COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860. Effective Date: August 31, 2006
|
|
|
- Osborn Chapman
- 5 years ago
- Views:
Transcription
1 COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated 6/1/05 1. PURPOSE: To provide guidelines for the prevention of cross contamination and infection of employees and individuals served by the proper collection, handling, storage, and transportation of biohazardous wastes. 2. POLICY: 1. All biohazardous (infectious) wastes shall be collected, handled, stored, transported, and disposed of separately from all other solid waste and only as described in the procedure outlined below. 2. The entire system for dealing with biohazardous (infectious) wastes shall be approved periodically in writing by the Fresno County Department of Environmental Health Service and shall not be revised in any way without their approval and the approval of the Coalinga State Hospital Infection Control Committee. 3. Non-Biohazardous (non-infectious) waste should not be placed in the "Biohazardous (Infectious)" Waste receptacles. 4. Unit nursing personnel are responsible for the collection and, in most areas, the disposal of infectious waste. 5. The fluid blood and moist body substance of all individuals served, staff, and visitors shall be treated as though they were contagious. 6. Individuals served are never to handle any contaminated materials. -1-
2 3. DEFINITION OF WASTE: -Significant laboratory wastes including microbiologic specimens (including cultures) from Individual, and disposable fomites; discarded live or attenuated vaccine or containers. -Human tissues and disposable materials from medical/dental clinics and unit examinations and treatment rooms suspected of being contaminated with infectious agents contagious to humans. -Waste which contains recognizable fluid blood or blood products (free, on equipment, or in containers) -Waste containing discarded materials contaminated with excretion, exudate, or secretions of individuals served placed in isolation room to protect others from highly communicable diseases. -Coalinga State Hospital also treats as Biohazardus Waste the following categories that are listed by CAL-OSHA as Regulated Waste. Regulated waste is defined as: contaminated items that would release blood or other potentially infectious materials ( OPIM ) if compressed, or that are caked with dried blood or OPIM, or that contains liquid OPIM. OPIM is defined to include: semen (except for discarded condoms), vaginal secretions (except for discarded peri pads or tampons), saliva from dental procedures, and fluid from internal body cavities. DOES NOT INCLUDE: -Paper towels, paper products, urine, feces, saliva (except from dental procedures) sputum, nasal secretions, sweat, tears, or vomits unless any of the preceding contain fluid blood. 3. PRECAUTIONS: 1. In all reasonably anticipated exposures to blood or other potentially infectious material personal protective equipment and engineering controls shall be used. Engineering controls isolate or remove the blood borne pathogen hazard from the work place (i.e. sharps containers). Personal protective equipment is special clothing or equipment worn by an employee for protection against a hazard. General work clothes (e.g., uniforms, pants, shirts, or blouses) not intended to function as protection against a hazard are not considered to be personal protective equipment. 2. It is the responsibility of all nursing personnel who handle "Biohazardous Waste" to ensure that it is not mixed with "non-infectious waste." -2-
3 3. If "Biohazardous Waste" is accidentally placed in a non-infectious container, the entire contents of the container must then be considered contaminated. It must be immediately bagged and disposed of as "Biohazardous Waste." The container from which it was taken shall be cleansed with detergent-germicide solution. If noninfectious waste is accidentally placed in the "Biohazardous Waste Container" it is considered contaminated and may not be removed. 4. IMPLEMENTATION AND INTERVENTION: PLACING INFECTIOUS WASTE IN RECEPTACLE: NURSING ACTION A. Biohazardous waste must be placed in the special receptacles marked Biohazardous Waste Only or Biohazardous with symbol on the cover and on visible lateral sides. B. These receptacles should be considered contaminated on all surfaces and care taken not to touch them except as necessary and with proper hand hygiene after each exposure. C. Never place biohazardous waste in a biohazardous receptacle which does not have the appropriate lettering and symbols D. All biohazardous waste receptacles will have a foot pedal to open the lid. When the lid mechanism is not working properly, the receptacle should be replaced. E. The lids of the biohazardous waste receptacles must be closed except when briefly opened for placement of biohazardous material inside. No container without a lid should be used for infectious waste. KEY POINTS A. Infectious waste must not be mixed with non-infectious waste. B. All waste should be considered hazardous but known biohazardous waste requires special precautions. C. All receptacles should have a transport container inside. An intact can liner prevents contamination of the receptacle. Placing contaminated waste directly in receptacle grossly contaminates it and it will require special cleaning with detergent germicidal solution immediately. D. Proper procedures require functioning equipment. E. The closed lid prevents airborne contamination and helps control odors. -3-
