During the normal aging process, there is a loss
|
|
|
- Marshall Davis
- 5 years ago
- Views:
Transcription
1 COSMETIC DERMATOLOGY Soft Tissue Augmentation Ranella J. Hirsch, MD; Joel L. Cohen, MD Recent additions to the soft tissue augmentation armamentarium have greatly increased the dermatologic surgeon s choices in optimizing facial contouring and the treatment of acne scars. In this article, we review the science of fillers and look at the future of dermal fillers. Cutis. 2006;78: Solish and Pollack, 1 pinch the skin around the scar to be treated and observe the amount of correction to the scar complete correction with taut stretch predicts successful treatment with fillers. Recently, a plethora of new products and developments for soft tissue augmentation have been introduced. We review current treatment options and look at the future of soft tissue augmentation. During the normal aging process, there is a loss of connective and subcutaneous tissue, most notably on the face, neck, and hands. Subcutaneous tissue augmentation with injectable fillers can fill lines, replace lost volume, and reposition sagging structures. Thus, these materials may be used for a rejuvenating effect to soften the appearance of aging, provide a fuller more youthful face, and recontour lines that form over time. In addition to rejuvenation, fillers have an important role in repairing defects such as scars. Scars from acne, surgery, or trauma predominantly result from loss or contraction of tissue, and each also can be greatly improved with fillers. Scars best suited for therapy with filler materials are soft and easily distensible. A stretch test can be performed and is recommended preoperatively. According to Accepted for publication March 14, Dr. Hirsch is from Skincare Doctors, Cambridge, Massachusetts. Dr. Cohen is from AboutSkin Dermatology and DermSurgery, Denver, Colorado. Dr. Hirsch is a consultant and has received honoraria from Allergan, Inc, and Medicis Pharmaceutical Corporation. Dr. Cohen is a clinical trial participant, consultant, national trainer, and speaker for Allergan, Inc, and Medicis Pharmaceutical Corporation. He also is a consultant and trainer for Dermik Laboratories, a consultant for Inamed Corporation, and a clinical trial participant for Ipsen. Reprints: Ranella J. Hirsch, MD, Skincare Doctors, 777 Concord Ave, Suite 206, Cambridge, MA Optimizing Results With Fillers To optimize results using fillers for soft tissue augmentation, the practitioner must be knowledgeable about several important variables, including preprocedural instructions, choice of filler material, proper placement in the desired location, amount of implant deposited, postprocedural instructions, and timing of repeat injections. Each filler has individual properties that must be considered when selecting the product that is appropriate for a given patient and his/her goals. Different fillers have different tissue residence times, ranging from months to years. Selection of the perfect product for a given indication in a particular patient does not guarantee a perfect outcome. Precise placement of any filling substance is critical, requiring anatomic knowledge of the areas being treated as well as an understanding of the intended depth of placement of the material being utilized. We also feel that to optimize results, it is important that patients obtain incremental benefit from reevaluation and touch-up injections before an implant is completely absorbed. Other factors affecting filler selection include budget and time frame. Depending on the amount and type of filler used, it also is prudent to avoid soft tissue augmentation for approximately 10 to 14 days prior to an important business or social event, such as a wedding. Patients should be advised to avoid aspirin, nonsteriodal anti-inflammatory drugs, vitamin E, and other herbal supplements for VOLUME 78, SEPTEMBER
2 10 days prior to treatment with any fillers to decrease the risk for and degree of swelling and bruising. Preprocedure and postprocedure photography is essential for documentation; it can help show patients the degree of correction achieved. Too often, patients forget what you made go away and simply focus on what is leftover. Prior to any injection with soft tissue augmentation products, makeup should be completely removed and the treatment area cleaned with alcohol. Subsequently, a chlorhexidine preparation of the area is recommended. Postprocedure, ice to the treatment site can be effective at decreasing swelling and minimizing bruising. We have patients ice treatment areas for 20 minutes in the office to ensure the icing process is understood and started. We also tell patients to avoid exercise for 12 hours to decrease augmentation of blood to the area and to refrain from putting on makeup until the following day because of the potential risk for transfer of any bacterial or viral products from makeup; contamination is more than just a theoretic risk. Fillers can be classified in many different ways. Typically, products are classified as temporary, semipermanent, or permanent fillers. Determining whether the material is from an animal, nonanimal, or autologous source is another method of classifying fillers. The Holy Grail of dermal fillers is permanence coupled with a lack of antigenicity. 2 At present, no filler substance exists that is permanent without some level of antigenicity and inflammation. With dermal filling, less inflammation represents an improved chance at longevity because less inflammation is associated with a longer-lasting fill effect. These criteria are not met by fat or collagen from any source (bovine or cadaver), all of which are resorbed by the body over time. 3 Collagen Fillers Many dermal fillers are available for soft tissue augmentation. Zyderm 1, Zyderm 2, and Zyplast are suspensions of bovine dermal collagen packaged with lidocaine. The anesthetic tempers the discomfort of the intradermal injection. Zyderm 1 is available in a concentration of 35 mg/ml in phosphate-buffered saline solution with lidocaine added for anesthesia. Zyderm 2 is twice as concentrated at 65 mg/ml. 4 Zyplast is a more stable product because it contains collagen cross-linked with % gluteraldehyde, which strengthens the collagen fibers, rendering it more resistant to decomposition by the host. 5 Zyderm 1 is used to treat fine lines, shallow scars, and thin-skinned areas. 6 Zyderm 2 is indicated for the treatment of moderate lines, wrinkles, scars, and thicker-skinned areas. Zyplast is best used for contouring and shaping and to treat pronounced scars, lines, and furrows, as well as to define the lip border. A widely accepted practice is the layering of Zyderm 1 over Zyplast when both volume fill and subtle contour are required for optimal correction. While Zyplast is placed in the mid dermis, the injection technique for Zyderm is to raise a papillary dermal bleb as the material flows along superficial lines. It is necessary to overcorrect by approximately 200% with Zyderm as it is diluted with saline. When using this product, it is necessary to explain to patients that the saline is reabsorbed over 24 hours. When treating the glabella, botulinum toxin chemodenervation frequently is combined with Zyderm injections to enhance and prolong the correction obtained. Cutaneous necrosis, however, has been associated with soft tissue augmentation of the glabella, most likely resulting from deep product placement, causing compression of the watershed branches of the supratrochlear vessels or direct intravascular injection of the material. 6 The primary advantage of bovine collagen is its proven safety record over many years in expert hands. The largest drawback to therapy with bovine material is the risk for hypersensitivity in 3% to 5% of treated patients, which has been documented in the aesthetic dermatology literature. 6 Because a substantial number of patients with one negative skin test subsequently have developed a reaction at the treatment site, double skin testing is widely practiced (lowering the risk from 1% to less than 0.5%). 7 It is recommended that if a patient has not received treatment for more than one year, a single retest should be done with evaluation at 2 weeks. There has been some concern about the possibility of bovine collagen inciting crossover autoantibodies to human collagen, with theoretic risk for subsequent induction of collagen vascular disease. Various studies and panels have indicated that this type of induction has not occurred and would be extremely remote. 6-8 Public opinion also has swayed practitioners away from some animal-based products. Practitioners should avoid treating patients who have a history of lidocaine sensitivity, an anaphylactoid event, beef allergy, and, of course, previous sensitivity to bovine collagen. To reduce the risk for sensitivity to a bovine product, quell any possible concerns of potential risk for transferring some animal-hosted infectious processes, and obviate the need for skin testing, CosmoDerm and CosmoPlast human-derived collagen products were launched in early CosmoDerm is bioengineered human collagen 166 CUTIS
3 obtained from fibroblasts of neonatal foreskin. It is ideal for patients sensitive to bovine- and cadaverbased collagens. This human material has been extensively tested for viruses, tumorigenicity, and retroviruses, as well as known genetic diseases. The product etymology is similar to the respective bovine derivatives Zyderm and Zyplast. CosmoDerm and CosmoPlast have the additional advantage of requiring no pre-skin testing, so they can be used at the initial consultation visit. They provide immediate correction and are valuable adjuncts to the fillers category. 9 No important data have shown CosmoDerm and CosmoPlast to yield a measurably longer correction as compared with the bovine product, and our anecdotal experience has not suggested increased durability either. Autologen is an autograft composed of collagen removed from the donor patient during harvesting. 10 The tissue is frozen and sent for processing, where it can be stored for up to 5 years. When needed, it can be directly processed so that 1 sq in of harvested skin produces about 3 cc of Autologen product. The syringes of this processed material are stable for up to 6 months. Because it is autologous collagen, no skin testing is required, and patient anxiety about use of animal or another person s material is alleviated. The product contains no anesthetic, so topical anesthesia is necessary, and sometimes local infiltration blocks also are necessary. The injection sessions usually are spaced at 2-week intervals until the desired correction is achieved. The main disadvantages are the need for large tissue harvesting (usually skin from abdominoplasty or a face-lift procedure), the delay required for tissue processing, and the high cost of the product. It is composed of intact human dermal collagen fibers with intact telopeptides still attached, thus rendering it very resistant to enzymatic degradation, with reports suggesting durability of the injected material up to 18 months. 10 Isolagen is another autologous collagen product; however, harvesting requires only a small piece of tissue, typically a 3-mm punch biopsy harvested from the postauricular sulcus. 10 The harvested material is sent to the company where it is cultured and grown in vitro for 6 weeks. The material is then returned to the physician s office in 1- to 1.5-cc aliquots. The injections are similar to traditional collagen injections. Like Autologen, the material does not contain any anesthetic. Injections usually are spaced at 2- to 3-week intervals until the desired correction is reached. Since the material is autologous, there should not be any risk for allergic or immunologic reactions. Nevertheless, skin testing is recommended so that any by-product allergy can be detected. Finally, because viable fibroblasts, collagen matrix, and other materials required for replenishment of the dermal support structures are being injected, the Isolagen process offers the opportunity to have a durable correction, but there are few data on the exact duration of the product. 