microfine) 1% w/w and clioquinol 1% w/ w Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about Hydroform Cream 1%. It
Hydroform Cream 1% Hydrocortisone (microfine) 1% w/w and clioquinol 1% w/ w Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about Hydroform Cream 1%. It
PATIENT INFORMATION LEAFLET. Timodine Cream
 PATIENT INFORMATION LEAFLET Timodine Cream Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again. If you have any further questions, ask
PATIENT INFORMATION LEAFLET Timodine Cream Read all of this leaflet carefully before you start using this medicine Keep this leaflet. You may need to read it again. If you have any further questions, ask
Eumovate Ointment clobetasone butyrate
 [GlaxoSmithKline Logo] Package Leaflet: Information for the User Eumovate Ointment clobetasone butyrate Read all of this leaflet carefully before you start using this medicine because it contains important
[GlaxoSmithKline Logo] Package Leaflet: Information for the User Eumovate Ointment clobetasone butyrate Read all of this leaflet carefully before you start using this medicine because it contains important
DP Fusidic Acid Cream
 New Zealand Consumer Medicine Information DP Fusidic Acid Cream Fusidic Acid 20 mg/g (2 %) Cream What is in this leaflet Please read this leaflet carefully before you start using DP Fusidic Acid Cream.
New Zealand Consumer Medicine Information DP Fusidic Acid Cream Fusidic Acid 20 mg/g (2 %) Cream What is in this leaflet Please read this leaflet carefully before you start using DP Fusidic Acid Cream.
: Promat Glue K84 : Viscous liquid. : Glue, resistant to high temperature
 SECTION 1 Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Trade name Identification of the product Type of product : : Viscous liquid. : Glue, resistant to
SECTION 1 Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Trade name Identification of the product Type of product : : Viscous liquid. : Glue, resistant to
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Vaniqa 11.5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of cream contains 115 mg of eflornithine (as hydrochloride
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Vaniqa 11.5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of cream contains 115 mg of eflornithine (as hydrochloride
Retin-A 0.05% w/w cream
 PACKAGE LEAFLET: INFORMATION FOR THE USER Retin-A 0.05% w/w cream Tretinoin Retin-A is a registered trademark Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER Retin-A 0.05% w/w cream Tretinoin Retin-A is a registered trademark Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. TRAVATAN Z Travoprost Ophthalmic Solution
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr TRAVATAN Z Travoprost Ophthalmic Solution Read this carefully before you start taking TRAVATAN Z and each time you
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr TRAVATAN Z Travoprost Ophthalmic Solution Read this carefully before you start taking TRAVATAN Z and each time you
New Zealand Data Sheet
 1 New Zealand Data Sheet 1 (2% Ointment) MUPIDER 2% Ointment 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mupider 2% Ointment contains 20 mg mupirocin per gram in polyethylene glycol base. For the full list
1 New Zealand Data Sheet 1 (2% Ointment) MUPIDER 2% Ointment 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mupider 2% Ointment contains 20 mg mupirocin per gram in polyethylene glycol base. For the full list
PACKAGE LEAFLET: INFORMATION FOR THE USER. CURANAIL 5%, medicated nail lacquer Amorolfine
 Medicinal product name Outline PACKAGE LEAFLET: INFORMATION FOR THE USER CURANAIL 5%, medicated nail lacquer Amorolfine Read all of this leaflet carefully before you start using this medicine because it
Medicinal product name Outline PACKAGE LEAFLET: INFORMATION FOR THE USER CURANAIL 5%, medicated nail lacquer Amorolfine Read all of this leaflet carefully before you start using this medicine because it
Each gram of gel contains 10 mg (1%) clindamycin (as clindamycin phosphate) and 0.25 mg (0.025%) tretinoin.