4 F. Bio-hazardous waste should always be gently and carefully placed in the receptacle. F. Avoid all forceful agitation of infectious waste. Biohazardous waste can be life threatening. G. Care should be taken to place the waste within the plastic bag can liner without contamination of the lid or outer surface(s) of the receptacle. H. Do not overfill the biohazardous waste receptacles! I. Broken glass fragments and other sharp infectious waste items should be contained in a sharps disposal container. H. Full means 2/3 full. Failure to follow this procedure will result in serious contamination of the environment. I. The sharps container must be delivered to the biohazardous waste barrel be the next day if blood or tissue is placed in it. EMPTYING WASTE RECEPTACLES: NURSING ACTION A. Get one (1) red plastic bag labeled Biohazardous Waste with a biohazard symbol. B. Care must be taken not to spill any of the biohazardous waste at any time. C. Open the biohazardous waste receptacle, place hands under the cuff, and gently close the bag and tie. Lift transport container (with tied red bag inside) from the waste can and put transport container lid in place. D. Place a new transport container into the biohazardous waste receptacle. -4- KEY POINTS A. Double bagging is no longer required. Transport containers are used. B. If a spill should occur, clean up immediately. Wearing gloves, apply a solution of Powder Keg or other approved disinfectant to contaminated surface(s). Clean-up Kit should be used before wiping up liquid blood spills. C. Do not compress bag prior to tying. D. The rigid container with tight fitting cover prevents accidental injury and spills, which may result in contamination of the environment.
5 E. Place the clean red bag marked with the biohazard symbol and labeled Biohazardous Waste in the transport container. Carefully fold a cuff down around the top outside of the transport container. F. If the large drum is full, return to unit with biohazardous waste in transport container and notify Housekeeping or Public Health office. E. Handle only the inside surfaces of the new can liner. F. If after hours, leave word on the voic system. TYPES OF CONTAINERS, WHERE OBTAINED AND WHERE DISPOSED OF: MATERIAL TO BE BAGGED Infectious Linen Contaminated Laundry (Double Bagged) WASTE (Does Not Include Infectious Sharps UNLESS IN SHARPS CONTAINER) Non-disposable Infectious Items (i.e. instruments, etc.) TYPE OF BAG OR CONTAINER Clear, Water Soluble Bag Yellow Plastic (outer) Bag A SINGLE, RED PLASTIC BAG MARKED WASTE Clean with soap and water on the unit and place in a puncture-resistant, plastic contained labeled WHERE OBTAINED Laundry Main Warehouse Central Supply WHERE DISPOSED OF Soiled Linen Room Place in a WASTE Barrel Return to Central Supply -5-
Deadly Bloodborne Diseases
 What and Why This Refresher Blood Borne Pathogens on-line training is offered for all returning employees of Harnett County Schools who have previously completed the Initial BBP training video. This is
What and Why This Refresher Blood Borne Pathogens on-line training is offered for all returning employees of Harnett County Schools who have previously completed the Initial BBP training video. This is
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College
 Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
Regulated Medical Waste. Be sure to sign in!
 Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Bloodborne Pathogens Exposure Control Plan. December 2003
 Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Infection Control 101
 Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
REQUEST FOR QUOTE. Community Initiatives Bureau. Biohazardous Cleaning Service
 REQUEST FOR QUOTE Community Initiatives Bureau Biohazardous Cleaning Service December 7, 2017 OVERVIEW The Boston Public Health Commission (BPHC) protects the health protects the health of Bostonians and
REQUEST FOR QUOTE Community Initiatives Bureau Biohazardous Cleaning Service December 7, 2017 OVERVIEW The Boston Public Health Commission (BPHC) protects the health protects the health of Bostonians and
Biological Safety Training
 Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
Bloodborne Pathogens
 Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
Bloodborne Pathogens: Exposure In The Workplace Employee Handbook
 Bloodborne Pathogens: Exposure In The Workplace Employee Handbook Introduction There s a danger in the workplace that s not even visible to the naked eye, yet it could change your life forever if you re
Bloodborne Pathogens: Exposure In The Workplace Employee Handbook Introduction There s a danger in the workplace that s not even visible to the naked eye, yet it could change your life forever if you re
Annual Associate Safety Module. Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan
 Annual Associate Safety Module Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan Since you work in a healthcare facility, you may have potential exposure to blood or body fluids.