10 Cadaver-Derived Implants Cymetra and Fascian are alternative soft tissue augmentation products. Both are acellular freezedried dermal grafts processed from human cadaver dermis. No skin testing is required. Fascian is derived from human gastrocnemius (crural) fascia or fascia lata that is then processed, freeze dried, and ethylene-oxide sterilized to insure sterility. Harvested from screened human donors, Fascian is an injectable formulation of the same substance that is applicable for soft tissue repair and augmentation. Fascian is prepared in accordance with US Food and Drug Administration (FDA) regulations for processing nonviable human tissue, though cadaver-derived implants Cymetra, Fascian, and AlloDerm are not FDA approved. 11 Provided in preloaded syringes, all 3 products have several advantageous features. It does not require refrigeration, and, because of low allergenicity, test implantation is not required. Five different particle sizes (0.10 mm, 0.25 mm, 0.50 mm, 1.0 mm, and 2.0 mm) of Fascian are available in preloaded syringes containing 80 mg. Each particle size treats problems of different sizes and types. The syringe content can be hydrated in anesthetic solution to a volume of 1 to 3 cc and then injected through a needle. The product tends to spread evenly through the tissues. It is easy to handle and is stored at room temperature. 12 The process of tissue particulation has not altered the inherent graft properties, and Fascian seems to evoke the same process of collagen replacement that routinely occurs in larger grafts. Hyaluronic Acid Based Fillers Over the past 2 years, the limelight has been on hyaluronic acid (HA) based fillers. HA is a universal component of the extracellular matrix that, unlike collagen, is chemically identical across species and thus confers less risk for antigenicity. 13 In the past 2 years, the FDA approved several HA fillers including Restylane and Hylaform, and, more recently, Hylaform Plus and Captique. HA itself is a glycosaminoglycan polysaccharide composed of alternating residues of the monosaccharides d-glucuronic acid and N-acetyl-d-glucosamine VOLUME 78, SEPTEMBER
4 forming a long unbranched polyanionic chain. Glycosaminoglycans are abundant in fetal skin and are present in low levels in adulthood. The chains in these HA products are hydrated and coil upon themselves, giving the material both elasticity and viscosity. 14 The product is known to have exquisite water-binding capacity and thus is responsible for dermal hydration. 15 Restylane and Captique are derived from the fermentation of specific strains of streptococci, which are then alcohol precipitated, filtered, and dried. Hylaform is another HA derivative made from rooster combs. 16 Captique was FDA-approved in November 2004 and is very similar to Hylaform. Like Hylaform, it has a concentration of 5.5 mg/ml, contains 500 µm particles, and is cross-linked by divinyl sulfone. It also is injected with a 30-gauge needle. Captique, aside from its bacterial source, so closely resembles Hylaform that it is considered to be an extension of it as a new manufacturing source rather than a new product. Captique seems to be a useful product for areas such as the lips, which require minimal edema. Restylane, Perlane, and Restylane Fine Lines are biocompatible biotechnologically manufactured gels of HAs. 17 Though all 3 of these Restylane products are available in much of the world, including Europe and Canada, currently only Restylane is FDA approved in the United States. Restylane is packaged with sodium chloride and a phosphate buffer and comes as a preloaded 0.4- or 1.0-mL syringe. Restylane is more viscous (20 mg/ml) than Hylaform gel (5.5 mg/ml); Hylaform is more elastic. Both comprise the ground substance and are extremely hydrophilic. 17 All 3 of the Restylane products contain a concentration of 20 mg/ml. The difference between each of these products is their unique particle size. Restylane Fine Lines has the smallest particle size (200,000 gel particles/ml) and is the least viscous of the 3 products. It is injected through a 31-gauge needle and is intended for the correction of superficial defects. Restylane is of a larger particle size (100,000 gel particles/ml). It is injected through a 30-gauge needle and is intended for placement in the papillary dermis for correction of moderate rhytides. Perlane is of the largest particle size (10,000 gel particles/ml), is injected via a 27-gauge needle, and is best suited for deep rhytides and sculpting. With these products as well as other HA products, anesthesia can be provided through judicious use of ice, topical anesthetics, and nerve blocks. It is common practice to combine the use of an HA product such as Restylane for deeper rhytides with CosmoDerm or Zyderm for more superficial wrinkles. In addition, many practitioners are utilizing both botulinum toxin type A and dermal fillers to take advantage of their synergistic effects in tissue. Botulinum toxin type A functions to disrupt the muscle s ability to move; thus, it effectively prevents dynamic wrinkles from forming by keeping the musculature from pulling and imprinting lines in the overlying skin. By contrast, filler substances add volume. Used together, the results of each product last longer and are enhanced. In a prospective randomized study comparing the use of Restylane alone to the use of Restylane and botulinum toxin type A to treat severe glabellar rhytides, Carruthers and Carruthers 18 reported a better response and longer maintenance of correction. Current standard of care with HA does not include pretreatment allergy testing. Delayed reactions with Restylane, defined as occurring 14 days or more after the last injection, occur in 1 of every 25,000 injections. 19 Anecdotally, these reactions seem to be transient, lasting between 2 weeks and 2 months, and only rarely require the use of topical or injectable corticosteroids. It is imperative for practitioners to comprehend the distinct difference between delayed hypersensitivity reactions and true allergy. Lupton and Alster 20 reported on a particular case of hypersensitivity secondary to an impurity of the bacterial fermentation of the modified HA gel. Fernández-Aceñero et al 21 reported a case of a foreign body reaction after injection of HA. Histologic examination confirmed the presence of a nodule in the subcutaneous fat that stained intensely with Alcian blue at ph 2.7, confirming the presence of HA. 21 There are, as of the writing of this piece, 3 reported cases of HArelated allergy, which represents an extremely low complication rate It is not clear if these cases were the result of impurities in the material injected or true allergies. Restylane is a nonanimal stabilized HA (NASHA) with an adverse reaction profile of 0.15% in the pre material and 0.06% in the post-1999 product. A decrease in adverse incidents is attributed to the introduction of a more purified HA raw material post Nonetheless, practitioners must review the risks of the rare hypersensitivity reaction and/or the very rare allergy with prospective patients during the informed consent process. Worldwide, HA is not only manufactured under the names of Hylaform and Captique but also Juvéderm and Rofilan. The full spectrum of Juvéderm products (Juvéderm 18, Juvéderm 24, Juvéderm 24HV, Juvéderm 30, Juvéderm 30HV) currently is marketed 168 CUTIS
5 in Europe and Canada. Recently, the FDA approved Juvéderm 24HV, Juvéderm 30, and Juvéderm 30HV. 23 Based on our experience, it is a promising agent. Juvéderm, like Restylane and Captique, is a nonanimal-derived HA made from bacterial fermentation of streptococcus and cross-linked by butane-diol-diglycidyl ether. Juvéderm, however, is a homogeneous (nonparticulate) gel-based HA, in contrast to the other hyalurons that contain particles of different sizes. It is believed that the homogeneous structure of Juvéderm gel leads to increased longevity because there is less surface area available for host enzymatic processes to attack and degrade the HA. The homogeneous Juvéderm has less friction on injection (ie, more pliable and viscoelastic) and could be potentially less inflammatory than other HA products. The recently FDA-approved products include Juvéderm 24HV, Juvéderm 30, and Juvéderm 30HV. Juvéderm 24HV is highly cross-linked for increased versatility in contouring and volumizing middermal defects, Juvéderm 30 is intended for the subtle correction of facial wrinkles and folds, and Juvéderm 30HV is more highly cross-linked for volumizing and correcting deeper folds and wrinkles. 23 Unlike Juvéderm 18, which is composed of 18 mg of cross-linked HA, Juvéderm 24 and 30 contain 24 mg of cross-linked nonanimalderived HA. These products also do not contain anesthetic and thus require either topical anesthetic or injectable anesthetic prior to treatment in most patients. As with other hyalurons, injection technique is variable with linear threading, serial puncture, and cross hatching being advocated by various skilled injectors. Aquamid gel is 2.5% polyacrylamide and 97.5% water. Given the lack of particles and a very high concentration of water, it is homogeneous, stable, and nonbiodegradable. Aquamid has been authorized for sale in Europe since March de Cassia Novaes and Berg 24 present experience with Aquamid in 59 subjects (43 subjects [72%] received lip augmentation and 8 subjects [13%] received cheekbone enlargement and nasolabial fold correction with high levels of patient satisfaction). Currently, in the United States, HA-based products are favored fillers among dermatologic surgeons given their longevity, ease of use, and safety data. 25 The Federal Food, Drug, and Cosmetic Act allows any legally marketed FDA-approved product to be administered for any condition in a doctorpatient relationship. 26 The term for this is off-label use and it is a widely accepted practice in the United States. Permanent and Semipermanent Fillers Recently, there has been renewed interest in more permanent fillers. Worldwide, permanent and semipermanent skin implants include New-Fill, Artecoll, Dermalive, and Evolence. The rights to New-Fill, renamed Sculptra in the United States, have been purchased and the product was fast-tracked through the FDA to gain approval for the treatment of facial lipoatrophy in patients with human immunodeficiency virus. 27 New-Fill is polylactic acid that occurs naturally and is known to stimulate the body s own collagen production. Safety data presently are lacking, as no large trials have been reported yet in the United States. The deeper level of placement, large volume of dilution, and longer reconstitution times seem to anecdotally be greatly reducing the fairly high rates of nodule formation reported in Europe. Artecoll is a nonbiodegradable permanent filler containing polymethylmethacralate microspheres 25% of 30 to 40 µm in diameter suspended in bovine collagen solution with saline and lidocaine 0.3% to minimize discomfort during injection. 28 It is designed for implantation into the deep reticular dermis. It has a 2-fold function: (1) an immediate filler, and (2) an inert long-lasting scaffold to stimulate permanent deposition of autologous collagen around the microspheres. The bovine collagen suspension component has an expected lifespan of 3 to 6 months and necessitates skin testing. Artecoll has been widely used in Europe for the last 10 years. 28 The bovine collagen in Artecoll is obtained from calves aged up to 6 months that only have been fed milk and vegetables. 28 The goal of this diet is to avoid administration of hormones or antibiotics. To prepare the solution, the collagen is washed with sodium hydroxide to protect against contamination with bovine spongiform encephalitis and other viruses. The telopeptide immunogenic ends then are removed and the material filtered to lower antigenicity. 28 The promise of long-standing correction leads the list of advantages and disadvantages. While the notion of long-standing correction is appealing to both patients and practitioners, lateonset granulomas have been reported in the aesthetic and dermatologic literature. Alcalay et al 29 recently reported the case of a 54-year-old woman who presented with firm nodules at the site of an Artecoll injection 14 months prior. Histologic examination revealed histiocytic granulomas with giant cells and vacuoles. If the product is injected too superficially, blanching will be observed and there is a substantial risk for VOLUME 78, SEPTEMBER