 1. NAME OF THE MEDICINAL PRODUCT Acnatac 10 mg/g + 0.25 mg/g gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of gel contains 10 mg (1%) clindamycin (as clindamycin phosphate) and 0.25 mg (0.025%)
1. NAME OF THE MEDICINAL PRODUCT Acnatac 10 mg/g + 0.25 mg/g gel 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram of gel contains 10 mg (1%) clindamycin (as clindamycin phosphate) and 0.25 mg (0.025%)
Daktacort 2% / 1% w/w cream
 PACKAGE LEAFLET: INFORMATION FOR THE USER Daktacort 2% / 1% w/w cream Miconazole nitrate / hydrocortisone Daktacort is a registered trademark Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER Daktacort 2% / 1% w/w cream Miconazole nitrate / hydrocortisone Daktacort is a registered trademark Read all of this leaflet carefully before you start using this
Package leaflet: Information for the patient. Onytec 80 mg/g medicated nail lacquer For adults Ciclopirox
 Package leaflet: Information for the patient Onytec 80 mg/g medicated nail lacquer For adults Ciclopirox [For medicines available only on prescription] Read all of this leaflet carefully before you start
Package leaflet: Information for the patient Onytec 80 mg/g medicated nail lacquer For adults Ciclopirox [For medicines available only on prescription] Read all of this leaflet carefully before you start
Material Safety Data Sheet
 Material Safety Data Sheet 1. PRODUCT AND COMPANY IDENTIFICATION Product: Design Essentials Natural Honey Curl Forming Custard with Honey &Chamomile Manufacturer: Draga Laboratories 3435 Empire Blvd Atlanta,
Material Safety Data Sheet 1. PRODUCT AND COMPANY IDENTIFICATION Product: Design Essentials Natural Honey Curl Forming Custard with Honey &Chamomile Manufacturer: Draga Laboratories 3435 Empire Blvd Atlanta,
BAXTER ANNOUNCES U.S. FDA APPROVAL AND COMMERCIAL LAUNCH OF READY-TO-USE CLINDAMYCIN INJECTION IN SALINE
 FOR IMMEDIATE RELEASE Media Contact Eric Tatro, (224) 948-5353 media@baxter.com Investor Contact Clare Trachtman, (224) 948-3085 BAXTER ANNOUNCES U.S. FDA APPROVAL AND COMMERCIAL LAUNCH OF READY-TO-USE
FOR IMMEDIATE RELEASE Media Contact Eric Tatro, (224) 948-5353 media@baxter.com Investor Contact Clare Trachtman, (224) 948-3085 BAXTER ANNOUNCES U.S. FDA APPROVAL AND COMMERCIAL LAUNCH OF READY-TO-USE
PACKAGE LEAFLET: INFORMATION FOR THE USER. Clindamycin 300 mg Capsules Clindamycin (as hydrochloride)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Clindamycin 300 mg Capsules Clindamycin (as hydrochloride) Read all of this leaflet carefully before you start taking this medicine because it contains important
PACKAGE LEAFLET: INFORMATION FOR THE USER Clindamycin 300 mg Capsules Clindamycin (as hydrochloride) Read all of this leaflet carefully before you start taking this medicine because it contains important
Dermovate Ointment clobetasol propionate
 Dermovate Ointment clobetasol propionate Package Leaflet: Information for the user Read all of this leaflet carefully before you start using this medicine because it contains important information for
Dermovate Ointment clobetasol propionate Package Leaflet: Information for the user Read all of this leaflet carefully before you start using this medicine because it contains important information for
PRODUCT INFORMATION DIFFERIN ADAPALENE 0.1% TOPICAL GEL
 PRODUCT INFORMATION DIFFERIN ADAPALENE 0.1% TOPICAL GEL NAME OF THE MEDICINE DIFFERIN Topical Gel: Adapalene 1 mg/g (0.1%) Common Name: Adapalene Chemical Name: 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic
PRODUCT INFORMATION DIFFERIN ADAPALENE 0.1% TOPICAL GEL NAME OF THE MEDICINE DIFFERIN Topical Gel: Adapalene 1 mg/g (0.1%) Common Name: Adapalene Chemical Name: 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic
ClobaDerm 500 micrograms/g Cream & Ointment (clobetasol propionate)
 ClobaDerm 500 micrograms/g Cream & Ointment (clobetasol propionate) PATIENT INFORMATION LEAFLET Read all of this leaflet carefully before you start using this medicine because it contains important information
ClobaDerm 500 micrograms/g Cream & Ointment (clobetasol propionate) PATIENT INFORMATION LEAFLET Read all of this leaflet carefully before you start using this medicine because it contains important information
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Herbal Essences Totally Twisted Curl Silkening Detangler (99926434) Company
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Herbal Essences Totally Twisted Curl Silkening Detangler (99926434) Company
SAFETY DATA SHEETS. This SDS packet was issued with item: N/A
 SAFETY DATA SHEETS This SDS packet was issued with item: 078866723 N/A 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Material Empirical Formula Synonyms Manufacturer Distributor
