Policy: C-08-PRO Reviewed: 7/08
|
|
|
- Barry Wells
- 6 years ago
- Views:
Transcription
1 Written: 1979 Policy: Reviewed: 7/08 Procedure Policy Manual Revised: 81, 84, 85, 86, 87, 88, 89, 90, 92, 93, 95 Ambulatory Care Division 2/97, 7/98, 7/00, 7/02, 7/04, 7/06, 2/09, 7/10 LSU Health Sciences Center-Shreveport, LA 8/10, 5/13 Page 1 of 5 PURPOSE CIDEX (GLUTERALDEHYDE) and CIDEX OPA (ORTHOPHTHALAHYDE) HIGH LEVEL DISINFECTANT USED FOR INSTRUMENTS To provide guidelines for high level disinfection of ultrasound probes, scopes, and other instruments approved by the Infection Control Committee for processing in Ambulatory Care Clinics. POLICY 1. The following outpatient clinics are approved to use glutaraldehyde 2.4% (Cidex Activated Dialdehyde Solution) or ortho-phthalahyde 0.55% (Cidex OPA) for high level disinfection of the specified instruments: Clinics/Room Endoscopy (B3-4) Urology (3036) ENT (3086) Feist-Weiller Cancer Center (B334, B anteroom) Women & Children Health Center (2-170) Radiology ACB (1192) Usage- For High Level Disinfection of Gastric motility probes and balloons, flexible endoscopes that cannot be processed in Steris. Rectal ultrasound probes Nasopharyngeal scopes Panendoscopes, nasal scopes and laryngeal scopes Vaginal ultrasound probes Ultrasound probes 2. Only trained staff with documented competency are allowed to perform high level disinfection using glutaraldehyde or ortho-phthalahyde solutions. Annual competency is documented by the unit manager. 3. Glutaraldehyde and ortho-phthalahyde are kept in a controlled area. Containers are kept covered or closed to prevent fumes from disseminating into the air. 1
2 Cidex Activated Dialdehyde Solution/Cidex OPA High Level Disinfectant Used for Instruments Page 2 of 5 PROCEDURE Preparing Glutaraldehyde or Ortho-phthadahyde 1. Obtain a gallon bottle of solution. 2. Put on PPE (eye protection, mask, gloves, and gown). 3. Read and prepare the solution according to the manufacturer s directions on the label. (Glutaraldehyde requires mixing, ortho-phthalahyde does not.) 4. Carefully pour solution into an approved soak container. Do not splash or spill. Do not breathe the vapors. Replace the bottle cap. Place a lid on the soak container. 5. Label the soak container with the chemical solution, date of expiration, and initials. (If pouring from a previously opened bottle, check the expiration date on the original container. Solution in soak basins expires in 14 days or upon the expiration date of the original container, whichever comes first.) 6. If left over solution remains in the bottle, label the bottle with date of expiration and initials. Cidex 2.4% that remains in the bottle is good for only 14 days once activated. Cidex OPA that remains in the bottle is good for 75 days once opened. Quality Assurance for Test Strips When a new bottle of test strips is opened, perform the following quality assurance test. Ensure that the test strips you are using match the chemical (Cidex 2.4% test strips for Cidex 2.4%; Cidex OPA test strips for Cidex OPA). 1. Check the expiration date on the test strip bottle. If expired, throw away and obtain a new bottle. 2. Check the chemical solution you are using to be sure that the expiration date has not been exceeded. If it has been exceeded, do not use it, and obtain a new bottle of the chemical. 3. Prepare a control solution by diluting one part of full strength chemical solution to one part of tap water. 4. Dip 3 strips in the diluted solution and 3 strips in the full strength solution in accordance with the time specified on the bottle. Remove from the solution, stand on a paper towel, and read at the time specified on the bottle. 5. The 3 strips dipped in the full strength solution should change to the passing color on the bottle. 6. The 3 strips dipped in the diluted solution should match the fail color or not change color. 7. If your results differ from those described in number 4 and 5 above, discard the test strips, obtain a new bottle of test strips, and repeat the Quality Assurance test. 8. Document the results on the log sheet. 9. If more than one bottle fails the Quality Assurrance test, contact Infection Control. 2
3 Cidex Activated Dialdehyde Solution/Cidex OPA High Level Disinfectant Used for Instruments Page 3 of 5 Testing Glutaraldehyde/Orthophthalahyde for Efficacy 1. Glutaraldehyde/orthophthalahyde must be tested for efficacy before each use. 2. Before soaking any instrument: A. Put on a pair of unsterile gloves and eye protection. B. Check the expiration date on the soak basin to ensure that the expiration date has not been exceeded. If the expiration date has been exceeded, discard the solution. C. Check the bottle of test strips to ensure that you are using test strips that match the chemical and concentration you are testing, and to ensure that the expiration date has not been exceeded. If the test strips are expired, discard the bottle. Check the date the test strip bottle was opened. Discard any unused test strips after the bottle has been opened for 90 days. If opening the test strip bottle for the first time, place the date you opened it on the outside of the bottle and initial. Do not leave the test strip bottle open for more than 30 minutes. D. Remove a test strip from the bottle and replace the cap tightly. E. Read and follow directions on the bottle. Dip the indicating pad of the test strip into the soak solution. Follow test strip bottle directions and ensure that the strip is exposed to the chemical for the correct amount of time. F. DO NOT shake the test strip after removal from the solution. G. Remove excess solution by standing the strip upright on a paper towel. Discard the paper towel after one use. H. Read the results at the appropriate time interval according to label directions. I. If the color comparison matches the color for effective dilution of the high level disinfectant solution, record this in the log book and proceed with instructions for disinfection. J. If the color comparison does not match the results for effective dilution, record this result, and discard the solution. (Follow instructions below for discarding solution.) Cleaning and Decontaminating Instruments Prior to Disinfection Carefully measure enzyme cleaner and water, mix according to manufacturer s label instructions prior to beginning the cleaning process. 1. Instruments must be thoroughly cleaned and decontaminated prior to disinfection. 2. Thoroughly clean the instrument in accordance with the manufacturer s directions. 3. Wipe gel, blood, body fluids, or other debris off of ultrasound probes or scopes using clean 4 x 4 s or clean towel soaked in enzyme cleaner. Discard the 4 x 4 s or towel after one use. 4. If instruments have lumens or channels, they must be opened, caps removed, and thoroughly cleaned with enzyme cleaner. Channels must be brushed, and disposable channel brushes discarded after use on one instrument. 5. After cleaning with enzyme cleaner, rinse the instruments thoroughly, and dry with a clean towel or 4x4 s. Discard the towel or 4x4 s after one use. 6. Instrument lumens or channels should be blown dry with forced air. 3