Annual Associate Safety Module Blood & Body Fluids: How To Prevent Exposure Your Exposure Control Plan Since you work in a healthcare facility, you may have potential exposure to blood or body fluids.
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS
 Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
Michigan State University Athletic Training Students BLOOD BORNE PATHOGENS AND UNIVERSAL PRECAUTIONS The following principles must be applied when employees are potentially exposed to bloodborne pathogens:
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN
 FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
Appendix C. Infectious Waste Guidelines
 Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
INFECTION PREVENTION AND CONTROL PLAN (IPCP)
 INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
What is infection control?
 Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions
 Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions 83.100 Health & Safety Definitions Clarity for licensee on health, safety & sanitation responsibilities
Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions 83.100 Health & Safety Definitions Clarity for licensee on health, safety & sanitation responsibilities
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT
 UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
UNIVERSITY OF NORTH FLORIDA BIOMEDICAL WASTE MANAGEMENT PLAN DEVELOPED BY: ENVIRONMENTAL HEALTH, SAFETY, INSURANCE & RISK MANAGEMENT September 2010 Table of Contents Section Page Background 1 Definitions
Provide a brief description of the procedure and infectious organisms used:
 Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS
 SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
Bloodborne Pathogens Exposure Control Plan
 Bloodborne Pathogens Exposure Control Plan Environmental Health, Safety, and Risk Management Department Box 6113, SFA Station Nacogdoches, Texas 75962-6113 January 2011 Revised May 2017 APPLICABILITY These
Bloodborne Pathogens Exposure Control Plan Environmental Health, Safety, and Risk Management Department Box 6113, SFA Station Nacogdoches, Texas 75962-6113 January 2011 Revised May 2017 APPLICABILITY These
Disposal of Biological Waste
 Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
Disposal of Biological Waste Biological Waste Disposal / Supplies Biological Waste Boxes Available in designated areas of research buildings (consult EH&S, Department administrator, other researchers)
Sterilization A Training Module
 Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
BSL-2 Emergency Plan
 BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
x. ANNUAL REVIEW SIGNATURE SHEET
 x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
x. ANNUAL REVIEW SIGNATURE SHEET PROCEDURE TITLE: UNIVERSAL PRECAUTIONS Signature on this page insures that each procedure has been reviewed annually. Any changes will be reflected on the procedure by
CCS Administrative Procedure T Biosafety for Laboratory Settings
 CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
CCS Administrative Procedure 2.30.05-T Biosafety for Laboratory Settings Implementing Board Policy 2.30.05 Contact: College Biosafety Hygiene Officers, (phone # to be determined) 1.0 Purpose Community
UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach
 Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
Procedure #: UNIVERSITY OF SOUTHERN MAINE Office of Research Integrity & Outreach IBC-001 Date Adopted: October 10, 2017 Last Updated: Prepared By: Casey Webster, Research Compliance Administrator Reviewed
RULES OF TENNESSEE BOARD OF COSMETOLOGY AND BARBER EXAMINERS CHAPTER SANITARY REQUIREMENTS TABLE OF CONTENTS
 RULES OF TENNESSEE BOARD OF COSMETOLOGY AND BARBER EXAMINERS CHAPTER 0200-03 SANITARY REQUIREMENTS TABLE OF CONTENTS 0200-03-.01 Applicability 0200-03-.02 Violations 0200-03-.03 Location 0200-03-.04 Communicable
RULES OF TENNESSEE BOARD OF COSMETOLOGY AND BARBER EXAMINERS CHAPTER 0200-03 SANITARY REQUIREMENTS TABLE OF CONTENTS 0200-03-.01 Applicability 0200-03-.02 Violations 0200-03-.03 Location 0200-03-.04 Communicable
ECU Radiation, Biosafety and Hazardous Substances Committee
 Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
The most up-to-date version of this policy can be viewed at the following website:
 Page 1 of 9 Review SOP Objective To ensure that patients with head lice are cared for appropriately and actions are taken to minimise the risk of cross-infection. This SOP applies to all staff employed