SAFETY DATA SHEETS This SDS packet was issued with item: 078866723 N/A 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Material Empirical Formula Synonyms Manufacturer Distributor
MENTAX -TC (butenafine HCl) Cream, 1% Rx Only DESCRIPTION CLINICAL PHARMACOLOGY. Package Insert
 Package Insert MENTAX -TC (butenafine HCl) Cream, 1% Rx Only DESCRIPTION MENTAX -TC Cream, 1%, contains the synthetic antifungal agent, butenafine hydrochloride. Butenafine is a member of the class of
Package Insert MENTAX -TC (butenafine HCl) Cream, 1% Rx Only DESCRIPTION MENTAX -TC Cream, 1%, contains the synthetic antifungal agent, butenafine hydrochloride. Butenafine is a member of the class of
Safety Data Sheet 1. IDENTIFICATION 2. HAZARDS IDENTIFICATION
 Safety Data Sheet 1. IDENTIFICATION Product Identifier QV Skin Lotion Other Means of Identification None Recommended Uses and Restrictions on Use Soothing, light moisturiser to help prevent and relieve
Safety Data Sheet 1. IDENTIFICATION Product Identifier QV Skin Lotion Other Means of Identification None Recommended Uses and Restrictions on Use Soothing, light moisturiser to help prevent and relieve
Item/Package Details Size Item Bottle ph Shelf Life 1.0 oz/29.6 ml 1101 Lucite Matte Silver Pump months
 B-Kleer 10 Lotion Use of mild to moderate acne. Gently exfoliates encrustation and acne pustules. Penetrates pores to eliminate most acne pimples. Reduces the severity of acne blemishes. Help prevent new
B-Kleer 10 Lotion Use of mild to moderate acne. Gently exfoliates encrustation and acne pustules. Penetrates pores to eliminate most acne pimples. Reduces the severity of acne blemishes. Help prevent new
Package leaflet: Information for the user. Tactuo 0.1% / 2.5% gel Adapalene/Benzoyl Peroxide
 Package leaflet: Information for the user Tactuo 0.1% / 2.5% gel Adapalene/Benzoyl Peroxide Read all of this leaflet carefully before you start using this medicine because it contains important information
Package leaflet: Information for the user Tactuo 0.1% / 2.5% gel Adapalene/Benzoyl Peroxide Read all of this leaflet carefully before you start using this medicine because it contains important information
ClobaDerm 0.05% w/w Cream & Ointment (clobetasol propionate)
 Package leaflet: Information for the patient ClobaDerm 0.05% w/w Cream & Ointment (clobetasol propionate) Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient ClobaDerm 0.05% w/w Cream & Ointment (clobetasol propionate) Read all of this leaflet carefully before you start using this medicine because it contains important
STAGES OF PHARMACEUTICAL MANUFACTURING
 STAGES OF PHARMACEUTICAL MANUFACTURING API Finished Product API Primary Packaging Secondary Packaging Excipients Starting Materials (Chemicals) 1 PHARMACEUTICAL MANUFACTURING OF NORMAL DOSAGE FORMS 2 Dosage
STAGES OF PHARMACEUTICAL MANUFACTURING API Finished Product API Primary Packaging Secondary Packaging Excipients Starting Materials (Chemicals) 1 PHARMACEUTICAL MANUFACTURING OF NORMAL DOSAGE FORMS 2 Dosage
Package leaflet: Information for the patient. Epiduo 0.3% / 2.5% gel adapalene / benzoyl peroxide
 Package leaflet: Information for the patient Epiduo 0.3% / 2.5% gel adapalene / benzoyl peroxide Read all of this leaflet carefully before you start using this medicine because it contains important information
Package leaflet: Information for the patient Epiduo 0.3% / 2.5% gel adapalene / benzoyl peroxide Read all of this leaflet carefully before you start using this medicine because it contains important information
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Cover Girl Eye Enhancers Eye Shadow (95815924, 95819198, 95841744,
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Cover Girl Eye Enhancers Eye Shadow (95815924, 95819198, 95841744,
What is in this leaflet 1. What Dalacin Cream is and what it is used for
 Package leaflet: Information for the patient PFIZER Dalacin Cream 2% Clindamycin phosphate Read all of this leaflet carefully before you start using this medicine because it contains important information
Package leaflet: Information for the patient PFIZER Dalacin Cream 2% Clindamycin phosphate Read all of this leaflet carefully before you start using this medicine because it contains important information
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Max Factor ColorGenius Face Powder (95887081, 95887099, 95884879, 95884826,
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Max Factor ColorGenius Face Powder (95887081, 95887099, 95884879, 95884826,
O N C E A DAY. In this leaflet FOR WOMEN
 FOR WOMEN O N C E A DAY Regaine for Women ONCE A DAY Scalp Foam Minoxidil 5% This medicine is used to treat female pattern hair loss in women aged 18 65 years. This product should only be used ONCE A DAY.