4 Cidex Activated Dialdehyde Solution/Cidex OPA High Level Disinfectant Used for Instruments Page 4 of 5 Disinfection 1. Check the temperature of the solution and record on the log. If temperature is too cold, warm the room and test again. Acceptable temperatures are: Cidex 2.4% Activated Dialdehyde Solution: 68 F (20 C) or higher for a minimum soak of 20 minutes* Cidex OPA: 68 F (20 C) or higher for a minimum soak of 12 minutes* *Some departments may specify longer soak times. Check and follow your department policy. 2. Place clean, dry instrument into the soak basin. Ensure that the item is completely covered with the solution. If the instrument has inner lumens or channels, ensure that they are filled with the solution by injecting with a syringe. Replace the container lid. Remove and discard soiled gloves and gown. Wash your hands. 3. Record the time the instrument was placed in the solution as the start time or use a timer. The approved minimum soak time for glutaraldehye 2.4% is 20 minutes. Approved soak time for Cidex OPA is 12 minutes. The soak container should be labeled with the time the soak is complete or a timer should be used. 4. After the appropriate soak time has passed, prepare to rinse the instruments. Check to ensure that the soak time is complete. Wash your hands. 5. Open a sterile soak pan and sterile water. Pour sterile water into the soak pan. (Vaginal and rectal ultrasound probes may be rinsed in a clean soak pan using tap water or using copious amounts of running tap water.) 6. Put on a mask and eye protection. 7. Carefully remove the lid of the soak container and set aside. Put on a clean gown and wear sterile gloves. Remove the instrument from the soak solution, using care not to contaminate the instrument by touching contaminated surfaces or sides of the soak container. 8. Place the instrument in the basin of water. Rinse three times thoroughly to remove all traces of soak solution inside and outside of the instrument. Blot dry with sterile surgical towels or 4X4s. 9. If the instrument is lumened or channeled, use a syringe to force sterile water through the lumens, followed by purging the channels with 70% isopropyl alcohol, then dry with forced air. 10. Wrap the instrument in a clean towel for immediate use. Wrap bronchoscopes in a sterile towel. If the item is to be stored, place it in a clean container or bag, and hang long flexible scopes to dry in such a manner that the tip of the scope does not touch the bottom of the cabinet. 11. Replace the lid on the soak pan. Empty rinse basins after one use, dry, and send to CMS for cleaning and sterilization. If disposable rinse basins are used, discard after use on one instrument. 12. Remove and discard gown and gloves. 13. Keep the door to the processing room closed as much as possible. 4
5 Cidex Activated Dialdehyde Solution/Cidex OPA High Level Disinfectant Used for Instruments Page 5 of 5 Discarding Glutaraddehyde/Orthophthalahyde 1. Put on a mask, eye protection, clean gloves, and gown. 2. Place the discard bucket near the soak container. 3. Open the lid of the discard bucket and the lid of the soak container. 4. Carefully lift the soak container and pour the contents into the discard bucket, using care not to splash or spill the solution. 5. Place the lid on the discard container and, when ¾ full, call the Safety Office at extension If a spill occurs-first, contain the spill by placing cloth or paper towels to absorb it. Then, immediately call the safety office at extension Rinse the empty soak basin carefully in the sink using tap water, dry with clean towels, and send to Central Medical Supply for further cleaning and sterilization. Discard the towels after one use. 5
Cleaning and Disinfection Protocol for Emergency Services Fire, Ambulance, Police, Search & Rescue
 This document has been developed in accordance with current applicable infection control and regulatory guidelines. It is intended for use as a guideline only. At no time should this document replace existing
This document has been developed in accordance with current applicable infection control and regulatory guidelines. It is intended for use as a guideline only. At no time should this document replace existing
Package Leaks. OH&S Biosafety Emergency Response Document. Examine outer packaging. Leaks or evidence of leaks. No evidence of leaks
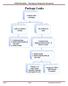 Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Package Leaks Examine outer packaging Leaks or evidence of leaks No evidence of leaks 1. Contain package 2. Notify UAB Biosafety @ 934-2487 3. Notify Sender Examine inner pkging/contents in BSC - if there
Emergency Procedures Specific Biological Spill Clean-Up Guidelines
 Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
Emergency Procedures 3.1.1. Biological Spills Spill kit materials and written procedures shall be kept in each laboratory where work with microorganisms is conducted. Basic equipment includes concentrated
Sterilization A Training Module
 Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
Sterilization A Training Module In This Training Module, You Will Learn: Definition of sterilization and disinfection What needs to be sterilized and disinfected What Personal Protective Equipment is required
What is infection control?
 Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
Infection control What is infection control? It is the discipline concerned with preventing healthcareassociated infection. It is an essential part of the infrastructure of health care. Standard principles
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION
 State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
State of Kuwait Ministry of Health Infection Control Directorate SAFE INJECTION May 2010 Contents I. Introduction II. Prevention strategies III. Best practices for injection A. General safety practices
Table 6: Detailed Infection Prevention and Control Procedures for Tattooing and Micropigmentation. Use During Tattooing
 FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
FACT SHEET Table 6: Detailed Infection Prevention and Control Procedures for and Micropigmentation 1. Skin Preparation Spray bottle with a solution of soap and water Single use disposable razor The skin
BSL-2 Emergency Plan
 BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
BSL-2 Emergency Plan Spills General Spill Cleanup Guidelines: Know how to get the HVAC unit servicing the lab space shut down in order to limit the spread of contamination. Wear gloves and lab coat. Use
A SAFE ANDCONNECTED APPROACH
 Endoscopy Solutions A SAFE ANDCONNECTED APPROACH TO ENDOSCOPE REPROCESSING One Integrated Approach to Healthcare. The Revital-Ox Difference. When you reprocess your endoscopy devices using the Revital-Ox
Endoscopy Solutions A SAFE ANDCONNECTED APPROACH TO ENDOSCOPE REPROCESSING One Integrated Approach to Healthcare. The Revital-Ox Difference. When you reprocess your endoscopy devices using the Revital-Ox
Hand Hygiene. Policy Title: Hand Hygiene Policy Number: 05. Effective Date: 6/10/2013 Review Date: 6/10/2016
 Hand Hygiene 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in hand hygiene. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings. 2.2. To promote awareness for
Hand Hygiene 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in hand hygiene. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings. 2.2. To promote awareness for
BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN GUIDELINE
 Ventura County Environmental Health Division 800 S. Victoria Ave., Ventura CA 93009-1730 TELEPHONE: 805/654-5007 FAX: 805/477-1595 Internet Web Site Address: https://vcrma.org/body-art-program BODY ART
Ventura County Environmental Health Division 800 S. Victoria Ave., Ventura CA 93009-1730 TELEPHONE: 805/654-5007 FAX: 805/477-1595 Internet Web Site Address: https://vcrma.org/body-art-program BODY ART
SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL
 ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
ENVIRONMENTAL AND EMERGENCY MANAGEMENT Environmental Health and Safety University Crossing Suite 140 Lowell MA 01854 http://www.uml.edu/eem/ SOP BIO-002 FOR SHARPS USAGE AND DISPOSAL SCOPE This policy
Body Art Facility Infection Prevention And Control Plan Guideline
 Body Art Facility Infection Prevention And Control Plan Guideline In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection
Body Art Facility Infection Prevention And Control Plan Guideline In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection
#74 - CHANGING A MOIST TO DRY DRESSING (TEST)
 #74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
#74 - CHANGING A MOIST TO DRY DRESSING (TEST) I acknowledge I have physically practiced and successfully learned the following skill(s): Student: Date: TIME LIMIT:30 Minutes TEST INCLUDES SKILLS FROM #56,
MSDS WRITTEN PLAN REQUIREMENTS. Written Plan Education & Training. Site Specific Updated Annually Training. Material Safety Data Sheet.
 The Hazard Communication Standard can be found in the U.S. Code of Federal Regulations 29CFR1910.1200. The standard is available online at http://www.oshaslc.gov/oshstd_toc/osh A_Std_toc_1910_SUBP ART_Z.html.
The Hazard Communication Standard can be found in the U.S. Code of Federal Regulations 29CFR1910.1200. The standard is available online at http://www.oshaslc.gov/oshstd_toc/osh A_Std_toc_1910_SUBP ART_Z.html.
WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL
 WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL Part of White Woods Academy Trust BODILY FLUID HYGIENE POLICY Approved by Governors: September 2017 Review Date: September 2019 Statement of intent At Whiston
WHISTON WORRYGOOSE JUNIOR AND INFANT SCHOOL Part of White Woods Academy Trust BODILY FLUID HYGIENE POLICY Approved by Governors: September 2017 Review Date: September 2019 Statement of intent At Whiston
Ventricular Assist Device (VAD) Exit Site Care Guidelines
 Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
Ventricular Assist Device (VAD) Exit Site Care Guidelines I. STATEMENT OF PURPOSE The Ventricular Assist Device (VAD) Exit Site Care Guidelines are intended to provide standardization and continuity of
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860. Effective Date: August 31, 2006
 COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
COALINGA STATE HOSPITAL NURSING POLICY AND PROCEDURE MANUAL SECTION Infection Control POLICY NUMBER: 860 Effective Date: August 31, 2006 SUBJECT: (INFECTIOUS) WASTE This cancels Nursing Procedure 860 dated
Current Status: Active PolicyStat ID: Original Policy: 10/1986 Last Reviewed: 01/2016 Last Revised: 01/2016 Next Review: 01/2019
 Current Status: Active PolicyStat ID: 2085666 Original Policy: 10/1986 Last Reviewed: 01/2016 Last Revised: 01/2016 Next Review: 01/2019 Owner: Policy Area: References: Applicability: Tracy Dodson: Director
Current Status: Active PolicyStat ID: 2085666 Original Policy: 10/1986 Last Reviewed: 01/2016 Last Revised: 01/2016 Next Review: 01/2019 Owner: Policy Area: References: Applicability: Tracy Dodson: Director
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # Approved: 10/3/18
 Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
Enhanced BSL2 (BSL2+) Lab Policy IBC Policy # 150.1 Approved: 10/3/18 DIRECTIONS: All lab members must review this policy and sign/date the confirmation page at the end. I. GENERAL INFORMATION A. Institutional
TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee
 Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
Page 1 of 5 TEMPLE UNIVERSITY - Research Administration Institutional Biosafety Committee STANDARD OPERATING PROCEDURE SOP# 1.0 BIOSAFETY LEVEL 1 (BSL1) PROCEDURES A. Purpose This standard operating procedure
CHAPTER 13 PROCEDURE checklists
 PROCEDURE checklists 179 CHAPTER 13 PROCEDURE checklists Procedure 13-1 Handwashing 1. Gather needed supplies if not present at the handwashing area: soap or the cleansing agent specified by your facility,
PROCEDURE checklists 179 CHAPTER 13 PROCEDURE checklists Procedure 13-1 Handwashing 1. Gather needed supplies if not present at the handwashing area: soap or the cleansing agent specified by your facility,
Procedure 19 Changing A Clean Dressing. Procedure 20 Applying A Bandage. Procedure 21 Applying A Sterile Dressing