Page 1 of 9 Review SOP Objective To ensure that patients with head lice are cared for appropriately and actions are taken to minimise the risk of cross-infection. This SOP applies to all staff employed
OSHA: Occupational Safety and Health Administration PPE Personal protective equipment
 Bloodborne Pathogens University of Tennessee Safety Program HM-010 Document Contact: EHS Date effective: March 15, 2011 Revision Date: October 2, 2017 Purpose The purpose of this written program is to
Bloodborne Pathogens University of Tennessee Safety Program HM-010 Document Contact: EHS Date effective: March 15, 2011 Revision Date: October 2, 2017 Purpose The purpose of this written program is to
List any references used for the procedure design (research publications, etc.):
 Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Safety Rules for Laboratory
 Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Medical Waste Management Plan
 Medical Waste Management Plan Safety Services - Biosafety University of California, Davis Version 1.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website:
Medical Waste Management Plan Safety Services - Biosafety University of California, Davis Version 1.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website:
HEAD LICE. The most up-to-date version of this policy can be viewed at the following website:
 Page Page 1 of 9 Policy Objective To ensure that patients with head lice are cared for appropriately and actions are taken to minimise the risk of cross-infection. This policy applies to all staff employed
Page Page 1 of 9 Policy Objective To ensure that patients with head lice are cared for appropriately and actions are taken to minimise the risk of cross-infection. This policy applies to all staff employed
Disposal of Biohazard Wastes
 4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
4.24.1 POLICY Exceptions Radioactive Materials Administrators and principal investigators (PIs) are responsible for ensuring that biohazard wastes generated by University units are collected and disposed
A ppendix 15 WUStL Bloodborne Pathogens Exposure Control Plan Research Laboratory-Specific Work Practices
 Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee
 Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers
 Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Laboratory Orientation. Biological Screening
 Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
MEDICAL WASTE MANAGEMENT PLAN
 MEDICAL WASTE MANAGEMENT PLAN University of California, Davis Version 2.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website: safetyservices.ucdavis.edu
MEDICAL WASTE MANAGEMENT PLAN University of California, Davis Version 2.0 Main Office: 276 Hoagland Hall, Davis, CA, 95616 Phone: (530) 752-1493 Fax: (530) 752-4527 Website: safetyservices.ucdavis.edu
KERN HEALTH SYSTEMS POLICIES AND PROCEDURES 2.21-P
 Page 1 of5 RESPONSIBLE DEPARTMENT HEAD: Director of Quality Improvement, Health Education and Disease Management Review Date 08/29/97 08/00 01102 08/2005 OS/2010 Effective Date 09/01103 11118/05 DS/;ZOhtJ
Page 1 of5 RESPONSIBLE DEPARTMENT HEAD: Director of Quality Improvement, Health Education and Disease Management Review Date 08/29/97 08/00 01102 08/2005 OS/2010 Effective Date 09/01103 11118/05 DS/;ZOhtJ
Original Date:
 Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
PUBLIC HEALTH DEPARTMENT
 ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
General Lab Safety Rules and Practices SOP-GLSRP-01
 Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
Standard Operating Procedure General Lab Safety Rules and Practices SOP Number: SOP-GLSRP-01 Category: Lab Process Supersedes: N/A Effective Date: December 1, 2017 Pages 5 Subject: General Lab Safety Rules
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET
 TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
Bloodborne Pathogens Exposure Control Plan
 Bloodborne Pathogens Exposure Control Plan Document History Version Date Comments 0.2 January, 2018 Program Review Foreword This written program is site specific to UVa Facilities Management and is in
Bloodborne Pathogens Exposure Control Plan Document History Version Date Comments 0.2 January, 2018 Program Review Foreword This written program is site specific to UVa Facilities Management and is in
San Bernardino Valley College. Blood Borne Pathogens. Exposure Control Program