6 palpable permanent beading. Also of note is the risk for delayed sensitization to the bovine collagen within Artecoll. 30 Radiesse is a calcium hydroxylapatite injectable filler currently FDA approved for oral/maxillofacial defects, vocal fold insufficiency, and radiographic tissue marking. 31 Radiesse is the longest lasting nonpermanent wrinkle filler available with results that can last up to 2 to 5 years. Its effects are longer lasting than any other treatment available, but there is concern that it can interfere with the reading of dental x-rays. Long-term clinical studies on its use as a soft tissue filler are lacking. 31 Of critical importance with any injectable material is both the skill of the injector and the reliability of the product that is injected. It is imperative that patients carefully inquire about who is injecting and the product that is being injected. In a report on the development of orofacial granulomas after the use of filler substances, only 3 of 11 patients involved knew what substance had been injected. 32 Silicone Silicone, a synthetic compound composed of long polymers of dimethysiloxanes, likely is the most commonly implanted material in all of medical practice. 33 It has existed in an assortment of incarnations over many decades and can possess a variety of physical characteristics depending on its chemical structure. Its consistency can range from a gel form to an expansive shell (used as a tissue expander) to a stable solid compound. Solid silicone facial implants are highly inert and resistant to degradation but by nature are hydrophobic and nonporous. Consequently, silicone does not allow for fibrovascular ingrowth that, when achieved, yields several important benefits, including immobility and infection resistance. Thus, part of the implantation process for dermal implants is the fenestration with a scalpel or large bore needle to incite fibrous ingrowth. 33 In the United States, use of silicone for soft tissue augmentation is not FDA approved and constitutes an off-label use of the product. 34 Side effects include the development of granulomas and a variety of rare immunologic manifestations. 35 Currently, there are 2 FDA-approved medical-grade liquid injectables (approved for ophthalmic use to tamponade retinal detachment) AdatoSil 5000 and Silikon Polytetrafluorethylene in the form of the Softform implant has a fibrillar nature that permits fibrovascular ingrowth into a microporous surface, permitting its widespread use in both soft tissue and bony augmentation, including the lips, nasolabial folds, and malar areas. Its history is poor with a substantial complication profile since its introduction in The 4 most frequent postoperative complications are extrusion, movement of the product, infection, and granulomatous swelling necessitating 8% removal of the device after implantation. 34 Autologous Fat Transplantation Unlike dermal fillers such as collagen and HA that function by the replacement of volume to a dead space, autologous fat transplantation is the grafting of viable donor tissue. Fat transplantation has many potential dermatologic uses, including the augmentation of congenital defects, management of scarring disease such as morphea, and use in lipodystrophy in patients with human immunodeficiency virus. 36 Best recipient sites are distensible scars associated with subdermal atrophy. 37 It also is used for cosmetic purposes, including breast augmentation and rejuvenation of aging hands. 38 Many techniques exist, but the precise conditions yielding the most favorable survival of the transplanted fat are poorly understood. 39 The distinct advantage is the lack of immune response and the fact that this product can be frozen and reused. Meticulous care to prevent clerical error between stored donor and recipient tissue is crucial. The main disadvantages are the time and procedure required for the harvest and the unpredictable correction secondary to irregular resorption. 39 Comment The skill of the injector and the reliability of what is injected are of supreme importance for optimum results with any filler material. It is imperative that patients are treated by qualified injectors who understand the precise anatomy to be treated and know exactly what is being injected. Practitioners are fortunate to have many choices available to treat patients with acne scars and scars from other dermatologic conditions. Judicious patient selection, appropriate product selection, and meticulous technique are the best ways to ensure high levels of patient satisfaction and optimize clinical improvement. REFERENCES 1. Solish N, Pollack SV. Approaches to acne scarring: a review. J Cutan Med Surg. 1998;2(suppl 3): CUTIS
7 2. Krauss MC. Recent advances in soft tissue augmentation. Semin Cutan Med Surg. 1999;18: Allen O. Response to subdermal implantation of texturized microimplants in humans. Aesthetic Plast Surg. 1992;16: Hirsch RJ, Lewis AB. Treatment of acne scarring. Semin Cutan Med Surg. 2001;20: Bailin MD, Bailin PM. Case studies: correction of surgical scars, acne scars, and rhytids with Zyderm and Zyplast implants. J Dermatol Surg Oncol. 1988;14(suppl 1): DeLustro F, Mackinnon V, Swanson NA. Immunology of injectable collagen in human subjects. J Dermatol Surg Oncol. 1988;14(suppl 1): Elson ML. The role of skin testing in the use of collagen injectable materials. J Dermatol Surg Oncol. 1989;15: Klein AW. Bonfire of the wrinkles. J Dermatol Surg Oncol. 1991;17: Klein AW. Tissue fillers [letter]. J Drugs Dermatol. 2003;2: Fagien S. Facial soft-tissue augmentation with injectable autologous and allogeneic human tissue collagen matrix (autologen and dermalogen). Plast Reconstr Surg. 2000;105: ; discussion Burres S. Preserved particulate fascia lata for injection: a new alternative. Dermatol Surg. 1999;25: Burres S. Soft-tissue augmentation with Fascian. Clin Plast Surg. 2001;28: Larsen NE, Pollack CT, Reiner K, et al. Hylan gel biomaterial: dermal and immunologic compatibility. J Biomed Mater Res. 1993;27: Comper WD, Laurent TC. Physiological function of connective tissue polysaccharides. Physiol Rev. 1978;58: Meyer LJ, Stern R. Age-dependent changes of hyaluronan in human skin. J Invest Dermatol. 1994;102: Harry C. A comparison of today s treatment options for soft tissue augmentation. Skin & Aging. March 1999;3: Duranti F, Salti G, Bovani B, et al. Injectable hyaluronic acid gel for soft tissue augmentation. a clinical and histological study. Dermatol Surg. 1998;24: Carruthers J, Carruthers A. A prospective, randomized, parallel group study analyzing the effect of BTX-A (Botox) and nonanimal sourced hyaluronic acid (NASHA, Restylane) in combination compared with NASHA (Restylane) alone in severe glabellar rhytids in adult female subjects: treatment of severe glabellar rhytids with a hyaluronic acid derivative compared with the derivative and BTX-A. Dermatol Surg. 2003;29: Friedman PM, Mafong EA, Kauvar AN, et al. Safety data of injectable nonanimal stabilized hyaluronic acid for soft tissue augmentation. Dermatol Surg. 2002;28: Lupton JR, Alster TS. Cutaneous hypersensitivity reaction to injectable hyaluronic acid gel. Dermatol Surg. 2000;26: Fernández-Aceñero MJ, Zamora E, Borbujo J. Granulomatous foreign body reaction against hyaluronic acid: report of a case after lip augmentation. Dermatol Surg. 2003;29: Raulin C, Greve B, Hartschuh W, et al. Exudative granulomatous reaction to hyaluronic acid (Hylaform). Contact Dermatitis. 2000;43: Allergan announces FDA approval of Juvéderm gel family of products for the treatment of facial wrinkles and folds [press release]. Irvine, Calif: Allergan, Inc; June 5, de Cassia Novaes W, Berg A. Experiences with a new nonbiodegradable hydrogel (Aquamid): a pilot study. Aesthetic Plast Surg. 2003;27: Narins RS, Brandt F, Leyden J, et al. A randomized, double-blind, multicenter comparison of the efficacy and tolerability of Restylane versus Zyplast for the correction of nasolabial folds. Dermatol Surg. 2003;29: US Food and Drug Administration. Federal Food, Drug, and Cosmetic Act. Available at: /opacom/laws/fdcact/fdctoc.htm. Accessed August 17, Moyle G, Lysakova L, Brown S, et al. Polylactate (NewFill) injections subjectively and objectively improve appearance and reduce anxiety and depression scores in HIV positive persons with facial lipoatrophy: a randomized, open label, immediate vs. delayed therapy study. Poster presented at: 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy; September 27-30, 2002; San Diego, California. 28. Lemperle G, Romano JJ, Busso M. Soft tissue augmentation with Artecoll: 10-year history, indications, techniques, and complications. Dermatol Surg. 2003;29: Alcalay J, Alkalay R, Gat A, et al. Late-onset granulomatous reaction to Artecoll. Dermatol Surg. 2003;29: McClelland M, Egbert B, Hanko V, et al. Evaluation of Artecoll polymethylmethacrylate implant for soft-tissue augmentation: biocompatibility and chemical characterization. Plast Reconstr Surg. 1997;100: Lemperle G, Morhenn V, Charrier U. Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthetic Plast Surg. 2003;27: ; discussion Rubin JP, Yaremchuk MJ. Complications and toxicities of implantable biomaterials used in facial reconstructive and aesthetic surgery: a comprehensive review of the literature. Plast Reconstr Surg. 1997;100: Rapaport MJ, Vinnik C, Zarem H. Injectable silicone: cause of facial nodules, cellulites, ulceration, and migration. Aesthetic Plast Surg. 1996;20: VOLUME 78, SEPTEMBER
8 34. Brody HJ. Complications of expanded polytetrafluoroethylene (e-ptfe) facial implant. Dermatol Surg. 2001;27: Lombardi T, Samson J, Plantier F, et al. Orofacial granulomas after injection of cosmetic fillers. histopathologic and clinical study of 11 cases. J Oral Pathol Med. 2004;33: Moskowitz L. Treatment of facial hemiatrophy by transplantation of fat tissues. Med Clin. 1930;26: Pinski KS. Microlipoinjection and autologous collagen. Dermatol Clin. 1995;13: Vita VT. Lipoinjection and aging hands. Am J Cosmet Surg. 1989;6: Rohrich RJ, Sorokin ES, Brown SA. In search of improved fat transfer viability: a quantitative analysis of the role of centrifugation and harvest site. Plast Reconstr Surg. 2004;113: CUTIS
Facial Fillers Dr Tarek Said Professor of Plastic Surgery Cairo University 2010
 Facial Fillers Dr Tarek Said Professor of Plastic Surgery Cairo University 2010 Synthetic Fillers (HA fillers, Non-HA fillers including Collagen) Autologous Fat Facial Implantables (AlloDerm & Gore-Tex)
Facial Fillers Dr Tarek Said Professor of Plastic Surgery Cairo University 2010 Synthetic Fillers (HA fillers, Non-HA fillers including Collagen) Autologous Fat Facial Implantables (AlloDerm & Gore-Tex)
PRESS MATERIAL. Contents: Appendix: Backgrounder Q-Med 2 Backgrounder RESTYLANE 3 Questions and answers 5 Recommended reading 7
 PRESS MATERIAL Contents: Backgrounder Q-Med 2 Backgrounder RESTYLANE 3 Questions and answers 5 Recommended reading 7 Appendix: Patient brochure Physician brochure Before and after pictures Clinical study
PRESS MATERIAL Contents: Backgrounder Q-Med 2 Backgrounder RESTYLANE 3 Questions and answers 5 Recommended reading 7 Appendix: Patient brochure Physician brochure Before and after pictures Clinical study
Informed Consent for Dermal Filler
 Informed Consent for Dermal Filler NAME: DATE OF BIRTHG: ADDRESS: CELL PHONE: EMAIL: www.medicaleyecenter.com Please initial all of the following sections confirming that you have read and understand each
Informed Consent for Dermal Filler NAME: DATE OF BIRTHG: ADDRESS: CELL PHONE: EMAIL: www.medicaleyecenter.com Please initial all of the following sections confirming that you have read and understand each
Complete Dermal Integration. Proven Duration.