 Chapter 5 Wound Care Procedure 19 Changing A Clean Dressing Procedure 20 Applying A Bandage Procedure 21 Applying A Sterile Dressing Procedure 22 Applying A Dressing Around A Drain Procedure 23 Changing
Chapter 5 Wound Care Procedure 19 Changing A Clean Dressing Procedure 20 Applying A Bandage Procedure 21 Applying A Sterile Dressing Procedure 22 Applying A Dressing Around A Drain Procedure 23 Changing
PUBLIC HEALTH DEPARTMENT
 ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
ROBIN HODGKIN, M.P.A. Director STEPHEN W. MUNDAY, M.D., M.S. Health Officer COUNTY OF IMPERIAL PUBLIC HEALTH DEPARTMENT DIVISION OF ENVIRONMENTAL HEALTH 797 Main Street, Ste. B El Centro, CA 92243 Phone
DISINFECTION AND CLEANING
 1Disinfection and cleaning DISINFECTION AND CLEANING 9 Document version: 2v1.01 DISINFECTION AND CLEANING 1 PREPARING PROBES 1 CAUTION: Risk of allergic reaction. Some probe faces and probe sheaths contain
1Disinfection and cleaning DISINFECTION AND CLEANING 9 Document version: 2v1.01 DISINFECTION AND CLEANING 1 PREPARING PROBES 1 CAUTION: Risk of allergic reaction. Some probe faces and probe sheaths contain
rooo.lb IOWA COUNTY ORDINANCE NO TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS:
 .. rooo.lb IOWA COUNTY ORDINANCE NO. 4-196 TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS: SECTION I: The following ordinance of Iowa County, Wisconsin is hereby created
.. rooo.lb IOWA COUNTY ORDINANCE NO. 4-196 TATTOO ARTIST REGULATIONS THE IOWA COUNTY BOARD OF SUPERVISORS DO ORDAIN AS FOLLOWS: SECTION I: The following ordinance of Iowa County, Wisconsin is hereby created
BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN
 BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection Prevention
BODY ART FACILITY INFECTION PREVENTION AND CONTROL PLAN In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection Prevention
Hygienic requirements for tattoo and piercing studios
 Hygienic requirements for tattoo and piercing studios Activities injuring the skin or mucus membrane are linked to an increased infection risk for diseases transferred by blood and serum. To avoid transferable
Hygienic requirements for tattoo and piercing studios Activities injuring the skin or mucus membrane are linked to an increased infection risk for diseases transferred by blood and serum. To avoid transferable
Standard Operating Procedure for Biosafety Cabinet Use
 NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
NIPISSING UNIVERSITY ENVIRONMENTAL HEALTH AND SAFETY Standard Operating Procedure for Biosafety Cabinet Use PREPARED BY: DAVE VADNAIS JULY 27, 2016 R EVIEWED NOVEMBER 30, 2017 STANDARD OPERATING PROCEDURE
Updated by S. McNew, March Deborah Jung Microbiology Preparation Technician
 Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
Southeast Missouri State University PROTOCOL FOR SCIENCE EQUIPMENT USAGE AT REGIONAL CAMPUSES WITH EMPHASIS ON BS240/BS242 MICROORGANISMS AND THEIR HUMAN HOSTS Updated by S. McNew, March 2018 Personnel
STANDARDIZED CLINICAL PROTOCOLS. Sterilization Protocols. Aravind Eye Care System 1, Anna Nagar, Madurai , Tamilnadu, India
 STANDARDIZED CLINICAL PROTOCOLS Sterilization Protocols Aravind Eye Care System 1, Anna Nagar, Madurai - 625 020, Tamilnadu, India i.exe Standardized Sterilization Protocol STANDARDIZED STERILIZATION PROCEDURE
STANDARDIZED CLINICAL PROTOCOLS Sterilization Protocols Aravind Eye Care System 1, Anna Nagar, Madurai - 625 020, Tamilnadu, India i.exe Standardized Sterilization Protocol STANDARDIZED STERILIZATION PROCEDURE
Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions
 Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions 83.100 Health & Safety Definitions Clarity for licensee on health, safety & sanitation responsibilities
Texas Department of Licensing & Regulation Health & Safety Sanitation Standards Topic Definitions 83.100 Health & Safety Definitions Clarity for licensee on health, safety & sanitation responsibilities
CLEANING, SANITIZING, AND DISINFECTING
 CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
CLEANING, SANITIZING, AND DISINFECTING This section provides general information about cleaning, sanitizing, and disinfecting; guidelines for specific items commonly used in childcare and school settings;
Aseptic Milk Sampling for Bacteriology
 Aseptic Milk Sampling for Bacteriology Year Group: BVSc1 + Document number: CSL_F02 Equipment for this station: Equipment list: Cow model (fitted with milking udder) Wet wipes Paper towel Teat dip bottle
Aseptic Milk Sampling for Bacteriology Year Group: BVSc1 + Document number: CSL_F02 Equipment for this station: Equipment list: Cow model (fitted with milking udder) Wet wipes Paper towel Teat dip bottle
A ppendix 15 WUStL Bloodborne Pathogens Exposure Control Plan Research Laboratory-Specific Work Practices
 Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Specifc Work Practices Check List for Principal Investigators and Laboratory M anagers Discuss with staff tasks that involve handling of potentially infectious materials and how to perform such tasks in
Appendix C. Infectious Waste Guidelines
 Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
Appendix C. Infectious Waste Guidelines C.1 Infectious Waste Generation and Treatment, as required by Ohio Administrative Code (OAC) Section 3745-27, is registered with the Ohio Environmental Protection
INFECTION PREVENTION AND CONTROL PLAN
 INFECTION PREVENTION AND CONTROL PLAN FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: ( ) The owner, employees and practitioners of the above body art facility have developed
INFECTION PREVENTION AND CONTROL PLAN FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: ( ) The owner, employees and practitioners of the above body art facility have developed
INFECTION PREVENTION AND CONTROL PLAN (IPCP)
 INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
INFECTION PREVENTION AND CONTROL PLAN (IPCP) FACILITY NAME: FACILITY ID: ADDRESS: CITY: STATE: ZIP: OWNER S NAME: PHONE: CONTACT PERSON: EMAIL: The owner, employees and practitioners of the above body
under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC
 Page 1 of 7 under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC New Miana Pura East, Roras Road, Sialkot - Pakistan Tel: +92-52-3560135 Fax: +92-52-3563647 E-mail: info@longstoneintl.com
Page 1 of 7 under Council Medical Devices Directive (93/42/EEC) as updated directive 2007/47/EEC New Miana Pura East, Roras Road, Sialkot - Pakistan Tel: +92-52-3560135 Fax: +92-52-3563647 E-mail: info@longstoneintl.com
Bloodborne Pathogens
 Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
Bloodborne Pathogens This PowerPoint is designed to inform those who may be exposed to blood and other bodily functions how to prevent spreading, avoid exposure, and what to do if exposed to infectious
Please Read Before Applying General Directions: Note: You may want to print these for future use!