 San Bernardino Valley College Blood Borne Pathogens Exposure Control Program December 7, 2009 I. PURPOSE The Blood Borne Pathogens Exposure Control Program (BBP) has been developed by San Bernardino Valley
San Bernardino Valley College Blood Borne Pathogens Exposure Control Program December 7, 2009 I. PURPOSE The Blood Borne Pathogens Exposure Control Program (BBP) has been developed by San Bernardino Valley
INFECTION PREVENTION AND CONTROL PLAN
 INFECTION PREVENTION AND CONTROL PLAN FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: ( ) The owner, employees and practitioners of the above body art facility have developed
INFECTION PREVENTION AND CONTROL PLAN FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: ( ) The owner, employees and practitioners of the above body art facility have developed
Roosevelt Biosafety Training. Created 10/2015
 Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Safety Office -- Laboratory Inspection Form
 RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
The following standard practices, safety equipment, and facility requirements apply to BSL-1:
 Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
Standard Microbiological Practices for Biosafety Level 1 Laboratories at the University of Tennessee-Knoxville, Institute of Agriculture and Graduate School of Medicine Overview and Definitions Standard
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION
 State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
Package Leaks. OH&S Biosafety Emergency Response Document. Examine outer packaging. Leaks or evidence of leaks. No evidence of leaks
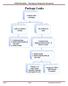 Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
STANDARD: Laboratory Safety Effective: March 20, 2018
 University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
University of North Dakota Department of Medical Laboratory Science Grand Forks, ND STANDARD: Laboratory Safety Effective: March 20, 2018 PURPOSE This standard establishes general safe practices in the
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # Approved: 10/3/18
 Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Bloodborne Pathogens Safety in Your Workplace
 Bloodborne Pathogens Safety in Your Workplace COPYRIGHT NOTICE Copyright 2017 by Judy Adams. ALL RIGHTS RESERVED No part of this publication may be copied or distributed, transmitted, transcribed, stored
Bloodborne Pathogens Safety in Your Workplace COPYRIGHT NOTICE Copyright 2017 by Judy Adams. ALL RIGHTS RESERVED No part of this publication may be copied or distributed, transmitted, transcribed, stored
Biohazardous Waste. 1. Solid Biohazardous Waste (non-sharps) Storage
 Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Standard Operating Procedure for Biosafety Cabinet Use
 NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
METHODS OF IMPLEMENTATION AND CONTROL
 Universal Precautions: METHODS OF IMPLEMENTATION AND CONTROL All employees will utilize universal precautions (MIOSHA Rule 325.70005) Rule 5. Universal precautions shall be observed to prevent contact
Universal Precautions: METHODS OF IMPLEMENTATION AND CONTROL All employees will utilize universal precautions (MIOSHA Rule 325.70005) Rule 5. Universal precautions shall be observed to prevent contact
Brazosport College Life Science Laboratory Safety Rules and Regulations
 Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
Brazosport College Life Science Laboratory Safety Rules and Regulations Laboratory Safety Procedures for Biology Labs Permanent Link: http://bit.ly/bc-labsafety The risks incurred in the biology laboratories
University Of Florida. Bloodborne Pathogen Program. Standard Operating Procedures
 University Of Florida Bloodborne Pathogen Program Standard Operating Procedures Revised February 9, 2011 Updated (annually) BBP Standard Operating Procedures Page 1 of 13 University Of Florida Bloodborne
University Of Florida Bloodborne Pathogen Program Standard Operating Procedures Revised February 9, 2011 Updated (annually) BBP Standard Operating Procedures Page 1 of 13 University Of Florida Bloodborne
BLOODBORNE PATHOGENS
 MARICOPA COUNTY SHERIFF S OFFICE POLICY AND PROCEDURES Subject Related Information CRITICAL POLICY PURPOSE BLOODBORNE PATHOGENS Supersedes CP-6 (08-14-15) Policy Number CP-6 Effective Date 11-22-16 The
MARICOPA COUNTY SHERIFF S OFFICE POLICY AND PROCEDURES Subject Related Information CRITICAL POLICY PURPOSE BLOODBORNE PATHOGENS Supersedes CP-6 (08-14-15) Policy Number CP-6 Effective Date 11-22-16 The