 Complete Dermal Integration. Proven Duration. Introducing BELOTERO BALANCE Dermal Filler. BELOTERO BALANCE Dermal Filler is uniquely manufactured with CPM Technology to give you precision to treat a wide
Complete Dermal Integration. Proven Duration. Introducing BELOTERO BALANCE Dermal Filler. BELOTERO BALANCE Dermal Filler is uniquely manufactured with CPM Technology to give you precision to treat a wide
The first step: Choose a surgeon you can trust COPYRIGHT ASPS
 / INJECTABLE FILLERS The Symbol of Excellence in Plastic Surgery A public education service of the American Society of Plastic Surgeons. The first step: Choose a surgeon you can trust Plastic surgery involves
/ INJECTABLE FILLERS The Symbol of Excellence in Plastic Surgery A public education service of the American Society of Plastic Surgeons. The first step: Choose a surgeon you can trust Plastic surgery involves
INJECTABLES. Botox Cosmetic Page 1 of 7. FAQ s
 290 Country Club Drive, Stockbridge, Georgia 30281 770.506.9123 www.schillingmedicalspa.com FAQ s INJECTABLES Botox Cosmetic WHAT EXACTLY IS BOTOX COSMETIC? BOTOX Cosmetic is a purified protein produced
290 Country Club Drive, Stockbridge, Georgia 30281 770.506.9123 www.schillingmedicalspa.com FAQ s INJECTABLES Botox Cosmetic WHAT EXACTLY IS BOTOX COSMETIC? BOTOX Cosmetic is a purified protein produced
Understanding and Using Hyaluronic Acid. Seth L. Matarasso, MD
 Understanding and Using Hyaluronic Acid Seth L. Matarasso, MD Understanding and Using Hyaluronic Acid Injectable synthetic hyaluronic acid is biodegradable and biocompatible with human hyaluronic acid.
Understanding and Using Hyaluronic Acid Seth L. Matarasso, MD Understanding and Using Hyaluronic Acid Injectable synthetic hyaluronic acid is biodegradable and biocompatible with human hyaluronic acid.
Hyaluronic acid and the advanced thixotropic
 Made in Germany by Hyaluronic acid and the advanced thixotropic technology (ATT) Hyaluronic acid (HA) has been used in medicine for a long time, and over many years has been proven to have an excellent
Made in Germany by Hyaluronic acid and the advanced thixotropic technology (ATT) Hyaluronic acid (HA) has been used in medicine for a long time, and over many years has been proven to have an excellent
MID FACE VOLUMIZING 6/30/2015 DISCLOSURES. No Industry Disclosures
 MID FACE VOLUMIZING Heather D. Rogers Clinical lassistant Professor of Dermatology UW School of Medicine Seattle, WA DISCLOSURES No Industry Disclosures Generic names when possible Trade name when necessary
MID FACE VOLUMIZING Heather D. Rogers Clinical lassistant Professor of Dermatology UW School of Medicine Seattle, WA DISCLOSURES No Industry Disclosures Generic names when possible Trade name when necessary
FACIAL ARTS. Manufacturer: naturelize GmbH Kasseler Straße Bad Emstal Germany Phone: +49 (0)
 Manufacturer: naturelize GmbH Kasseler Straße 47 34308 Bad Emstal Germany Phone: +49 (0) 5624 926 7630 www.naturelize.com Fax: +49 (0) 5624 926 7639 Mail: customer support@naturelize.com supporting esthetic
Manufacturer: naturelize GmbH Kasseler Straße 47 34308 Bad Emstal Germany Phone: +49 (0) 5624 926 7630 www.naturelize.com Fax: +49 (0) 5624 926 7639 Mail: customer support@naturelize.com supporting esthetic
The wealth of new options offers opportunities for patients and clinicians. Here s how to make fillers work in your office.
 The wealth of new options offers opportunities for patients and clinicians. Here s how to make fillers work in your office. The emergence of new filler agents in the past year and a half has been nothing
The wealth of new options offers opportunities for patients and clinicians. Here s how to make fillers work in your office. The emergence of new filler agents in the past year and a half has been nothing
Collagen
 Collagen Injectable fillers are one of the most popular facial rejuvenation techniques. As we age, the underlying tissues that keep our skin looking youthful and firm begin to break down due to the effects
Collagen Injectable fillers are one of the most popular facial rejuvenation techniques. As we age, the underlying tissues that keep our skin looking youthful and firm begin to break down due to the effects
FAQs DERMAL FILLERS. 1 P age
 Dermal fillers (also called soft tissue fillers) are a non-surgical injectable treatment used to restore facial volume, create youthful facial contours, add volume to lips, and smooth out and reduce the
Dermal fillers (also called soft tissue fillers) are a non-surgical injectable treatment used to restore facial volume, create youthful facial contours, add volume to lips, and smooth out and reduce the
Hyaluronic acid. and the Advanced. Thixotropic
 HYAcorp_Broschüre_A5_RZ 04.01.16 15:53 Seite 2 Hyaluronic acid Hyaluronic acid (HA) is in use in medicine for a long time with a long safety profile. HA in his natural form has a and the Advanced short
HYAcorp_Broschüre_A5_RZ 04.01.16 15:53 Seite 2 Hyaluronic acid Hyaluronic acid (HA) is in use in medicine for a long time with a long safety profile. HA in his natural form has a and the Advanced short
Successful treatment of the nasolabial fold (NLF)
 Treatment of Nasolabial Folds With Fillers According to the author, injectable dermal fillers can be used effectively to treat nasolabial folds. He offers advice on evaluating the depth and classifying
Treatment of Nasolabial Folds With Fillers According to the author, injectable dermal fillers can be used effectively to treat nasolabial folds. He offers advice on evaluating the depth and classifying
Injectable Soft Tissue Fillers: Practical Applications. Karol A Gutowski, MD, FACS
 Injectable Soft Tissue Fillers: Practical Applications Karol A Gutowski, MD, FACS Disclosures Instructor for Suneva (Bellafill) Will describe off-label uses Will use brand names Injectable Tissue Filler
Injectable Soft Tissue Fillers: Practical Applications Karol A Gutowski, MD, FACS Disclosures Instructor for Suneva (Bellafill) Will describe off-label uses Will use brand names Injectable Tissue Filler
Five-Year Safety and Efficacy of a Novel Polymethylmethacrylate Aesthetic Soft Tissue Filler for the Correction of...
 See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/5762787 Five-Year Safety and Efficacy of a Novel Polymethylmethacrylate Aesthetic Soft Tissue
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/5762787 Five-Year Safety and Efficacy of a Novel Polymethylmethacrylate Aesthetic Soft Tissue
A Randomized, Double-Blind, Multicenter Comparison of the Efficacy and Tolerability of Restylane Versus Zyplast for the Correction of Nasolabial Folds
 A Randomized, Double-Blind, Multicenter Comparison of the Efficacy and Tolerability of Restylane Versus Zyplast for the Correction of Nasolabial Folds RHODA S. NARINS, MD,PC, n FREDRIC BRANDT, MD, w JAMES
A Randomized, Double-Blind, Multicenter Comparison of the Efficacy and Tolerability of Restylane Versus Zyplast for the Correction of Nasolabial Folds RHODA S. NARINS, MD,PC, n FREDRIC BRANDT, MD, w JAMES
Informed Consent Injectable Fillers
 Informed Consent Injectable Fillers INSTRUCTIONS This is an informed-consent document which has been prepared to help your plastic surgeon inform you concerning Juvederm & Juvederm Ultra Plus with Lidocaine
Informed Consent Injectable Fillers INSTRUCTIONS This is an informed-consent document which has been prepared to help your plastic surgeon inform you concerning Juvederm & Juvederm Ultra Plus with Lidocaine
MARK D. EPSTEIN, M.D. F.A.C.S. Hyaluronic Acid (HA) INJECTION - INFORMATION FOR PATIENTS
 Hyaluronic Acid (HA) INJECTION - INFORMATION FOR PATIENTS INSTRUCTIONS This is an informed-consent document which has been prepared to help you understand hyaluronic acid (Juvederm, Restylane, Belotero)
Hyaluronic Acid (HA) INJECTION - INFORMATION FOR PATIENTS INSTRUCTIONS This is an informed-consent document which has been prepared to help you understand hyaluronic acid (Juvederm, Restylane, Belotero)
NORMAL OCCURRENCES DURING TISSUE FILLER INJECTIONS, INCLUDING HYLAFORM and JUVEDERM
 INSTRUCTIONS This informed-consent document has been prepared to help inform you about various soft tissue filler materials, their use, risks, and alternative treatments. It is important that you read
INSTRUCTIONS This informed-consent document has been prepared to help inform you about various soft tissue filler materials, their use, risks, and alternative treatments. It is important that you read
EVERYONE WILL NOTICE. No One Will Know.