 Please Read Before Applying General Directions: Note: You may want to print these for future use! Page One: GENERAL DIRECTIONS Page Two: SPECIAL DIRECTIONS FOR CARPET AND UPHOLSTERY Page Three: SPECIAL
Please Read Before Applying General Directions: Note: You may want to print these for future use! Page One: GENERAL DIRECTIONS Page Two: SPECIAL DIRECTIONS FOR CARPET AND UPHOLSTERY Page Three: SPECIAL
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS
 SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
SHARPS MANAGEMENT AND DISPOSAL OF SHARPS, SYRINGES & CONTAMINATED PRODUCTS Purpose To ensure the safe disposal of potentially contaminated sharps, syringes, clothing and any other waste products. Scope
MEDICAL WASTE MANAGEMENT
 MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
MEDICAL WASTE MANAGEMENT Biological Safety INTRODUCTION PURPOSE Regulated medical waste is a designation for wastes that may contain pathogenic microorganisms which was previously termed infectious waste.
Laboratory Orientation. Biological Screening
 Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Laboratory Orientation Laboratory Orientation Safety Clean technique Reagent preparation Use of basic equipment Quality assurance : Laboratory Orientation 2 Safety National Forensic Science Technology
Bloodborne Pathogens Exposure Control Plan. December 2003
 Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Bloodborne Pathogens Exposure Control Plan December 2003 H://winfiles/safety/bloodborne pathogens/ofd Bloodborne Pathogens Plan.doc pg 2 PURPOSE: The purpose of this exposure control plan is to: 1. Eliminate
Preventing infection saves lives Prevent infection by keeping germs away Clean your hands and wear protective clothing...
 C hapter 5 Preventing infection In this chapter: Preventing infection saves lives...49 Infection is caused by germs...49 How germs get into the body...50 Prevent infection by keeping germs away...52 Clean
C hapter 5 Preventing infection In this chapter: Preventing infection saves lives...49 Infection is caused by germs...49 How germs get into the body...50 Prevent infection by keeping germs away...52 Clean
Roosevelt Biosafety Training. Created 10/2015
 Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Roosevelt Biosafety Training Created 10/2015 Objectives Identify risks and hazards in biological laboratories Understand biosafety levels for laboratories and the proper procedures for working in them
Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System. Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe
 Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Chapter 6 Phlebotomy Procedure 29 Performing A Venipuncture Procedure 30 Collecting A Blood Specimen Using The Vacuum-Tube System Procedure 31 Collecting A Blood Specimen Using A Needle And Syringe Procedure
Technical Information. Clorox Healthcare Bleach Germicidal Cleaner OVERVIEW. Clorox Healthcare Bleach Germicidal
 OVERVIEW Bleach Germicidal Cleaner is engineered as a ready-to-use sporicidal cleaner-disinfectant system for healthcare facilities. This EPA-registered disinfectant contains sodium hypochlorite and other
OVERVIEW Bleach Germicidal Cleaner is engineered as a ready-to-use sporicidal cleaner-disinfectant system for healthcare facilities. This EPA-registered disinfectant contains sodium hypochlorite and other
Biohazardous Waste. 1. Solid Biohazardous Waste (non-sharps) Storage
 Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Biohazardous Waste There are 4 general categories of biohazardous wastes based on the physical form of the waste. Each form must be segregated, identified, decontaminated and disposed of in an appropriate
Procedure/ Care Plan for Domiciliary Care Workers/ Support Workers - Application of Prescribed Creams/ Ointments/ Lotions (Adult)
 Application of Prescribed Creams/ Ointments/ Lotions (Adult) CLINICAL GUIDELINES ID TAG Medicines Management Specific Title: Procedure: Application of prescribed Creams/ Ointments/ Lotions (Adult) Author:
Application of Prescribed Creams/ Ointments/ Lotions (Adult) CLINICAL GUIDELINES ID TAG Medicines Management Specific Title: Procedure: Application of prescribed Creams/ Ointments/ Lotions (Adult) Author:
Safety Rules for Laboratory
 Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
Safety Rules for Laboratory These protocols are intended to protect you and make your laboratory experience enjoyable and productive. Section I: CVM General Laboratory Protocols (these rules apply to all
PLAN REVIEW APPLICATION PACKET BODY ART ESTABLISHMENTS
 PLAN REVIEW APPLICATION PACKET BODY ART ESTABLISHMENTS Greene County Public Health 360 Wilson Drive Xenia, OH 45385 (937) 374-5600 / (937) 374-5607 www.gcph.info Submit completed plan review packet, Infection
PLAN REVIEW APPLICATION PACKET BODY ART ESTABLISHMENTS Greene County Public Health 360 Wilson Drive Xenia, OH 45385 (937) 374-5600 / (937) 374-5607 www.gcph.info Submit completed plan review packet, Infection
HAND HYGIENE QUIZ. 1. Why is hand hygiene so important? (1 point) a. It is one of the single most effective measures for
 HAND HYGIENE QUIZ 1. Why is hand hygiene so important? (1 point) a. It is one of the single most effective measures for preventing the spread of infection. 2. Name five reasons why staff do not wash their
HAND HYGIENE QUIZ 1. Why is hand hygiene so important? (1 point) a. It is one of the single most effective measures for preventing the spread of infection. 2. Name five reasons why staff do not wash their
LESSON ASSIGNMENT. After completing this lesson, you should be able to:
 LESSON ASSIGNMENT LESSON 4 Procedures Used in Wound Care. LESSON ASSIGNMENT Paragraphs 4-1 through 4-14. LESSON OBJECTIVES After completing this lesson, you should be able to: 4-1. Identify the purposes
LESSON ASSIGNMENT LESSON 4 Procedures Used in Wound Care. LESSON ASSIGNMENT Paragraphs 4-1 through 4-14. LESSON OBJECTIVES After completing this lesson, you should be able to: 4-1. Identify the purposes
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN
 FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
FLORIDA GULF COAST UNIVERSITY DEPARTMENT OF REHABILITATION SCIENCES BIOSAFETY AND INFECTIOUS AGENTS CONTROL PLAN PURPOSE: This policy establishes minimum requirements for the handling, storage and disposal
VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers
 Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
Page 1 of 13 VGH Laboratory Guidelines Positive blood cultures from patients with suspect Ebola Virus Disease or other Viral Hemorrhagic Fevers Blood Culture technologist: 1. BACTEC FX signals positive
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET
 TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
TEN EASY STEPS FOR CLEANING A SPILL IN THE BIOSAFETY CABINET Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet For over 40 years, NuAire has been providing laboratory equipment that better enables