Infection Prevention Guidelines. Safe Use, Handling & Disposal of Sharps
 Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
CLEANING, SANITIZING, AND DISINFECTING
 CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
BIOLOGICAL SAFETY MANUAL
 BIOLOGICAL SAFETY MANUAL April, 2017 University of Northern Colorado Biological Safety Manual I. Introduction II. Definitions Human Blood and Other Potentially Infectious Materials Infectious Agents and
BIOLOGICAL SAFETY MANUAL April, 2017 University of Northern Colorado Biological Safety Manual I. Introduction II. Definitions Human Blood and Other Potentially Infectious Materials Infectious Agents and
Cleaning and Disinfection Protocol for Emergency Services Fire, Ambulance, Police, Search & Rescue
 This document has been developed in accordance with current applicable infection control and regulatory guidelines. It is intended for use as a guideline only. At no time should this document replace existing
This document has been developed in accordance with current applicable infection control and regulatory guidelines. It is intended for use as a guideline only. At no time should this document replace existing
Standard Operating Procedures
 Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
Standard Operating Procedures (V1_4/7/16) Safe Working Practices for Leica Laser Micro Dissection Microscope Table of Contents I. General Information II. Facility Orientation and Training III. Startup
Table 6: Detailed Infection Prevention and Control Procedures for Tattooing and Micropigmentation. Use During Tattooing
 FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
Emergency Procedures Specific Biological Spill Clean-Up Guidelines
 Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL
 WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL Part of White Woods Academy Trust BODILY FLUID HYGIENE POLICY Approved by Governors: September 2017 Review Date: September 2019 Statement of intent At Whiston
WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL Part of White Woods Academy Trust BODILY FLUID HYGIENE POLICY Approved by Governors: September 2017 Review Date: September 2019 Statement of intent At Whiston
TARLETON STATE UNIVERSITY Biohazardous Waste Program
 TARLETON STATE UNIVERSITY Biohazardous Waste Program Office of Risk Management and Safety June 2012 1. GENERAL The following information is provided to assist in developing requirements, guidelines and
TARLETON STATE UNIVERSITY Biohazardous Waste Program Office of Risk Management and Safety June 2012 1. GENERAL The following information is provided to assist in developing requirements, guidelines and
The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy
 The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy This policy applies to ALL sharps injuries where any hazardous substance (including, toxins, chemicals and human pathogens)
The Management of Inoculation (Sharps) Injury or Blood Borne Pathogen Exposure Policy This policy applies to ALL sharps injuries where any hazardous substance (including, toxins, chemicals and human pathogens)
Hand Hygiene ORGANIZATIONAL: Affects two or more departments.
 Hand Hygiene ORGANIZATIONAL: Affects two or more departments. Folder Infection Prevention Sub-Folder Original 1/1/1987 Scope All Effective Date Approved (Approver/Date) Last Reviewed/ Revised Date IPC:
Hand Hygiene ORGANIZATIONAL: Affects two or more departments. Folder Infection Prevention Sub-Folder Original 1/1/1987 Scope All Effective Date Approved (Approver/Date) Last Reviewed/ Revised Date IPC:
BIOLOGICAL SAFETY INSPECTION CHECKLIST
 BIOLOGICAL SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
BIOLOGICAL SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
Scabies Identification, Treatment and Environmental Cleaning
 Scabies Identification, Treatment and Environmental Cleaning Level III Purpose The purpose of this procedure is to treat residents infected with and sensitized to Sarcoptes scabiei and to prevent the spread
Scabies Identification, Treatment and Environmental Cleaning Level III Purpose The purpose of this procedure is to treat residents infected with and sensitized to Sarcoptes scabiei and to prevent the spread
ILLINOIS STATE UNIVERSITY BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN
 ILLINOIS STATE UNIVERSITY BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN Revised 11/10/2017 Table of Contents 1. PURPOSE AND SCOPE... 2 2. RESPONSIBILITIES... 2 a. ENVIRONMENTAL HEALTH AND SAFETY... 2 b. STUDENT