FOR WOMEN O N C E A DAY Regaine for Women ONCE A DAY Scalp Foam Minoxidil 5% This medicine is used to treat female pattern hair loss in women aged 18 65 years. This product should only be used ONCE A DAY.
MUPIROCIN OINTMENT 2% W/W TUBE New Zealand Data Sheet
 MUPIROCIN OINTMENT 1 PRODUCT NAME Medicianz Mupirocin Ointment Mupirocin 2% w/w ointment. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin 2% w/w equivalent to 20 mg/g For the full list of excipients,
MUPIROCIN OINTMENT 1 PRODUCT NAME Medicianz Mupirocin Ointment Mupirocin 2% w/w ointment. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mupirocin 2% w/w equivalent to 20 mg/g For the full list of excipients,
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Head and Shoulders Itchy Scalp Care Pyrithione Zinc Dandruff Conditioner
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Head and Shoulders Itchy Scalp Care Pyrithione Zinc Dandruff Conditioner
Creams and Lotions. Ointment/Special Creams. Soaps to be used With or Without Water
 Skin Care Creams and Lotions. Ointment/Special Creams. Soaps to be used With or Without Water 1 Skin Care Abena s skin care concept is based on our long experience within international health care sectors.
Skin Care Creams and Lotions. Ointment/Special Creams. Soaps to be used With or Without Water 1 Skin Care Abena s skin care concept is based on our long experience within international health care sectors.
Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None. Emergency telephone number: CHEMTREC
 1. Product and Company Identification Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None Recommended Use: Uses Advised Against: No information available Supplier information: Garcoa
1. Product and Company Identification Product Name: Acne Medication Benzoyl Peroxide 10% Lotion Synonyms: None Recommended Use: Uses Advised Against: No information available Supplier information: Garcoa
MATERIAL SAFETY DATA SHEET Finished Product
 MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Olay Regenerist UV Defense Regenerating Lotion (95411526) Company Identification:
MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Olay Regenerist UV Defense Regenerating Lotion (95411526) Company Identification:
SAFETY DATA SHEET Clean Choice Antibacterial Foam Soap
 SAFETY DATA SHEET 1. Identification Product identifier Product name Product number 0617376 Internal identification 58211 / 0001 Recommended use of the chemical and restrictions on use Application Antibacterial
SAFETY DATA SHEET 1. Identification Product identifier Product name Product number 0617376 Internal identification 58211 / 0001 Recommended use of the chemical and restrictions on use Application Antibacterial
PRODUCT INFORMATION DALACIN T TOPICAL LOTION NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. DALACIN T clindamycin 1% (10 mg/ml) (as phosphate) lotion.
 PRODUCT INFORMATION DALACIN T TOPICAL LOTION NAME OF TE MEDICINE DALACIN T clindamycin 1% (10 mg/ml) (as phosphate) lotion. The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-
PRODUCT INFORMATION DALACIN T TOPICAL LOTION NAME OF TE MEDICINE DALACIN T clindamycin 1% (10 mg/ml) (as phosphate) lotion. The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-
Dermovate Scalp Application clobetasol propionate
 Dermovate Scalp Application clobetasol propionate Package Leaflet: Information for the user Read all of this leaflet carefully before you start using this medicine because it contains important information
Dermovate Scalp Application clobetasol propionate Package Leaflet: Information for the user Read all of this leaflet carefully before you start using this medicine because it contains important information
Betnovate Scalp Application betamethasone valerate. 1 What Betnovate is and what it is used for. 2 What you need to know before you use Betnovate
 Package Leaflet: Information for the User Betnovate Scalp Application betamethasone valerate Read all of this leaflet carefully before you start using this medicine because it contains important information
Package Leaflet: Information for the User Betnovate Scalp Application betamethasone valerate Read all of this leaflet carefully before you start using this medicine because it contains important information
Dermovate Ointment clobetasol propionate
 Dermovate Ointment clobetasol propionate Package Leaflet: Information for the User Read all of this leaflet carefully before you start using this medicine because it contains important information for
Dermovate Ointment clobetasol propionate Package Leaflet: Information for the User Read all of this leaflet carefully before you start using this medicine because it contains important information for
Complete Dermal Integration. Proven Duration.