SUBCHAPTER 14H - SANITATION SECTION SANITATION
 SUBCHAPTER 14H - SANITATION SECTION.0100 - SANITATION 21 NCAC 14H.0101 COPY OF RULES TO COSMETOLOGY STUDENTS Cosmetic art schools shall give a copy of the sanitation rules governing the practice of the
SUBCHAPTER 14H - SANITATION SECTION.0100 - SANITATION 21 NCAC 14H.0101 COPY OF RULES TO COSMETOLOGY STUDENTS Cosmetic art schools shall give a copy of the sanitation rules governing the practice of the
Infection Control 101
 Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Infection Control 101 Infection Control Nosocomial vs. HAIs Standard Precautions/Body Substance Isolation (BSI) Protective environment to prevent HAIs PPE (latex precautions) Biohazard Waste Transmission-based
Bearing cleaning manual
 Bearing cleaning manual *you can click on the pictures for more info on them in the webshop. Step 1: Get your bearings out of your wheels by using the Bones Bearing Tool. This tool will prevent damage
Bearing cleaning manual *you can click on the pictures for more info on them in the webshop. Step 1: Get your bearings out of your wheels by using the Bones Bearing Tool. This tool will prevent damage
NATIONAL ELECTROLOGY PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN
 NATIONAL ELECTROLOGY PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information regarding
NATIONAL ELECTROLOGY PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information regarding
NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN
 NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information
NATIONAL WAX TECHNICIAN PRACTICAL EXAMINATION CANDIDATE INFORMATION BULLETIN Please visit www.nictesting.org for the most current bulletin prior to testing. This bulletin contains important information
Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5
 Environmental Health and Safety Manual Policy Number: EH&S 4-4 Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5 PURPOSE: To establish safe handling practices and use of
Environmental Health and Safety Manual Policy Number: EH&S 4-4 Title: Formaldehyde Safety Effective Date: 10/94 Revision: 2/97 Number of Pages: 5 PURPOSE: To establish safe handling practices and use of
Prepared by Laurel Arrigona, Matt Bavougian, Michael Crea, John Johnson, Steve Joyner, Sarah Robbin, and KC Stevenson
 122 nd AFDO Educational Conference Burlington, Vermont Body Art Committee June 10, 2018 Prepared by Laurel Arrigona, Matt Bavougian, Michael Crea, John Johnson, Steve Joyner, Sarah Robbin, and KC Stevenson
122 nd AFDO Educational Conference Burlington, Vermont Body Art Committee June 10, 2018 Prepared by Laurel Arrigona, Matt Bavougian, Michael Crea, John Johnson, Steve Joyner, Sarah Robbin, and KC Stevenson
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml)
 Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml) Important Information The device is a single-dose disposable pen. It
Instructions For Use PRALUENT (PRAHL-u-ent) (alirocumab) Injection, for Subcutaneous Injection Single-Dose Pre-Filled Pen (75 mg/ml) Important Information The device is a single-dose disposable pen. It
Hand Hygiene & PPE Policy
 Hand Hygiene & PPE Policy AIM This policy specifies Dragon s Daycare approach to effective hand hygiene practices and outlines best practice with regards to personal protective equipment (PPE). BACKGROUND
Hand Hygiene & PPE Policy AIM This policy specifies Dragon s Daycare approach to effective hand hygiene practices and outlines best practice with regards to personal protective equipment (PPE). BACKGROUND
ECU Radiation, Biosafety and Hazardous Substances Committee
 Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Standard Operating Procedure (SOP) Title (Samples Collected from Internal and External Agencies/Institutions) Note: As the infectious status of a patient s sample is unknown, precautions against exposure
Provide a brief description of the procedure and infectious organisms used:
 Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
Western Carolina University Standard Operating Procedure for the Safe Handling of Infectious Organisms at BSL-2 Containment Section 1. Contact Information Procedure Title: Procedure Author: Date of Creation/Revision:
LAB 5 Blood Collection
 LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
LAB 5 Blood Collection Purpose The Purpose of this lab is to learn how to collect a sample of blood properly. From this blood sample disease such as anemia, viral infections, iron deficiency, spherocytosis,
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College
 Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
Standard Operating Procedure for Blood Borne Infectious Disease Control Measures at Calvin College Clean up should be done by non-student employees and trained personnel only Cleaning Up BODY FLUIDS from
Infection Prevention Guidelines. Safe Use, Handling & Disposal of Sharps
 Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
Infection Prevention Guidelines Safe Use, Handling & Disposal of Sharps Author: Anne Tateson Owner: Infection Prevention Team Publisher: Infection Prevention Date of Version Issue: February 2015 Version:
Infection Prevention & Control Guidelines
 Infection Prevention & Control Guidelines for Foot Care Settings 2008 Contact Information Pierce County Antibiotic Resistance Task Force Infection Control and Prevention Committee Gwenda Felizardo, RN,
Infection Prevention & Control Guidelines for Foot Care Settings 2008 Contact Information Pierce County Antibiotic Resistance Task Force Infection Control and Prevention Committee Gwenda Felizardo, RN,
Volume 14. Ultrasound Imaging Accessories. Infection Prevention Transducer Storage Needle Guidance Equipment Protection
 Volume 14 Ultrasound Imaging Accessories WWW.CIVCO.COM Infection Prevention Transducer Storage Needle Guidance Equipment Protection SONOSITE GENERAL PURPOSE GUIDES Ultra-Pro II Reusable Starter Kits Each
Volume 14 Ultrasound Imaging Accessories WWW.CIVCO.COM Infection Prevention Transducer Storage Needle Guidance Equipment Protection SONOSITE GENERAL PURPOSE GUIDES Ultra-Pro II Reusable Starter Kits Each
Safety Office -- Laboratory Inspection Form
 RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
RESEARCH DIVISION Safety Office -- Laboratory Inspection Form NOTES: Satisfactory laboratory inspection is required prior to initiation of research New inspection required if Biosafety Level changes Annual
Introductory Chemistry
 Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
Introductory Chemistry Lab 1: Introduction and Safety Objectives Learn how work to safely in the chemical laboratory Learn when and how to use the safety equipment in the chemical laboratory Learn the
Figure A. Figure B To prevent premature activation of the needle safety guard, do not touch the NEEDLE GUARD ACTIVATION CLIPS at any time during use.