ILLINOIS STATE UNIVERSITY BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN Revised 11/10/2017 Table of Contents 1. PURPOSE AND SCOPE... 2 2. RESPONSIBILITIES... 2 a. ENVIRONMENTAL HEALTH AND SAFETY... 2 b. STUDENT
TARLETON STATE UNIVERSITY Biohazardous Waste Program
 TARLETON STATE UNIVERSITY Biohazardous Waste Program Program Name: Biohazardous Waste Department Name: TSU Risk Management & Compliance Doc. No.: BIOS-04-L2-S0-CH0-001 Rev. No.: 2 Concurrence and Approval
TARLETON STATE UNIVERSITY Biohazardous Waste Program Program Name: Biohazardous Waste Department Name: TSU Risk Management & Compliance Doc. No.: BIOS-04-L2-S0-CH0-001 Rev. No.: 2 Concurrence and Approval
CHAPTER 13 PROCEDURE checklists
 PROCEDURE checklists 179 CHAPTER 13 PROCEDURE checklists Procedure 13-1 Handwashing 1. Gather needed supplies if not present at the handwashing area: soap or the cleansing agent specified by your facility,
PROCEDURE checklists 179 CHAPTER 13 PROCEDURE checklists Procedure 13-1 Handwashing 1. Gather needed supplies if not present at the handwashing area: soap or the cleansing agent specified by your facility,
BSL2 Exposure Control Plan: Human or Non Human Primate Materials
 Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Prepared/Revised by Tamara Casebolt, Ph.D Date 6/7/2017 Reviewed by Carolyn Keierleber, Ph.D Date 09/20/2017 A. Hazards Human blood or other primate cells and tissue have the potential to harbor infectious
Biohazard Waste Management Plan
 WAKE FOREST UNIVERSITY Biohazard Waste Management Plan Reynolda Campus WFU 1/9/2017 Questions or concerns regarding this plan should be directed to the Department of Environmental Health and Safety at
WAKE FOREST UNIVERSITY Biohazard Waste Management Plan Reynolda Campus WFU 1/9/2017 Questions or concerns regarding this plan should be directed to the Department of Environmental Health and Safety at
Biosafety Self-Audit Checklist
 Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Biosafety Self-Audit Checklist Principal Investigator: Biosafety Certificate #: Location: Audited By: Date: Posting: Dalhousie University Hazard Identification poster with biohazard symbol posted on lab
Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5
 Environmental Health and Safety Manual Policy Number: EH&S 4-4 Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5 PURPOSE: To establish safe handling practices and use of
Environmental Health and Safety Manual Policy Number: EH&S 4-4 Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5 PURPOSE: To establish safe handling practices and use of
Microdot Bleach Wipe
 EPA Reg. No. 69687-1-88459 EPA Est. No. 69687-IL-001 Cambridge Sensors USA, LLC 10051 Bode Rd Plainfield, IL 60585 PHONE: 630.428.4240 FAX: (630) 428 4255 www.microdotcs.com MATERIAL SAFETY DATA SHEET
EPA Reg. No. 69687-1-88459 EPA Est. No. 69687-IL-001 Cambridge Sensors USA, LLC 10051 Bode Rd Plainfield, IL 60585 PHONE: 630.428.4240 FAX: (630) 428 4255 www.microdotcs.com MATERIAL SAFETY DATA SHEET
Safe Handling and Disposal of Sharps. Reference Guide
 Safe Handling and Disposal of Sharps Reference Guide Safe Handling and Disposal of Syringes and other Sharps All staff involved in the administration of a drug or other substance should be trained in the
Safe Handling and Disposal of Sharps Reference Guide Safe Handling and Disposal of Syringes and other Sharps All staff involved in the administration of a drug or other substance should be trained in the
SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN
 NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information
NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information
Safe Handling and Disposal of Sharps
 Statement of Intent Safe Handling and Disposal of Sharps To provide clear guidelines for the safe handling and disposal of all sharps in order that the risk of inoculation injury and transmission of blood
Statement of Intent Safe Handling and Disposal of Sharps To provide clear guidelines for the safe handling and disposal of all sharps in order that the risk of inoculation injury and transmission of blood
Safe Sharps Disposal. Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program
 Safe Sharps Disposal Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program How to Safely Dispose of Household Sharps Millions of people use
Safe Sharps Disposal Learn how to safely dispose of used sharps including needles, lancets and syringes. Expanded Syringe Access Program How to Safely Dispose of Household Sharps Millions of people use