 THE WORLD S #1 SELLING DERMAL FILLER COLLECTION EVERYONE WILL NOTICE. No One Will Know. Get the natural-looking, long-lasting results you desire. Ask your aesthetic specialist about JUVÉDERM today. Actual
THE WORLD S #1 SELLING DERMAL FILLER COLLECTION EVERYONE WILL NOTICE. No One Will Know. Get the natural-looking, long-lasting results you desire. Ask your aesthetic specialist about JUVÉDERM today. Actual
(Injection of collagen, hyaluronic acid or other filler materials) INFORMED CONSENT FOR DERMAL FILLER
 INFORMED CONSENT FOR DERMAL FILLER (Injection of collagen, hyaluronic acid or other filler materials) INTRODUCTION Dermal fillers are injected just under the skin s surface in order to temporarily correct
INFORMED CONSENT FOR DERMAL FILLER (Injection of collagen, hyaluronic acid or other filler materials) INTRODUCTION Dermal fillers are injected just under the skin s surface in order to temporarily correct
designed to stimulate collagen
 Discover the volumizer designed to stimulate collagen with results that last over 2 years* Elaine: Age 40 (2.5 vials) Christine: Age 39 (2 vials) Veronica: Age 33 (4 vials) Actual Sculptra Aesthetic patients
Discover the volumizer designed to stimulate collagen with results that last over 2 years* Elaine: Age 40 (2.5 vials) Christine: Age 39 (2 vials) Veronica: Age 33 (4 vials) Actual Sculptra Aesthetic patients
Own Your Beauty. with the Belotero range. Enjoy natural results with a filler tailored to your needs.
 Own Your Beauty with the Belotero range Enjoy natural results with a filler tailored to your needs. Show your emotions with conf idencefi When was the last time you dared to show your emotions with self-assurance
Own Your Beauty with the Belotero range Enjoy natural results with a filler tailored to your needs. Show your emotions with conf idencefi When was the last time you dared to show your emotions with self-assurance
Sirtaz S. Sibia, D.O., FAOCO Austin Coleman, D.O., FAOCO Scottsdale, Arizona May 2016
 Sirtaz S. Sibia, D.O., FAOCO Austin Coleman, D.O., FAOCO Scottsdale, Arizona May 2016 Approach To The Cosmetic Patient Anatomic areas of concern to the patient Give the patient a mirror and have them point
Sirtaz S. Sibia, D.O., FAOCO Austin Coleman, D.O., FAOCO Scottsdale, Arizona May 2016 Approach To The Cosmetic Patient Anatomic areas of concern to the patient Give the patient a mirror and have them point
Avoiding Complications and Achieving Success in Filler Injections. Sammy Sinno, MD
 24TH Annual Meeting Avoiding Complications and Achieving Success in Filler Injections Sammy Sinno, MD Upon completion of this presentation, the participants will self-report an increase in knowledge about:
24TH Annual Meeting Avoiding Complications and Achieving Success in Filler Injections Sammy Sinno, MD Upon completion of this presentation, the participants will self-report an increase in knowledge about:
Safety of Storing and Reusing Hyaluronic Acid Fillers: A Retrospective Chart Review. Patrick K. Safo, MD, PhD; Christina Wahlgren, MD; Suzan Obagi, MD
 Study Safety of Storing and Reusing Hyaluronic Acid Fillers: A Retrospective Chart Review Patrick K. Safo, MD, PhD; Christina Wahlgren, MD; Suzan Obagi, MD Injectable dermal fillers are an integral component
Study Safety of Storing and Reusing Hyaluronic Acid Fillers: A Retrospective Chart Review Patrick K. Safo, MD, PhD; Christina Wahlgren, MD; Suzan Obagi, MD Injectable dermal fillers are an integral component
*Resilient Hyaluronic Acid
 *Resilient Hyaluronic Acid TEOSYAL RHA* DYNAMIC AESTHETICS EXPAND YOUR POSSIBILITIES FACIAL REJUVENATION High patient expectations 1,2 / High medical standards 1,2 Natural results Immediate and long lasting
*Resilient Hyaluronic Acid TEOSYAL RHA* DYNAMIC AESTHETICS EXPAND YOUR POSSIBILITIES FACIAL REJUVENATION High patient expectations 1,2 / High medical standards 1,2 Natural results Immediate and long lasting
Guide to Dermal FillerS for Facial Rejuvenation
 Guide to Dermal FillerS for Facial Rejuvenation Although no one likes the thought of aging, we can be thankful that we are living in this modern age when there are more facial cosmetic procedures than
Guide to Dermal FillerS for Facial Rejuvenation Although no one likes the thought of aging, we can be thankful that we are living in this modern age when there are more facial cosmetic procedures than
Guide to THE Types of dermal fillers
 Guide to THE Types of dermal fillers Injectable dermal fillers can give you a more youthful look for a fraction of what a traditional facelift costs. Most will fill hollows, lines, and wrinkles in less
Guide to THE Types of dermal fillers Injectable dermal fillers can give you a more youthful look for a fraction of what a traditional facelift costs. Most will fill hollows, lines, and wrinkles in less
INFORMED CONSENT Juvederm INJECTION
 INSTRUCTIONS This is an informed-consent document which has been prepared to help Dr. Jennifer Geoghegan inform you concerning Juvederm (Non-Animal Stabilized Hyaluronic Acid, Allergan Aesthetics) tissue
INSTRUCTIONS This is an informed-consent document which has been prepared to help Dr. Jennifer Geoghegan inform you concerning Juvederm (Non-Animal Stabilized Hyaluronic Acid, Allergan Aesthetics) tissue
NEWS RELEASE. CONTACTS: Investors: Lisa DeFrancesco (862) Media: Mark Marmur (862) Ember Garrett (714)
 NEWS RELEASE CONTACTS: Investors: Lisa DeFrancesco (862) 261-7152 Media: Mark Marmur (862) 261-7558 Ember Garrett (714) 246-3525 JUVÉDERM VOLBELLA XC APPROVED BY U.S. FDA FOR USE IN LIPS AND PERIORAL RHYTIDS
NEWS RELEASE CONTACTS: Investors: Lisa DeFrancesco (862) 261-7152 Media: Mark Marmur (862) 261-7558 Ember Garrett (714) 246-3525 JUVÉDERM VOLBELLA XC APPROVED BY U.S. FDA FOR USE IN LIPS AND PERIORAL RHYTIDS
Update on Dermal Fillers. Joely Kaufman, MD Voluntary Assistant Professor Dermatology University of Miami Miller School of Medicine
 Update on Dermal Fillers Joely Kaufman, MD Voluntary Assistant Professor Dermatology University of Miami Miller School of Medicine Relevant Disclosures Contracted Research: Merz Pharmaceuticals; Revance
Update on Dermal Fillers Joely Kaufman, MD Voluntary Assistant Professor Dermatology University of Miami Miller School of Medicine Relevant Disclosures Contracted Research: Merz Pharmaceuticals; Revance
Informed Consent Hyaluronic Acid Filler Injection
 Informed Consent Hyaluronic Acid Filler Injection INSTRUCTIONS This is an informed-consent document which has been prepared to help inform you about hyaluronic acidbased (non-animal stabilized) tissue
Informed Consent Hyaluronic Acid Filler Injection INSTRUCTIONS This is an informed-consent document which has been prepared to help inform you about hyaluronic acidbased (non-animal stabilized) tissue
Hyaluronic Acid Injections: Incorporating Advanced Microinjection Techniques Into Practice ReachMD Page 1 of 6
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
INFORMED CONSENT SOFT TISSUE FILLER INJECTION
 INSTRUCTIONS This informed-consent document has been prepared to help inform you about Hylaform (animal-origin, stabilized hyaluronic acid, INAMED) tissue-filler injection therapy Restylane (Non-Animal
INSTRUCTIONS This informed-consent document has been prepared to help inform you about Hylaform (animal-origin, stabilized hyaluronic acid, INAMED) tissue-filler injection therapy Restylane (Non-Animal
UNCORRECTED PROOF. Dermal Fillers and Combinations of Fillers for Facial Rejuvenation ARTICLE IN PRESS. Kenneth Beer, MD. derm.theclinics.
 1 2 3 4 5 6 7 ½Q2Š½Q3Š 8 9 ½Q5Š 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 ½Q4Š Dermal Fillers and Combinations of Fillers
1 2 3 4 5 6 7 ½Q2Š½Q3Š 8 9 ½Q5Š 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 ½Q4Š Dermal Fillers and Combinations of Fillers
Practical tips in Cosmetic Dermatology for the General Dermatologist. Dee Anna Glaser, MD
 Practical tips in Cosmetic Dermatology for the General Dermatologist Dee Anna, MD Disclosure Statement Cosmetic Dermatology Dee Anna, M.D. Professor & Vice Chairman Director Cosmetic and Laser Services
Practical tips in Cosmetic Dermatology for the General Dermatologist Dee Anna, MD Disclosure Statement Cosmetic Dermatology Dee Anna, M.D. Professor & Vice Chairman Director Cosmetic and Laser Services
The unique treatment that restores your skin s inner structure for a more youthful-looking appearance
 THE SECRET TO YOUTHFUL-LOOKING SKIN Actual patient. Individual results may vary. The unique treatment that restores your skin s inner structure for a more youthful-looking appearance Sculptra Aesthetic
THE SECRET TO YOUTHFUL-LOOKING SKIN Actual patient. Individual results may vary. The unique treatment that restores your skin s inner structure for a more youthful-looking appearance Sculptra Aesthetic
. DEFY LINES. along the sides of your nose and mouth ON YOUR FACE.