 INSTRUCTIONS FOR USE STELARA (stel ar a) (ustekinumab) injection, for subcutaneous use Instructions for injecting STELARA using a prefilled syringe. Read this Instructions for Use before you start using
INSTRUCTIONS FOR USE STELARA (stel ar a) (ustekinumab) injection, for subcutaneous use Instructions for injecting STELARA using a prefilled syringe. Read this Instructions for Use before you start using
Regulated Medical Waste. Be sure to sign in!
 Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Regulated Medical Waste Be sure to sign in! Waste Management Training You must receive this training if you: Add regulated medical waste into an accumulation container Determine if a material is regulated
Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing. drape the piercing site.
 FACT SHEET Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing Equipment / Single use towel 1. Client preparation A towel may be used to drape the piercing site. The towel should
FACT SHEET Table 5: Detailed Infection Prevention and Control Procedures for Body Piercing Equipment / Single use towel 1. Client preparation A towel may be used to drape the piercing site. The towel should
Muskogee Public Schools Custodial Procedures for Carpet Care
 CARPET CARE ENTRANCE MATS PURPOSE A. Exterior Mats 1. Remove sand and gravel from shoes prior to entering the building. 2. Minimize the amount of abrading material that will scratch and damage interior
CARPET CARE ENTRANCE MATS PURPOSE A. Exterior Mats 1. Remove sand and gravel from shoes prior to entering the building. 2. Minimize the amount of abrading material that will scratch and damage interior
Emergency Response and Biohazard Exposure Control Plan IBC Approved: 10/3/18
 Institutional Biosafety Committee Emergency Response and IBC Approved: 10/3/18 Table of Contents I. PURPOSE... 3 II. DEFINITIONS... 3 III. RESPONSIBILTIES... 4 IV. BIOHAZARDOUS SPILL EMERGENCY PREPAREDNESSS...
Institutional Biosafety Committee Emergency Response and IBC Approved: 10/3/18 Table of Contents I. PURPOSE... 3 II. DEFINITIONS... 3 III. RESPONSIBILTIES... 4 IV. BIOHAZARDOUS SPILL EMERGENCY PREPAREDNESSS...
Instructor Guide. Georgia Department of Technical and Adult Education. Decontamination and Infection Control
 Instructor Guide Georgia Department of Technical and Adult Education Decontamination and Infection Control Copyright October 2002 by. All rights reserved. No part of this manual may be reproduced or transmitted
Instructor Guide Georgia Department of Technical and Adult Education Decontamination and Infection Control Copyright October 2002 by. All rights reserved. No part of this manual may be reproduced or transmitted
CPTP COUNCIL FOR PIERCING AND TATTOO PROFESSIONALS
 To be completed by Tattoo / Body Piercing Establishment 1. Complete all applicable sections. 2. Sign and date the application. 3. Banking Details: FNB Olympus Account Name: SABATA Account nr: 622 697 745
To be completed by Tattoo / Body Piercing Establishment 1. Complete all applicable sections. 2. Sign and date the application. 3. Banking Details: FNB Olympus Account Name: SABATA Account nr: 622 697 745
CONSOLIDATION UPDATE: DECEMBER 11, 2002
 REPEALED BY THE BODY MODIFICATION BY-LAW NO. 40/2005 MARCH 23, 2005 (effective January 1, 2006) CONSOLIDATION UPDATE: DECEMBER 11, 2002 THE CITY OF WINNIPEG TATTOO STUDIO BY-LAW NO. 4653/87 A By-law of
REPEALED BY THE BODY MODIFICATION BY-LAW NO. 40/2005 MARCH 23, 2005 (effective January 1, 2006) CONSOLIDATION UPDATE: DECEMBER 11, 2002 THE CITY OF WINNIPEG TATTOO STUDIO BY-LAW NO. 4653/87 A By-law of
SUTTER COUNTY DEVELOPMENT SERVICES DEPARTMENT
 SUTTER COUNTY DEVELOPMENT SERVICES DEPARTMENT Building Inspection Planning Fire Services Road Maintenance Code Enforcement Environmental Health Engineering Water Resources SUMMARY OF THE SAFE BODY ART
SUTTER COUNTY DEVELOPMENT SERVICES DEPARTMENT Building Inspection Planning Fire Services Road Maintenance Code Enforcement Environmental Health Engineering Water Resources SUMMARY OF THE SAFE BODY ART
Biological Safety Training
 Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
Biological Safety Training Introduction to Biological Safety Biological Hazards are divided into 4 Biosafety Levels BSL 1 BSL 2 BSL 3 BSL4 Biosafety levels define the lab requirements, protective clothing,
List any references used for the procedure design (research publications, etc.):
 Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Western Carolina University Standard Operating Procedure for the Safe Handling of Animals A-BSL2 Containment Section 1. Contact Information Procedure Author: Date of SOP Creation/Revision: Name of Responsible
Green & Clean Program Nail Salon Application
 Green & Clean Program Nail Salon Application Name of Business Owner Name Manager Name Business Address Business Phone Number Secondary Phone Number Services Provided at Salon Hair styling (all types) Nails
Green & Clean Program Nail Salon Application Name of Business Owner Name Manager Name Business Address Business Phone Number Secondary Phone Number Services Provided at Salon Hair styling (all types) Nails
Airbrush Cleaning Manual. 1. Do's and Don'ts of Airbrush Cleaning
 Airbrush Cleaning Manual It's true of anything with working parts: proper upkeep means a longer life. One of the keys to keeping your airbrush in good working order is keeping it clean. Clean it after