 Complete Dermal Integration. Proven Duration. Introducing BELOTERO BALANCE Dermal Filler. BELOTERO BALANCE Dermal Filler is uniquely manufactured with CPM Technology to give you precision to treat a wide
Complete Dermal Integration. Proven Duration. Introducing BELOTERO BALANCE Dermal Filler. BELOTERO BALANCE Dermal Filler is uniquely manufactured with CPM Technology to give you precision to treat a wide
scalp solution 5% w/v minoxidil cutaneous solution to be applied to the scalp
 scalp solution 5% w/v minoxidil cutaneous solution to be applied to the scalp This medicine is used to treat hereditary hair loss in men aged 18 65 years. This product is for men only and should NOT be
scalp solution 5% w/v minoxidil cutaneous solution to be applied to the scalp This medicine is used to treat hereditary hair loss in men aged 18 65 years. This product is for men only and should NOT be
NEWS RELEASE. CONTACTS: Investors: Lisa DeFrancesco (862) Media: Mark Marmur (862) Ember Garrett (714)
 NEWS RELEASE CONTACTS: Investors: Lisa DeFrancesco (862) 261-7152 Media: Mark Marmur (862) 261-7558 Ember Garrett (714) 246-3525 JUVÉDERM VOLBELLA XC APPROVED BY U.S. FDA FOR USE IN LIPS AND PERIORAL RHYTIDS
NEWS RELEASE CONTACTS: Investors: Lisa DeFrancesco (862) 261-7152 Media: Mark Marmur (862) 261-7558 Ember Garrett (714) 246-3525 JUVÉDERM VOLBELLA XC APPROVED BY U.S. FDA FOR USE IN LIPS AND PERIORAL RHYTIDS
MATERIAL SAFETY DATA SHEET Finished Product
 MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Nioxin Scalp Recovery Moisturizing Conditioner (99428038) Company Identification:
MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Nioxin Scalp Recovery Moisturizing Conditioner (99428038) Company Identification:
MATERIAL SAFETY DATA SHEET Finished Product
 MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Olay Total Effects Night Firming Cream (95043213) Company Identification:
MATERIAL SAFETY DATA SHEET Finished Product SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Olay Total Effects Night Firming Cream (95043213) Company Identification:
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Cover Girl Sensitive Skin Pressed Powder (98917087, 98917088, 98917089,
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Cover Girl Sensitive Skin Pressed Powder (98917087, 98917088, 98917089,
MATERIAL SAFETY DATA SHEET Finished Product.
 MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Aussie Hair Insurance Leave-in Conditioning Spray (95260860) Company
MATERIAL SAFETY DATA SHEET Finished Product. SECTION 1 - CHEMICAL SUBSTANCE/PRODUCT AND COMPANY IDENTIFICATION Finished Product Name: Aussie Hair Insurance Leave-in Conditioning Spray (95260860) Company
Package leaflet: Information for the user. Versatis 700 mg medicated plaster Lidocaine
 Package leaflet: Information for the user Versatis 700 mg medicated plaster Lidocaine Read all of this leaflet carefully before you start using this medicine because it contains important information for
Package leaflet: Information for the user Versatis 700 mg medicated plaster Lidocaine Read all of this leaflet carefully before you start using this medicine because it contains important information for
COPPERPEX GEL. The product for lips with herpes TOPICAL USE. Medical Device
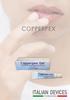 COPPERPEX GEL The product for lips with herpes TOPICAL USE Medical Device COPPERPEX GEL is an innovative gel for lips, studied for early treatment of acute and chronic herpes labials manifestations. The
COPPERPEX GEL The product for lips with herpes TOPICAL USE Medical Device COPPERPEX GEL is an innovative gel for lips, studied for early treatment of acute and chronic herpes labials manifestations. The