 . DEFY LINES. ( PARENTHESES HAVE NO PLACE) ON YOUR FACE. n Instantly smooths away the deeper lines along the sides of your nose and mouth n Provides natural-looking results Actual patient. Results may
. DEFY LINES. ( PARENTHESES HAVE NO PLACE) ON YOUR FACE. n Instantly smooths away the deeper lines along the sides of your nose and mouth n Provides natural-looking results Actual patient. Results may
Revitalize Rejuvenate ReNew
 Revitalize Rejuvenate ReNew The Revanesse Family of Products The Revanesse family of products incorporates the latest advancements in cross-linking technology, resulting in a high quality, safe, long lasting
Revitalize Rejuvenate ReNew The Revanesse Family of Products The Revanesse family of products incorporates the latest advancements in cross-linking technology, resulting in a high quality, safe, long lasting
ORIGINAL ARTICLE. Evaluation of Acellular Dermal Graft in Sheet (AlloDerm) and Injectable (Micronized AlloDerm) Forms for Soft Tissue Augmentation
 ORIGINAL ARTICLE Evaluation of Acellular Dermal Graft in Sheet (AlloDerm) and Injectable (Micronized AlloDerm) Forms for Soft Tissue Augmentation Clinical Observations and Histological Analysis Anthony
ORIGINAL ARTICLE Evaluation of Acellular Dermal Graft in Sheet (AlloDerm) and Injectable (Micronized AlloDerm) Forms for Soft Tissue Augmentation Clinical Observations and Histological Analysis Anthony
Massey Medical. Medical History (Dermal Filler) MEDICAL INFORMATION: I am interested in the following services: Juvederm: Botox:
 Medical History (Dermal Filler) Name: Date: _ Date of Birth: Phone: _ MEDICAL INFORMATION: I am interested in the following services: Juvederm: Botox: NO YES Allergies history of severe allergy or anaphylaxis.
Medical History (Dermal Filler) Name: Date: _ Date of Birth: Phone: _ MEDICAL INFORMATION: I am interested in the following services: Juvederm: Botox: NO YES Allergies history of severe allergy or anaphylaxis.
INFORMED CONSENT HYLAFORM INJECTION
 INSTRUCTIONS This informed-consent document has been prepared to help inform you about Hylaform (animal-origin, stabilized hyaluronic acid, INAMED) tissue-filler injection therapy, its risks, and alternative
INSTRUCTIONS This informed-consent document has been prepared to help inform you about Hylaform (animal-origin, stabilized hyaluronic acid, INAMED) tissue-filler injection therapy, its risks, and alternative
The Use of Restylane in Cosmetic Facial Surgery
 CURRENT THERAPY J Oral Maxillofac Surg 64:317-325, 2006 The Use of Restylane in Cosmetic Facial Surgery Joseph Niamtu, III, DMD* *Private Practice, Oral and Maxillofacial and Cosmetic Facial Surgery, Richmond,
CURRENT THERAPY J Oral Maxillofac Surg 64:317-325, 2006 The Use of Restylane in Cosmetic Facial Surgery Joseph Niamtu, III, DMD* *Private Practice, Oral and Maxillofacial and Cosmetic Facial Surgery, Richmond,
background on restylane skinboosters SB0017_Backgrounder_V04.indd 1 07/03/ :27
 background on restylane skinboosters SB0017_Backgrounder_V04.indd 1 07/03/2013 17:27 Introducing a new concept in skin care Components of the skin Restylane Skinboosters are a brand new approach to nourishing
background on restylane skinboosters SB0017_Backgrounder_V04.indd 1 07/03/2013 17:27 Introducing a new concept in skin care Components of the skin Restylane Skinboosters are a brand new approach to nourishing
Hyaluronic Acid Injections: Incorporating Advanced Microinjection Techniques Into Practice MELANIE D. PALM, MD, MBA, FAAD, FAACS
 Hyaluronic Acid Injections: Incorporating Advanced Microinjection Techniques Into Practice MELANIE D. PALM, MD, MBA, FAAD, FAACS Faculty Information Melanie D. Palm, MD, MBA Medical Director, ART OF SKIN
Hyaluronic Acid Injections: Incorporating Advanced Microinjection Techniques Into Practice MELANIE D. PALM, MD, MBA, FAAD, FAACS Faculty Information Melanie D. Palm, MD, MBA Medical Director, ART OF SKIN
I m living my life in colour. When I fulfil my potential, my personality shines through
 I m living my life in colour When I fulfil my potential, my personality shines through We want to look great, but more importantly, we want to feel great What if you could make a big difference to the
I m living my life in colour When I fulfil my potential, my personality shines through We want to look great, but more importantly, we want to feel great What if you could make a big difference to the
To acclimate skin to AHAs prior to a peel Anti-aging, exfoliation, builds collagen. Gentle cleanser to remove sebum, skin debris and makeup.
 Glycolic Acid Peels and Effective Home Care Products Rejuvenate and Complement the Benefits of Injectable Fillers and Botulinum Toxin Type A in the Treatment of Photoaged David Wrone, MD, Colleen J. Crane,
Glycolic Acid Peels and Effective Home Care Products Rejuvenate and Complement the Benefits of Injectable Fillers and Botulinum Toxin Type A in the Treatment of Photoaged David Wrone, MD, Colleen J. Crane,
A SuppleMeNt to JDD. Not Copy. Correction With a Ribose Cross-linked Collagen Dermal Filler. Penalties Apply
 A SuppleMeNt to JDD Lip Do Augmentation Not Copy and Contour Correction With a Ribose Cross-linked Collagen Dermal Filler ISSN: 1545 9616 March 2009 Volume 8 Issue 3 (Su p p l e m e n t) This supplement
A SuppleMeNt to JDD Lip Do Augmentation Not Copy and Contour Correction With a Ribose Cross-linked Collagen Dermal Filler ISSN: 1545 9616 March 2009 Volume 8 Issue 3 (Su p p l e m e n t) This supplement
Enhancing your appearance with a facelift
 PROCEDURE FACT SHEET PLASTIC SURGERY FACELIFT This is a guide for people who are considering a facelift surgery. We advise that you talk to a plastic surgeon and only use this information as a guide to
PROCEDURE FACT SHEET PLASTIC SURGERY FACELIFT This is a guide for people who are considering a facelift surgery. We advise that you talk to a plastic surgeon and only use this information as a guide to
S UPPLEMENT. Restylane SubQ, a Non-Animal Stabilized Hyaluronic Acid Gel for Soft Tissue Augmentation of the Mid- and Lower Face
 Augmentation of the Mid- and Lower Face Alexis Verpaele, MD; and Anders Strand, MD Dr. Verpaele is Assistant Clinical Professor, Department of Plastic Surgery, Ghent University Hospital and Director, Coupure
Augmentation of the Mid- and Lower Face Alexis Verpaele, MD; and Anders Strand, MD Dr. Verpaele is Assistant Clinical Professor, Department of Plastic Surgery, Ghent University Hospital and Director, Coupure
INFORMED CONSENT: RADIESSE INJECTIONS
 INSTRUCTIONS This is an informed-consent document which has been prepared to help your surgeon inform you concerning a number of available facial tissue filler injection therapies, their risks, and alternative
INSTRUCTIONS This is an informed-consent document which has been prepared to help your surgeon inform you concerning a number of available facial tissue filler injection therapies, their risks, and alternative
SCULPTRA INJECTIONS. New Age Medical Clinic 90 Millburn Ave., Suite 201 Millburn NJ (973) Dr Maria Romanenko, DO
 SCULPTRA INJECTIONS New Age Medical Clinic 90 Millburn Ave., Suite 201 Millburn NJ 07041 (973) 313-0028 Dr Maria Romanenko, DO Board Certified: Family Medicine & Aesthetics FREE CONSULTATION Original Contribution
SCULPTRA INJECTIONS New Age Medical Clinic 90 Millburn Ave., Suite 201 Millburn NJ 07041 (973) 313-0028 Dr Maria Romanenko, DO Board Certified: Family Medicine & Aesthetics FREE CONSULTATION Original Contribution
FACTS. about MemoryGel silicone gel-filled breast implants
 FACTS about MemoryGel silicone gel-filled breast implants Are you considering breast implant surgery but not certain which type of implant to choose? YOU RE NOT ALONE. Science-based information to empower
FACTS about MemoryGel silicone gel-filled breast implants Are you considering breast implant surgery but not certain which type of implant to choose? YOU RE NOT ALONE. Science-based information to empower
INFORMED CONSENT RADIESSE INJECTION
 Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and reproduce the modified version for use in the Purchaser's own practice only. All
Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and reproduce the modified version for use in the Purchaser's own practice only. All
Soft Tissue Augmentation
 Chapter Soft Tissue Augmentation Core Messages Facial aging is a growing concern among individuals in their 30s, 40s, and 50s and is driving increased demand for new products and techniques. Soft tissue
Chapter Soft Tissue Augmentation Core Messages Facial aging is a growing concern among individuals in their 30s, 40s, and 50s and is driving increased demand for new products and techniques. Soft tissue
Injectable Tissue Filler Consent
 Injectable Tissue Filler Consent Fillers are injectable gel is a colorless hyaluronic acid gel that is injected into facial tissue to smooth wrinkles and folds, especially around the nose and mouth. Hyaluronic
Injectable Tissue Filler Consent Fillers are injectable gel is a colorless hyaluronic acid gel that is injected into facial tissue to smooth wrinkles and folds, especially around the nose and mouth. Hyaluronic
INFORMED CONSENT HYLAFORM INJECTION
 2009 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and reproduce the modified version for use
2009 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein and reproduce the modified version for use
A NON-SURGICAL SKIN REJUVENATION TREATMENT
 A NON-SURGICAL SKIN REJUVENATION TREATMENT WHAT IS MESOFILLING? Mesofilling is a new concept of injectable treatment between dermal filling and mesotherapy. Mesotherapy, a safe, effective and established
A NON-SURGICAL SKIN REJUVENATION TREATMENT WHAT IS MESOFILLING? Mesofilling is a new concept of injectable treatment between dermal filling and mesotherapy. Mesotherapy, a safe, effective and established
Dermal Fillers Information Guide