Airbrush Cleaning Manual It's true of anything with working parts: proper upkeep means a longer life. One of the keys to keeping your airbrush in good working order is keeping it clean. Clean it after
PHARMACY PRACTICE I LAB PHCY 280L
 College of Pharmacy and Nursing School of Pharmacy PHARMACY PRACTICE I LAB PHCY 280L Lab Manual Fall 2017/18 Faculty: Ms Beena Jimmy Lecturer, Pharmacy Practice Name: Student Id: 1 Learning Objectives
College of Pharmacy and Nursing School of Pharmacy PHARMACY PRACTICE I LAB PHCY 280L Lab Manual Fall 2017/18 Faculty: Ms Beena Jimmy Lecturer, Pharmacy Practice Name: Student Id: 1 Learning Objectives
Spot Ex Endoscopic Tattoo. Take The N Ex t Step In The Fight Against Colon Cancer
 Spot Ex Endoscopic Tattoo Take The N Ex t Step In The Fight Against Colon Cancer Spot Ex Endoscopic Tattoo Take the next step in the fight against colon cancer Ex Ex Ex panded indications support adoption
Spot Ex Endoscopic Tattoo Take The N Ex t Step In The Fight Against Colon Cancer Spot Ex Endoscopic Tattoo Take the next step in the fight against colon cancer Ex Ex Ex panded indications support adoption
MASSACHUSETTS COSMETOLOGY FREQUENTLY ASKED QUESTIONS
 MASSACHUSETTS COSMETOLOGY FREQUENTLY ASKED QUESTIONS FREQUENTLY ASKED QUESTIONS CONCERNING TASKS We are aware that you may have questions concerning the State Board Examinations. Please check this FAQ
MASSACHUSETTS COSMETOLOGY FREQUENTLY ASKED QUESTIONS FREQUENTLY ASKED QUESTIONS CONCERNING TASKS We are aware that you may have questions concerning the State Board Examinations. Please check this FAQ
Instructions for Use: Decontamination Gown
 Instructions for Use: Decontamination Gown Brand Name of Product Decontamination Gown Generic Name of Product Decontamination Gown Product Code Number(s) MN110 SMMD, MN110 LGXL, MN110 2X3X, MN110 4X Intended
Instructions for Use: Decontamination Gown Brand Name of Product Decontamination Gown Generic Name of Product Decontamination Gown Product Code Number(s) MN110 SMMD, MN110 LGXL, MN110 2X3X, MN110 4X Intended
ENVIRONMENTAL HEALTH SERVICE REQUEST FORM 2019
 Environmental Health Division 1675 W. Garden of the Gods Rd., Suite 2044 Colorado Springs, CO 80907 (719) 578-3199 phone (719) 578-3188 fax www.elpasocountyhealth.org ENVIRONMENTAL HEALTH SERVICE REQUEST
Environmental Health Division 1675 W. Garden of the Gods Rd., Suite 2044 Colorado Springs, CO 80907 (719) 578-3199 phone (719) 578-3188 fax www.elpasocountyhealth.org ENVIRONMENTAL HEALTH SERVICE REQUEST
Original Date:
 Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
Title: Sharps Safety Index Number: (Func. - Categ. - Sr.No.) Function: Facility Management and Safety Category: Safety Scope of application: All Departments/Units/ Sections Original Date: 06.08.2008 Next
Technical Rescue-Personal Protective Equipment
 3.5.1.1 Technical Rescue-Personal Protective Equipment YOUR ORGANIZATION STANDARD OPERATING PROCEDURES/GUIDELINES TITLE: Technical Rescue-Personal Protective Equipment NUMBER: 3.5.1.1 PREPARED BY: SECTION/TOPIC:
3.5.1.1 Technical Rescue-Personal Protective Equipment YOUR ORGANIZATION STANDARD OPERATING PROCEDURES/GUIDELINES TITLE: Technical Rescue-Personal Protective Equipment NUMBER: 3.5.1.1 PREPARED BY: SECTION/TOPIC:
Focus on Quality Care Product Resource Guide
 Focus on Quality Care Product Resource Guide ChloraPrep Skin Antiseptic A complete line of skin preparation products to help reduce microorganisms on the skin that can cause infection. Every patient, every
Focus on Quality Care Product Resource Guide ChloraPrep Skin Antiseptic A complete line of skin preparation products to help reduce microorganisms on the skin that can cause infection. Every patient, every
2.6-1 SCIENCE EXPERIMENTS ON FILE Revised Edition. Cloud Chamber
 2.6-1 SCIENCE EXPERIMENTS ON FILE Revised Edition Cloud Chamber Topic Cloud formation Time 1 hour! Safety Please click on the safety icon to view safety precautions. Do not touch dry ice with bare hands.
2.6-1 SCIENCE EXPERIMENTS ON FILE Revised Edition Cloud Chamber Topic Cloud formation Time 1 hour! Safety Please click on the safety icon to view safety precautions. Do not touch dry ice with bare hands.
BlephEx...A Revolutionary New Treatment for Blepharitis. Owner s Manual
 BlephEx...A Revolutionary New Treatment for Blepharitis Owner s Manual Table of Contents Warnings...2 Intended Use...2 Instructions for Use...3 Cleaning Instructions...8 Maintenance Details...8 Safety
BlephEx...A Revolutionary New Treatment for Blepharitis Owner s Manual Table of Contents Warnings...2 Intended Use...2 Instructions for Use...3 Cleaning Instructions...8 Maintenance Details...8 Safety
This bulletin gives specific instructions for application of Avery Dennison translucent film to recommended flexible-face signage material.
 Application of Avery Dennison Translucent Pressure Sensitive Films to Avery Dennison Approved, Flexible-Faced Signage Material #4.01 (Revision 10) Dated: 12/31/14 1.0 Scope This bulletin gives specific
Application of Avery Dennison Translucent Pressure Sensitive Films to Avery Dennison Approved, Flexible-Faced Signage Material #4.01 (Revision 10) Dated: 12/31/14 1.0 Scope This bulletin gives specific