rooo.lb IOWA COUNTY ORDINANCE NO TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS:
 .. rooo.lb IOWA COUNTY ORDINANCE NO. 4-196 TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS: SECTION I: The following ordinance of Iowa County, Wisconsin is hereby created
.. rooo.lb IOWA COUNTY ORDINANCE NO. 4-196 TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS: SECTION I: The following ordinance of Iowa County, Wisconsin is hereby created
New Mexico Institute of Mining & Technology. Hazard Communication Policy
 New Mexico Institute of Mining & Technology Hazard Communication Policy BASIS: The OSHA Hazard Communication Standard establishes uniform requirements to ensure that the hazards of all chemicals used at
New Mexico Institute of Mining & Technology Hazard Communication Policy BASIS: The OSHA Hazard Communication Standard establishes uniform requirements to ensure that the hazards of all chemicals used at
Nova Scotia Safe Body Art Standards
 Nova Scotia Safe Body Art Standards Crown copyright, Province of Nova Scotia, 2018 Nova Scotia Safe Body Art Standards Environment, December 2018. ISBN 978-1-55457-905-1 Standard: Nova Scotia Safe Body
Nova Scotia Safe Body Art Standards Crown copyright, Province of Nova Scotia, 2018 Nova Scotia Safe Body Art Standards Environment, December 2018. ISBN 978-1-55457-905-1 Standard: Nova Scotia Safe Body
Hand Hygiene. Policy Title: Hand Hygiene Policy Number: 05. Effective Date: 6/10/2013 Review Date: 6/10/2016
 Hand Hygiene 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in hand hygiene. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings. 2.2. To promote awareness for
Hand Hygiene 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in hand hygiene. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings. 2.2. To promote awareness for
Ventricular Assist Device (VAD) Exit Site Care Guidelines
 Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
SUBCHAPTER 14H - SANITATION SECTION SANITATION
 SUBCHAPTER 14H - SANITATION SECTION.0100 - SANITATION 21 NCAC 14H.0101 COPY OF RULES TO COSMETOLOGY STUDENTS Cosmetic art schools shall give a copy of the sanitation rules governing the practice of the
SUBCHAPTER 14H - SANITATION SECTION.0100 - SANITATION 21 NCAC 14H.0101 COPY OF RULES TO COSMETOLOGY STUDENTS Cosmetic art schools shall give a copy of the sanitation rules governing the practice of the
Safe Handling and Disposal of Sharps
 SBC Children, Families And Community Health Service Statement of Intent Safe Handling and Disposal of Sharps To provide clear guidelines for the safe handling and disposal of all sharps in order that the
SBC Children, Families And Community Health Service Statement of Intent Safe Handling and Disposal of Sharps To provide clear guidelines for the safe handling and disposal of all sharps in order that the
RULES OF TENNESSEE STATE BOARD OF COSMETOLOGY CHAPTER SANITARY RULES TABLE OF CONTENTS
 RULES OF TENNESSEE STATE BOARD OF COSMETOLOGY CHAPTER 0440-2 SANITARY RULES TABLE OF CONTENTS 0440-2-.01 Definitions 0440-2-.10 Animals 0440-2-.02 Applicability 0440-2-.11 High Frequency Electric Current
RULES OF TENNESSEE STATE BOARD OF COSMETOLOGY CHAPTER 0440-2 SANITARY RULES TABLE OF CONTENTS 0440-2-.01 Definitions 0440-2-.10 Animals 0440-2-.02 Applicability 0440-2-.11 High Frequency Electric Current
Environmental Health Department 58 St Johns Road, Newport, Isle of Wight PO30 1LT
 Environmental Health Department 58 St Johns Road, Newport, Isle of Wight PO30 1LT 2 The Local Government (Miscellaneous Provisions) Act 1982 requires persons who practice acupuncture, tattooing, ear piercing
Environmental Health Department 58 St Johns Road, Newport, Isle of Wight PO30 1LT 2 The Local Government (Miscellaneous Provisions) Act 1982 requires persons who practice acupuncture, tattooing, ear piercing
Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing. drape the piercing site.
 FACT SHEET Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing Equipment / Single use towel 1. Client preparation A towel may be used to drape the piercing site. The towel should
FACT SHEET Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing Equipment / Single use towel 1. Client preparation A towel may be used to drape the piercing site. The towel should
#74 - CHANGING A MOIST TO DRY DRESSING (TEST)
 #74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
#74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