 Professionally trained in aesthetic procedures Dermal Fillers Information Guide What are Dermal Fillers? As the skin ages, it gradually loses some of its collagen and fat. These are the things that prevent
Professionally trained in aesthetic procedures Dermal Fillers Information Guide What are Dermal Fillers? As the skin ages, it gradually loses some of its collagen and fat. These are the things that prevent
Module 3: Injectable Dermal Fillers
 MODULE OVERVIEW Module 3: Injectable Dermal Fillers (2-day) consists of didactic & live patient hands-on training and examinations. This certification course is delivered in a small class size and high
MODULE OVERVIEW Module 3: Injectable Dermal Fillers (2-day) consists of didactic & live patient hands-on training and examinations. This certification course is delivered in a small class size and high
Hydryalix Advantages. Composition. Hydryalix
 2409 Hydryalix Hydryalix (Gentle / Volume / Deep / Ultra Deep / Lips) is an injectable, sterile, apyrogenic gel intended for soft tissue augmentation. It is composed of cross-linked hyaluronic acid from
2409 Hydryalix Hydryalix (Gentle / Volume / Deep / Ultra Deep / Lips) is an injectable, sterile, apyrogenic gel intended for soft tissue augmentation. It is composed of cross-linked hyaluronic acid from
Skin Boosting Technique: A New Approach to Hyaluronic Acid Microinjection
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
INFORMED CONSENT JUVÉDERM ULTRA/ULTRA PLUS FILLER INJECTION
 INSTRUCTIONS This is an informed-consent document which has been prepared to help Dr. Rothfield inform you concerning Juvederm -based (Non-Animal Stabilized) tissue filler injection therapy, its risks,
INSTRUCTIONS This is an informed-consent document which has been prepared to help Dr. Rothfield inform you concerning Juvederm -based (Non-Animal Stabilized) tissue filler injection therapy, its risks,
INFORMED CONSENT FOR FILLER INJECTION BELLAFILL BELOTERO PRODUCTS JUVEDERM PRODUCTS RADIESSE RESTYLANE PRODUCTS SCULPTRA
 INFORMED CONSENT FOR FILLER INJECTION BELLAFILL BELOTERO PRODUCTS JUVEDERM PRODUCTS RADIESSE RESTYLANE PRODUCTS SCULPTRA (PLEASE REVIEW AND BRING WITH YOU ON THE DAY OF YOUR PROCEDURE) PATIENT NAME KAROL
INFORMED CONSENT FOR FILLER INJECTION BELLAFILL BELOTERO PRODUCTS JUVEDERM PRODUCTS RADIESSE RESTYLANE PRODUCTS SCULPTRA (PLEASE REVIEW AND BRING WITH YOU ON THE DAY OF YOUR PROCEDURE) PATIENT NAME KAROL
balt6/zkw-iop/zkw-iop/zkw00208/zkw z xppws S 1 1/28/08 11:47 4/Color Figure(s): F1 3 Art: IOP Input-GA
 Ophthalmic Plastic and Reconstructive Surgery Vol. 24, No. 2, pp?????? 2008 The American Society of Ophthalmic Plastic and Reconstructive Surgery, Inc. AQ: A Patient-Preferred Sites of Restylane Injection
Ophthalmic Plastic and Reconstructive Surgery Vol. 24, No. 2, pp?????? 2008 The American Society of Ophthalmic Plastic and Reconstructive Surgery, Inc. AQ: A Patient-Preferred Sites of Restylane Injection
Review of soft tissue augmentation in the face
 Clinical, Cosmetic and Investigational Dermatology open access to scientific and medical research Open Access Full Text Article Review of soft tissue augmentation in the face r e v i e w James Newman Facial
Clinical, Cosmetic and Investigational Dermatology open access to scientific and medical research Open Access Full Text Article Review of soft tissue augmentation in the face r e v i e w James Newman Facial
TEOSYAL PEN: Personal experience after 12 months on 285 consecutive patients
 TEOSYAL PEN: Personal experience after 12 months on 285 consecutive patients Dr. Dell Avanzato Roberto AMWC 2016, 14 th Aesthetic & Anti-aging Medicine World Congress 31 March, 1 2 April, 2016 BACKGROUND
TEOSYAL PEN: Personal experience after 12 months on 285 consecutive patients Dr. Dell Avanzato Roberto AMWC 2016, 14 th Aesthetic & Anti-aging Medicine World Congress 31 March, 1 2 April, 2016 BACKGROUND
Lisa Chipps, MD, MS, FAAD Assistant Clinical Professor David Geffen School of Medicine at UCLA
 Lisa Chipps, MD, MS, FAAD Assistant Clinical Professor David Geffen School of Medicine at UCLA Presented at The American Academy of Facial Plastic and Reconstructive Surgery Meeting, September, 2011 Provided
Lisa Chipps, MD, MS, FAAD Assistant Clinical Professor David Geffen School of Medicine at UCLA Presented at The American Academy of Facial Plastic and Reconstructive Surgery Meeting, September, 2011 Provided
-5 th Generation- Poly-L-Lactic Acid. Thread Lifting
 PLLA -5 th Generation- Poly-L-Lactic Acid Thread Lifting Careis What is PLLA? 5 th Generation of polymer suture extracted from plants A type of AHA (Alpha-Hydroxy Acid) Originally developed and marketed
PLLA -5 th Generation- Poly-L-Lactic Acid Thread Lifting Careis What is PLLA? 5 th Generation of polymer suture extracted from plants A type of AHA (Alpha-Hydroxy Acid) Originally developed and marketed
Module 1. Introduction to Aesthetic Medicine: Nonsurgical
 Module 1 Introduction to Aesthetic Medicine: Nonsurgical What is aesthetic medicine? Well really it s about treatments, whether it be nonsurgical or surgical, to reshape normal structures of one s body
Module 1 Introduction to Aesthetic Medicine: Nonsurgical What is aesthetic medicine? Well really it s about treatments, whether it be nonsurgical or surgical, to reshape normal structures of one s body
INJECTING SCIENCE INTO YOUR BEAUTY
 INJECTING SCIENCE INTO YOUR BEAUTY REJUVENATE YOUR NATURAL BEAUTY WITH INSTANT, LASTING RESULTS PLURYAL MD SKIN SOLUTIONS LABORATORIES, THE CREATORS BEHIND BEAUTY SCIENCE UNDERSTANDING SKIN AGING > Due
INJECTING SCIENCE INTO YOUR BEAUTY REJUVENATE YOUR NATURAL BEAUTY WITH INSTANT, LASTING RESULTS PLURYAL MD SKIN SOLUTIONS LABORATORIES, THE CREATORS BEHIND BEAUTY SCIENCE UNDERSTANDING SKIN AGING > Due
NATIONAL CLEARINGHOUSE
 AMERICAN SOCIETY OF PLASTIC SURGEONS 2009 REPORT of the STATISTICS NATIONAL CLEARINGHOUSE of Plastic Surgery Statistics Established 1931. The Symbol of Excellence in Plastic Surgery Phone 847-228-9900
AMERICAN SOCIETY OF PLASTIC SURGEONS 2009 REPORT of the STATISTICS NATIONAL CLEARINGHOUSE of Plastic Surgery Statistics Established 1931. The Symbol of Excellence in Plastic Surgery Phone 847-228-9900
New Filler Approvals Refyne, Defyne, Vollure, Revanesse. Karol A Gutowski, MD, FACS Hot Topics
 New Filler Approvals Refyne, Defyne, Vollure, Revanesse Karol A Gutowski, MD, FACS Hot Topics Disclosures Merz - Advisory Board Suneva Medical - Instructor Will use brand names due to lack of distinguishing
New Filler Approvals Refyne, Defyne, Vollure, Revanesse Karol A Gutowski, MD, FACS Hot Topics Disclosures Merz - Advisory Board Suneva Medical - Instructor Will use brand names due to lack of distinguishing
Rejuvenating Effects of Facial Hydrofilling using Restylane Vital
 Rejuvenating Effects of Facial Hydrofilling using Restylane Vital Original Article Bong Moo Lee 1, Dong Gil Han 1, Won Seok Choi 2 1 Department of Plastic and Reconstructive Surgery, Catholic University
Rejuvenating Effects of Facial Hydrofilling using Restylane Vital Original Article Bong Moo Lee 1, Dong Gil Han 1, Won Seok Choi 2 1 Department of Plastic and Reconstructive Surgery, Catholic University
The Authoritative Source Current US Statistics on Cosmetic Surgery. Expanded data for 2007: Multi-year comparisons, 39 Cosmetic Procedures
 2 0 0 7 The American Society for Aesthetic Plastic Surgery The Authoritative Source Current US Statistics on Cosmetic Surgery 1997-2007 Cosmetic Surgery National Data Bank Statistics Expanded data for
2 0 0 7 The American Society for Aesthetic Plastic Surgery The Authoritative Source Current US Statistics on Cosmetic Surgery 1997-2007 Cosmetic Surgery National Data Bank Statistics Expanded data for
SkinCare. BodyShaping HairLoss JointsArthritis. best-care by your-cells
 SkinCare BodyShaping HairLoss JointsArthritis best-care by your-cells Beauty and Vitality are gifts from nature for those who live according to her laws (Leonardo da Vinci) Aging Skin Care face, neck,
SkinCare BodyShaping HairLoss JointsArthritis best-care by your-cells Beauty and Vitality are gifts from nature for those who live according to her laws (Leonardo da Vinci) Aging Skin Care face, neck,
PERFECT FILLERS TECHNIQUES FOR PHYSICIANS SWISS QUALITY
 PERFECT FILLERS TECHNIQUES FOR PHYSICIANS SWISS QUALITY OUR MISSION Suisselle is a privately owned company located in a biotech hub, Yverdon-les-Bains, Switzerland. Operating to the highest standard of
PERFECT FILLERS TECHNIQUES FOR PHYSICIANS SWISS QUALITY OUR MISSION Suisselle is a privately owned company located in a biotech hub, Yverdon-les-Bains, Switzerland. Operating to the highest standard of
Redensity Innovation in eye circle treatment
 T H E B E S T O F H Y A L U R O N I C A C I D presents Innovation in eye circle treatment As experts in aesthetic medicine and specialising in creating and manufacturing hyaluronic acid dermal fillers
T H E B E S T O F H Y A L U R O N I C A C I D presents Innovation in eye circle treatment As experts in aesthetic medicine and specialising in creating and manufacturing hyaluronic acid dermal fillers
Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle
 Clinical Study: Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle Luc Dewandre MD 1 ; Caroline Caperton MD, MSPH 2 ; James Fulton MD, PhD 2 1 Paris France; 2 University
Clinical Study: Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle Luc Dewandre MD 1 ; Caroline Caperton MD, MSPH 2 ; James Fulton MD, PhD 2 1 Paris France; 2 University
Patient Information Leaflet. Dermal Filler
 Patient Information Leaflet Dermal Filler When considering treatment with dermal fillers we want you to have a safe treatment. Some risks are unavoidable and out of your control. The following information
Patient Information Leaflet Dermal Filler When considering treatment with dermal fillers we want you to have a safe treatment. Some risks are unavoidable and out of your control. The following information
Rama Univ J Dent Sci 2016 Dec;3(4):27-31.
 Review Article Lip Rejuvenation- An Overview of Various Filler Materials and Techniques. Kumar V, Thapliyal GK, Singh N, Raj A, Choube A, Mehta KA Abstract: The lips enhance facial beauty and functionally
Review Article Lip Rejuvenation- An Overview of Various Filler Materials and Techniques. Kumar V, Thapliyal GK, Singh N, Raj A, Choube A, Mehta KA Abstract: The lips enhance facial beauty and functionally
Fillers- Post Treatment Information
 Fillers- Post Treatment Information Filler injections are a non-surgical procedure used to temporarily restore facial volume and help diminish folds and concavities. Common fillers used in our practice
Fillers- Post Treatment Information Filler injections are a non-surgical procedure used to temporarily restore facial volume and help diminish folds and concavities. Common fillers used in our practice
Artecoll: A Long-Lasting Injectable Wrinkle Filler Material: Report of a Controlled, Randomized, Multicenter Clinical Trial of 251 Subjects
 Cosmetic Artecoll: A Long-Lasting Injectable Wrinkle Filler Material: Report of a Controlled, Randomized, Multicenter Clinical Trial of 251 Subjects Foot Steven R. Cohen, M.D., and Ralph E. Holmes, M.D.
Cosmetic Artecoll: A Long-Lasting Injectable Wrinkle Filler Material: Report of a Controlled, Randomized, Multicenter Clinical Trial of 251 Subjects Foot Steven R. Cohen, M.D., and Ralph E. Holmes, M.D.
WHEN FRENCH TOUCH MEETS BEAUTY
 WHEN FRENCH TOUCH MEETS BEAUTY , MADE IN FRANCE BY SINCLAIR is manufactured 100% in France by SINCLAIR PHARMA in its laboratory specialized in the research, development and manufacturing of Hyaluronic
WHEN FRENCH TOUCH MEETS BEAUTY , MADE IN FRANCE BY SINCLAIR is manufactured 100% in France by SINCLAIR PHARMA in its laboratory specialized in the research, development and manufacturing of Hyaluronic
WONDERFUL SKIN. SB_202731_Skinboosters_Consumer Guide A5_V03.indd 1 20/02/ :28
 WONDERFUL SKIN SB_202731_Skinboosters_Consumer Guide A5_V03.indd 1 20/02/2013 13:28 Show your skin at its best You are wonderful and your skin should reflect your natural wonders within. That s why your
WONDERFUL SKIN SB_202731_Skinboosters_Consumer Guide A5_V03.indd 1 20/02/2013 13:28 Show your skin at its best You are wonderful and your skin should reflect your natural wonders within. That s why your
Semipermanent and Permanent Injectable Fillers
 Semipermanent and Permanent Injectable Fillers Derek H. Jones, MD KEYWORDS Semipermanent and permanent fillers Hyaluronic acid Calcium hyaluronic polylactic acid Polymethyl methacrylate Soft-tissue augmentation
Semipermanent and Permanent Injectable Fillers Derek H. Jones, MD KEYWORDS Semipermanent and permanent fillers Hyaluronic acid Calcium hyaluronic polylactic acid Polymethyl methacrylate Soft-tissue augmentation
Statistics. The American Society for Aesthetic Plastic Surgery. Cosmetic Surgery National Data Bank
 The American Society for Aesthetic Plastic Surgery The Authoritative Source Current US Statistics on Cosmetic Surgery Expanded data for 2006: Ten year comparisons, 34 Cosmetic Procedures Multi-specialty
The American Society for Aesthetic Plastic Surgery The Authoritative Source Current US Statistics on Cosmetic Surgery Expanded data for 2006: Ten year comparisons, 34 Cosmetic Procedures Multi-specialty
A brighter smile. A younger looking you.
 A brighter smile. A younger looking you. Facial cosmetic treatments, DENTO-FACIAL AESTHETICS delivered by your dentist. AVAILABLE EXCLUSIVELY TO KEYS DENTAL PATIENTS ONLY COSMETIC NON-SURGICAL REJUVENATION
A brighter smile. A younger looking you. Facial cosmetic treatments, DENTO-FACIAL AESTHETICS delivered by your dentist. AVAILABLE EXCLUSIVELY TO KEYS DENTAL PATIENTS ONLY COSMETIC NON-SURGICAL REJUVENATION
Hasson & Wong Lateral Slit Technique in Hair Transplantation Natural hair transplant results, minimized scarring
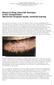 Hasson & Wong Lateral Slit Technique in Hair Transplantation Natural hair transplant results, minimized scarring The evolution of follicular unit hair transplants, which involves transplanting hair in
Hasson & Wong Lateral Slit Technique in Hair Transplantation Natural hair transplant results, minimized scarring The evolution of follicular unit hair transplants, which involves transplanting hair in
Refresh, Renew Rejuvenate Look years younger, with minimum downtime. The Quick-Recovery Facelift
 Refresh, Renew Rejuvenate Look years younger, with minimum downtime. The Quick-Recovery Facelift Discover How Easy Looking Younger Can Be. We have pioneered an exciting new facelift procedure that offers
Refresh, Renew Rejuvenate Look years younger, with minimum downtime. The Quick-Recovery Facelift Discover How Easy Looking Younger Can Be. We have pioneered an exciting new facelift procedure that offers
Evaluation of human allogeneic collagen gel for correction of nasolabial folds using non-invasive measurement techniques
 Original Article Evaluation of human allogeneic collagen gel for correction of nasolabial folds using non-invasive measurement techniques Mansour Nassiri Kashani, MD 1 Ali Rajabi Estarabadi, MD 1 Hamed
Original Article Evaluation of human allogeneic collagen gel for correction of nasolabial folds using non-invasive measurement techniques Mansour Nassiri Kashani, MD 1 Ali Rajabi Estarabadi, MD 1 Hamed
FACETITE: SUBDERMAL RADIOFREQUENCY SKIN TIGHTENING AND FACE CONTOURING
 FACETITE: SUBDERMAL RADIOFREQUENCY SKIN TIGHTENING AND FACE CONTOURING R. Stephen Mulholland, MD, FRCS(C)* and Michael Kreindel, PhD** *Private Plastic Surgery Practice, Toronto, Canada ** Chief Technology
FACETITE: SUBDERMAL RADIOFREQUENCY SKIN TIGHTENING AND FACE CONTOURING R. Stephen Mulholland, MD, FRCS(C)* and Michael Kreindel, PhD** *Private Plastic Surgery Practice, Toronto, Canada ** Chief Technology
BREAST RECONSTRUCTION
 BREAST RECONSTRUCTION YOUR OPTIONS FOR BREAST RECONSTRUCTION SURGERY The decision to pursue breast reconstruction is personal and your options vary based on your personal and medical history. This resource
BREAST RECONSTRUCTION YOUR OPTIONS FOR BREAST RECONSTRUCTION SURGERY The decision to pursue breast reconstruction is personal and your options vary based on your personal and medical history. This resource
Dermal fillers have steadily grown in use over the. Juvéderm Injectable Gel: A Multicenter, Double-Blind, Randomized Study of Safety and Effectiveness
 Juvéderm Injectable Gel: A Multicenter, Double-Blind, Randomized Study of Safety and Effectiveness Mark A. Pinsky, MD; Jane A. Thomas, AAS, CCRA; Diane K. Murphy, MBA; and Patricia S. Walker, MD, PhD;
Juvéderm Injectable Gel: A Multicenter, Double-Blind, Randomized Study of Safety and Effectiveness Mark A. Pinsky, MD; Jane A. Thomas, AAS, CCRA; Diane K. Murphy, MBA; and Patricia S. Walker, MD, PhD;
Society for Aesthetic. Cosmetic Surgery. National Data Bank. The Authoritative Source for Current US Statistics on.
 The American Society for Aesthetic Plastic Surgery 2005 0 Cosmetic Surgery National Data Bank Statistics The Authoritative Source for Current US Statistics on Cosmetic Surgery Expanded data for 2005: Nine
The American Society for Aesthetic Plastic Surgery 2005 0 Cosmetic Surgery National Data Bank Statistics The Authoritative Source for Current US Statistics on Cosmetic Surgery Expanded data for 2005: Nine
WHAT IS SILHOUETTE SOFT?
 WHAT IS SILHOUETTE SOFT? A technological innovation serving rejuvenation When women are asked what they consider their two main signs of facial ageing, the answer is invariably loss of skin tone causing
WHAT IS SILHOUETTE SOFT? A technological innovation serving rejuvenation When women are asked what they consider their two main signs of facial ageing, the answer is invariably loss of skin tone causing
Clinical studies with patients have been carried out on this subject of graft survival and out of body time. They are:
 Study Initial Date: July 21, 2016 Data Collection Period: Upon CPHS Approval to September 30, 2018 Study Protocol: Comparison of Out of Body Time of Grafts with the Overall Survival Rates using FUE Lead
Study Initial Date: July 21, 2016 Data Collection Period: Upon CPHS Approval to September 30, 2018 Study Protocol: Comparison of Out of Body Time of Grafts with the Overall Survival Rates using FUE Lead
